An XPS/AES comparative study of the surface behaviour of nano-silicon anodes for Li-ion batteries†
Abstract
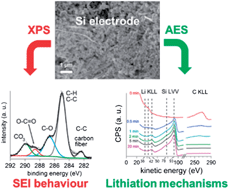
In this paper, the surface of composite electrodes made of nano-silicon is studied by using two surface characterization techniques: X-ray Photoelectron Spectroscopy (XPS) and Auger Electron Spectroscopy (AES). Several electrodes were analyzed at different States Of Charge (SOC) during the first electrochemical cycle. Firstly, the Solid Electrolyte Interphase (SEI) as well as the Si oxides present at the particle surface were thoroughly investigated. The behaviour of the SEI is close to that which forms on graphite: (i) its composition is similar and (ii) its thickness is greater at the lithiated state. Besides, our AES analyses carried out at the end of the first electrochemical cycle demonstrate that this behaviour strongly depends on the material it forms on: silicon or carbon fibers. Concerning SiO2, which is initially present at the Si particles' surface, it is reduced irreversibly during the first lithiation. Simultaneously, our results clearly show the formation of Li2O and LixSiOy; these species are still observed during the aging of the electrode and systematically react at each cycle. Secondly, by using AES combined with a sputtering technique, the first lithium insertion and the associated LixSi alloys were studied: several Si particles were investigated individually with respect to their electrode environment. After a capacity of 360 mA h g−1, the estimated Li concentration is different from one Si particle to another indicating that the first lithiation is a strongly heterogeneous process.

 Please wait while we load your content...
Please wait while we load your content...