Dissolution without disappearing: multicomponent gas exchange for CO2 bubbles in a microfluidic channel†
Abstract
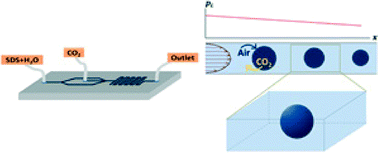
We studied the dissolution dynamics of CO2 gas bubbles in a microfluidic channel, both experimentally and theoretically. In the experiments, spherical CO2 bubbles in a flow of a solution of sodium dodecyl sulfate (SDS) first shrink rapidly before attaining an equilibrium size. In the rapid dissolution regime, the time to obtain a new equilibrium is 30 ms regardless of SDS concentration, and the equilibrium radius achieved varies with the SDS concentration. To explain the lack of complete dissolution, we interpret the results by considering the effects of other gases (O2, N2) that are already dissolved in the aqueous phase, and we develop a multicomponent dissolution model that includes the effect of surface tension and the liquid pressure drop along the channel. Solutions of the model for a stationary gas bubble show good agreement with the experimental results, which lead to our conclusion that the equilibrium regime is obtained by gas exchange between the bubbles and liquid phase. Also, our observations from experiments and model calculations suggest that SDS molecules on the gas–liquid interface form a diffusion barrier, which controls the dissolution behaviour and the eventual equilibrium radius of the bubble.

 Please wait while we load your content...
Please wait while we load your content...