Acyl protein thioesterase inhibitors as probes of dynamic S-palmitoylation
Abstract
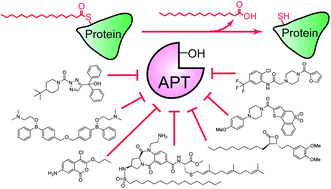
Protein palmitoylation describes the hydrophobic post-translational modification of cysteine residues in certain proteins, and is required for the spatial organization and composition of cellular membrane environments. Certain palmitoylated proteins are processed by acyl protein thioesterase (APT) enzymes, which catalyze thioester hydrolysis of palmitoylated cysteine residues. Inhibiting APT enzymes disrupts Ras trafficking and attenuates oncogenic growth signaling, highlighting these enzymes as potential therapeutic targets. As members of the serine hydrolase enzyme family, APT enzymes can be assayed by fluorophosphonate activity-based protein profiling (ABPP) methods, allowing rapid profiling of inhibitor selectivity and potency. In this review, we discuss recent progress in the development of potent and selective inhibitors to APT enzymes, including both reversible and irreversible inhibitor chemotypes. These examples highlight how ABPP methods integrate with medicinal chemistry for the discovery and optimization of inhibitors in complex proteomes.

 Please wait while we load your content...
Please wait while we load your content...