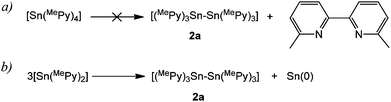Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Direct synthesis of the Janus-head ligand (MePy)3Sn–Sn(MePy)3 using an unusual pyridyl-transfer reaction (MePy = 6-methyl-2-pyridyl)†
Raúl
García-Rodríguez
* and
Dominic S.
Wright
Chemistry Department, Cambridge University, Lensfield Road, Cambridge, CB2 1EW, UK. E-mail: rg489@cam.ac.uk
First published on 6th August 2014
Abstract
The in situ reaction of the tripodal anion [EtAl(MePy)3]− (MePy = 6-Me-2-py) with SnCl2 gives the Janus-head ligand (MePy)3Sn–Sn(MePy)3, containing a Sn–Sn bond, through a novel, one-pot reaction involving a combination of pyridyl transfer and metal-oxidation.
Neutral tris(pyridyl) ligands of the general type [Y(Py)3] (Py = 2-pyridyl, where Y can be a range of non-metallic bridgehead atoms or groups (e.g. CR, COR, CH, N, P, P = O, As; Fig. 1a)), have been studied extensively in the last 30 years.1 These ligands and the related tris(pyrazolyl)borates and methanes2,3 have found a broad range of applications in organometallic, coordination and bioinorganic chemistry. Their applications include catalysis, stoichiometric organic transformations and as models for the coordination in enzymatic metal sites.4 However, it is only relatively recently that attention has turned to Group 13 and 14 congeners containing heavier, more metallic main group bridgeheads.5 These metal-based ligands introduce the possibility of redox activity and variable oxidation states at the bridgehead, as well as previously unknown anionic tris(pyridyl) ligands,6 such as tris(2-pyridyl)aluminates of the type shown in Fig. 1b.
 | ||
| Fig. 1 (a) Connectivity of tris(2-pyridyl) ligand systems containing bridgehead atoms or groups (Y) and (b) the anionic tris(2-pyridyl)aluminate ligands. | ||
In addition to the interesting coordination characteristics of this class of ligands revealed by a range of studies in the past 10 years,7 another potentially useful property of metal-based tris(pyridyl) arrangements is their ability to act as ‘soft’ pyridyl transfer reagents.5a,6 This is the result of the relatively low polarity of the C–Al bond, which is roughly comparable to the C–Zn bond. Unlike the Zn counterparts, however, 2-pyridyl-aluminium compounds are thermodynamically stable at room temperature and are therefore storable reagents. This soft nucleophilic behaviour was first suggested by the reaction of the lithium complex [MeAl(Py)3Li·THF] with CuCl, which gives the organo-CuI trimer [Cu(Py)]3. In contrast, direct reaction of (2-Li-py) with CuCl results only in decomposition.6
Here we illustrate a further new facet of the reactivity of tris(2-pyridyl) reagents, with the discovery that the reaction of [EtAl(MePy)3]Li·THF (1a) (MePy = 6-Me-2-pyridyl) with SnIICl2 gives unique access to the Janus-head ligand system (MePy)3Sn–Sn(MePy)3 (2a) in a single step, a reaction that involves a combination of 2-pyridyl ligand transfer and oxidation of the SnII centre (Scheme 1). The significance of this new reaction is that it has provided the simplest access yet to a donor-functionalised Sn–Sn bonded framework of this type. For example, the most obvious alternative precursors to 2a are the hexahalides Sn2X6 (X = halogen); however, these are difficult to prepare and are either highly unstable (X = Cl)8 or mixed valence compounds (X = F).9
The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reaction of [EtAl(MePy)3]Li·THF (1a) with SnCl2 at room temperature in toluene–THF affords an orange reaction mixture containing several unidentified pyridyl compounds (see the discussion below and Fig. S6 of the ESI†) from which colourless crystals of [(MePy)3Sn–Sn(MePy)3] (2a) were isolated in a total yield up to 26%. A finely-divided black precipitate of the Sn metal was also visible upon prolonged storage of the reaction mixture. Compound 2a was fully characterized by analytical and spectroscopic methods and its structure was determined by single-crystal X-ray diffraction. Particularly diagnostic are its 119Sn NMR spectrum (singlet δ −218.9 ppm) and the presence of 117,119Sn satellites for all of the C-atoms of the MePy ligands in the 13C{1H} NMR spectrum (see Fig. S1–S5, ESI†). The single-crystal X-ray analysis of 2a (Fig. 2) confirms the formation of a Sn–Sn single bond in this reaction, the observed Sn–Sn bond length [2.7183(6) Å] being at the short end of the range of hexa-organo di-tin compounds [R3Sn–SnR3].10
1 reaction of [EtAl(MePy)3]Li·THF (1a) with SnCl2 at room temperature in toluene–THF affords an orange reaction mixture containing several unidentified pyridyl compounds (see the discussion below and Fig. S6 of the ESI†) from which colourless crystals of [(MePy)3Sn–Sn(MePy)3] (2a) were isolated in a total yield up to 26%. A finely-divided black precipitate of the Sn metal was also visible upon prolonged storage of the reaction mixture. Compound 2a was fully characterized by analytical and spectroscopic methods and its structure was determined by single-crystal X-ray diffraction. Particularly diagnostic are its 119Sn NMR spectrum (singlet δ −218.9 ppm) and the presence of 117,119Sn satellites for all of the C-atoms of the MePy ligands in the 13C{1H} NMR spectrum (see Fig. S1–S5, ESI†). The single-crystal X-ray analysis of 2a (Fig. 2) confirms the formation of a Sn–Sn single bond in this reaction, the observed Sn–Sn bond length [2.7183(6) Å] being at the short end of the range of hexa-organo di-tin compounds [R3Sn–SnR3].10
Two obvious mechanisms might be responsible for the formation of the Sn–Sn bonded compound 2a, involving reductive elimination or disproportionation of SnII/IV intermediates. An in situ multinuclear NMR study of the reaction of an equimolar mixture of 1a with SnCl2 in d8-THF was used to gain insights into the mechanism by which 2a is formed. Within 15 min of mixing the reactants at ambient temperature an orange colouration developed and 2a was observed as the only Sn-containing species formed. The 27Al NMR spectrum showed that a characteristic sharp resonance for AlCl4− was also present (δ 103 ppm),11 together with other unidentified Al-containing species. No further changes in the 27Al or 119Sn spectra were observed after 24 h. At this stage, the 1H NMR spectrum confirmed that no precursor 1a remained. Importantly, no 6,6′-dimethyl-2,2′-dipyridyl is observed in the reaction at any point, despite the presence of several unidentified pyridyl signals (see Fig. S6 and S7, ESI†), ruling out a possible reductive elimination pathway to 2a which involves the SnIV intermediate Sn(MePy)4 (e.g., Scheme 2a). Confirming this, Sn(MePy)4 is not observed in the 119Sn NMR spectrum (lit. value δ −313 ppm in CDCl3)12 and we also find that heating an authentic sample of Sn(Py)4 at 80 °C in toluene for 5 days results in only negligible decomposition (Fig. S8, ESI†).
In a separate experiment, SnIICl2 was treated with 2 equivalents of 2-Li-MePy at −78 °C and the mixture was allowed to warm to room temperature. After 1 h, a mixture of at least five Sn species was formed, with the presence of 2a being apparent in both 1H and 119Sn NMR spectra (Fig. S9, ESI†). This observation provided initial evidence that a disproportionation pathway involving a 2-pyridyl SnII species (of the type [Sn(MePy)n]x−) is likely in the formation of 2a (e.g., Scheme 2b), which is also consistent with the formation of the Sn metal in the reaction.
The equimolar reaction of [EtAl(Py)3Li·THF] (1b) (containing unsubstituted 2-pyridyl groups, Py) with SnCl2 in d8-THF was also investigated in order to determine the substrate-dependent behavior of this reaction. An in situ119Sn NMR spectroscopic investigation shows that three major Sn-containing products are generated after 24 h (Fig. S10, ESI†), an unidentified species at δ −130 ppm (s), [(Py)3Sn–Sn(Py)3] (2b) [assigned on the basis of its chemical shift, δ −218.6 ppm (s)] and another species (3) observed as two singlets (δ −264.5 and −348.3 ppm). The in situ NMR spectroscopic studies therefore indicate that a similar reactivity occurs for the unsubstituted 2-pyridyl groups, but the reaction becomes slower and less selective.
In both the in situ NMR spectroscopic investigations and on the preparative scale, 3 precipitates cleanly from the reaction mixture. The single-crystal X-ray structural analysis shows that 3 is the heterometallic SnII/AlIII complex [{Sn(Py)3}Al{(μ-Cl)(Py)2Sn(Cl)}] (Fig. 3), containing a six-coordinate AlIII centre that is complexed by a monoanionic [SnII(Py)3]− and a dianionic [(Py)2SnIICl2]2− ligand in a sandwich-type arrangement. The presence of pyramidal and trigonal bipyramidal geometries at the two Sn centres (with stereochemically-active metal lone pairs) strongly supports the oxidation state formulation of the complex as SnII/AlIII/SnII. The structure of 3 is consistent with the previously mentioned 119Sn NMR spectrum, with the resonance at δ −266.3 being the SnII centre of the [SnII(Py)3]− ligand,13 while the resonance at δ −347.5 corresponds to the [(Py)2SnIICl2]2− ligand.14,15 Interestingly, there is also a very large change in chemical shift in the 27Al NMR spectrum on reaction of the precursor [EtAl(Py)3Li·THF] (1b) (δ 125) with the product 3 (δ 24), signifying the increase in the metal coordination number from four to six (Fig. S11–S13, ESI†).11b,16
Viewed as a model intermediate, 3 provides a snapshot of the likely stages of the reactions producing 2a and 2b. Although the precise details of the mechanism remain elusive at this stage, the key structural components of 3 provide further support for the conclusion that the formation of 2a and 2b involves (i) initial transfer of the 2-pyridyl groups from AlIII to SnII and (ii) the disproportionation of the resulting 2-pyridyl SnII complex into 2a and 2b (as concluded earlier in regard to Scheme 2b).
In summary, we have demonstrated in this paper a new feature of the reactivity of tris(2-pyridyl) aluminate anions, their ability to act as 2-pyridyl transfer reagents in combination with oxidation of metal centres. The formation of (MePy)3Sn–Sn(MePy)3 (2a) in a one-pot reaction avoids the use of highly unstable or poorly defined SnIV hexa-halides and may have extensive applications elsewhere in the p-block.17 Furthermore, the Janus-head ligand properties of 2a and related Group 14 arrangements should be of great interest in their own right. Future studies will be aimed at assessing the use of this new type of reaction for the synthesis of other main group Py′nM-MPy′n arrangements (Py′ = a substituted or unsubstituted pyridyl ligand) and the investigation of their coordination chemistry.
Experimental
All manipulations were carried out under dry, O2-free argon on a vacuum line. Compounds 1a and 1b were synthesized as described previously.7c,eSynthesis of 2a
To a Schlenk tube were added 1a (200 mg, 0.49 mmol) and SnCl2 (93 mg, 0.49 mmol) in a glove box. The Schlenk tube was transferred to a vacuum line, and toluene (15 mL) and THF (5 mL) were added. After the addition of THF a solution was obtained which quickly changed from pale yellow to deep orange. The mixture was stirred for 36 h at room temperature and then filtered through celite to remove a small amount of precipitate and an oily residue. The majority of the solvent was removed in vacuo until the precipitation of a solid was observed, which was heated gently back into solution. Storage at −15 °C (24 h) afforded a few colourless crystals of 2a along with an orange residue. The supernatant was transferred using a cannula to another Schlenk tube, concentrated and stored at −15 °C (48 h) affording colourless crystals of 2a. Total yield up to 26% (51 mg, 0.0645 mmol). Elem. anal. calc. for 2a: C: 54.7, H: 4.6, N: 10.6, found C: 54.4, H: 4.7, N: 10.2. 1H NMR (298 K, 500 MHz, thf-d8), δ = 7.68 (m, 6H, C(3)–H py), 7.36 (m, 6H, C(4)–H py), 6.94 (m, 6H, C(5)–H py), 2.39 (s, 18H, C(6)–Me). 13C{1H} NMR (298 K, 125.7 MHz, thf-d8), δ = 173.47 (C(2), 1J(13C–119/117Sn) = 550/525 Hz, 2J(13C–119/117Sn) = 74/71 Hz), 158.77 (C(6) 3J(13C–119/117Sn) = 76/73 Hz, 4J(13C–119/117Sn) = 5 Hz), 134.16 (C(4) 3J(13C–119/117Sn) = 38/36 Hz, 4J(13C–119/117Sn) = 3 Hz), 131.82 (C(3) 2J(13C–119/117Sn) = 97/93 Hz, 3J(13C–119/117Sn) = 9 Hz), 122.03 (C(5) 4J(13C–119/117Sn) = 12 Hz), 24.43 (C(6)–Me, 4J(13C–119/117Sn) = 4 Hz. 119Sn{1H} NMR (186.5 MHz, 298 K, rel. solution of SnMe4), δ = −218.7 [s(ν1/2 = 52 Hz)]. No 119Sn–117Sn coupling could be detected.Synthesis of 3
To a Schlenk tube were added 1b (181 mg, 0.49 mmol) and SnCl2 (93 mg, 0.49 mmol) in a glove box. The Schlenk tube was transferred to a vacuum line, and toluene (15 mL) and THF (5 mL) were added. After the addition of THF a yellow mixture was obtained which was stirred at room temperature for 40 h. A cloudy orange mixture was obtained. The supernatant was removed with a cannula and the resulting orange solid was dried in vacuo. Even though this orange solid was only partially soluble in THF, a few colourless crystals of 3 could be obtained from a concentrated sample in THF at room temperature. 1H NMR (298 K, 500 MHz, thf-d8), δ = 8.78 (d (6 Hz), 2H), 8.58 (d (7 Hz), 2H), 8.18 (d (7 Hz), 2H), 7.62–7.55 (m, 3H), 7.49 (t (7 Hz), 2H), 7.15 (m, 1H, overlapped with toluene resonances), 7.04 (t (7 Hz), 2H), 6.88 (d (6 Hz), 2H), 6.81 (t (7 Hz), 1H), 6.55 (t (7 Hz), 1H). 119Sn{1H} NMR (186.5 MHz, 298 K, rel. solution of SnMe4), δ = −264.5 (br, s), −348.3 (br, s). Changes up to 2 ppm were observed for the chemical shift. 27Al NMR (298 K, d8-thf-d8, 130.3 MHz, rel. solution of AlCl3·6H2O–D2O), δ = 23.8 (ν1/2 = 210 Hz).Crystal data: The data were collected on a Bruker SMART X2S diffractometer using a monochromatic Mo-Kα microfocus source with a BREEZE CCD detector, and equipped with an Oxford Cryosystem DeskTop cooler at 200 K. Data were collected using Bruker GIS, processed using Bruker SAINT and were refined using the SHELXTL-97 suite of programs. CCDC 1008866 and 1008867 contain the supplementary crystallographic data for 2a and 3, respectively.
R.G.-R. acknowledges the EU for a Marie Curie Intra European Fellowship and D.S.W. acknowledges the EU for an ERC Advanced Investigator Grant.
Notes and references
- L. F. Szczepura, L. M. Witham and K. J. Takeuchi, Coord. Chem. Rev., 1998, 174, 5 CrossRef CAS.
- (a) S. Trofimenko, Chem. Rev., 1993, 93, 943 CrossRef CAS; (b) D. L. Reger, Comments Inorg. Chem., 1999, 21, 1 CrossRef CAS.
- S. Trofimenko, Scorpionates: The Coordination Chemistry of Polypyrazolylborate Ligands, Imperial College Press, London, 1999 Search PubMed.
- (a) G. Parkin, Adv. Inorg. Chem., 1995, 42, 291 CrossRef CAS; (b) E. L. Hegg, R. Y. N. Ho and L. Que Jr., J. Am. Chem. Soc., 1999, 121, 1972 CrossRef CAS; (c) N. Kitajima and W. B. Tolman, Prog. Inorg. Chem., 1995, 43, 419 CrossRef CAS.
- (a) H. R. Simmonds and D. S. Wright, Chem. Commun., 2012, 48, 8617 RSC; (b) D. Morales, J. Perez, L. Riera, V. Riera and D. Miguel, Organometallics, 2001, 20, 4517 CrossRef CAS; (c) K. Zeckert, S. Zahn and B. Kirchner, Chem. Commun., 2010, 46, 2638 RSC; (d) K. Zeckert, J. Griebel, R. Kirmse, M. Weiss and R. Denecke, Chem. – Eur. J., 2013, 19, 7718 CrossRef CAS PubMed.
- F. Garcia, A. D. Hopkins, R. A. Kowenicki, M. McPartlin, M. C. Rogers and D. S. Wright, Organometallics, 2004, 23, 3884 CrossRef CAS.
- (a) C. S. Alvarez, F. Garcia, S. M. Humphrey, A. D. Hopkins, R. A. Kowenicki, M. McPartlin, R. A. Layfield, R. Raja, M. C. Rogers, A. D. Woods and D. S. Wright, Chem. Commun., 2005, 198 RSC; (b) F. Garcia, A. D. Hopkins, R. A. Kowenicki, M. McPartlin, M. C. Rogers, J. S. Silvia and D. S. Wright, Organometallics, 2006, 25, 2561 CrossRef CAS; (c) T. H. Bullock, W. T. K. Chan, D. J. Eisler, M. Streib and D. S. Wright, Dalton Trans., 2009, 1046 RSC; (d) T. H. Bullock, W. T. K. Chan and D. S. Wright, Dalton Trans., 2009, 6709 RSC; (e) R. García-Rodríguez, T. H. Bullock, M. McPartlin and D. S. Wright, Dalton Trans., 2014, 43, 14045–14053 RSC.
- E. Wiberg and H. Behringer, Z. Anorg. Allg. Chem., 1964, 329, 290 CrossRef CAS.
- (a) T. Birchall, J. P. Johnson and V. Manivannan, Hyperfine Interact., 1986, 28, 685 CrossRef CAS; (b) P. E. Lippens, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 60, 4576 CrossRef CAS.
- (a) M. Saito, R. Haga and M. Yoshioka, Chem. Commun., 2002, 1002 RSC; (b) H. Puff, B. Breuer, G. Gehrke-Brinkmann, P. Kind, H. Reuter, W. Schuh, W. Wald and G. Weidenbrueck, J. Organomet. Chem., 1989, 363, 265 CrossRef CAS.
- (a) K. Knabel and H. Noeth, Z. Naturforsch., B: Chem. Sci., 2005, 60, 1027 CAS; (b) G. E. Tranter, J. L. Holmes and J. C. Lindon, Encyclopedia of spectroscopy and spectrometry, San Diego, Calif, London, 2000, vol. 1, pp. 696–700 Search PubMed.
- M. Bette and D. Steinborn, Phosphorus, Sulfur Silicon Relat. Elem., 2012, 187, 1271 CrossRef CAS.
- K. Zeckert, Dalton Trans., 2012, 41, 14101 RSC.
- V. Chandrasekhar, S. Nagendran and V. Baskar, Coord. Chem. Rev., 2002, 235, 1 CrossRef CAS.
- A. G. Davies and I. Wiley, Tin chemistry: fundamentals, frontiers, and applications, John Wiley & Sons, Ltd, 2008, p. 17–283 Search PubMed.
- (a) R. Benn, A. Rufinska, H. Lehmkuhl, E. Janssen and C. Krueger, Angew. Chem., Int. Ed. Engl., 1983, 95, 808 CrossRef CAS; (b) J. M. Bakker, L. J. Barbour, G. B. Deacon, P. C. Junk, G. O. Lloyd and J. W. Steed, J. Organomet. Chem., 2010, 695, 2720 CrossRef CAS; (c) R. Benn, E. Janssen, H. Lehmkuhl and A. Rufinska, J. Organomet. Chem., 1987, 333, 155 CrossRef CAS.
- It can be noted that a similar Pb-compound [(6-tBuO-Py)3PbPb(6-tBuO-Py)3] has been obtained using oxidative coupling of the plumbate ion [(6-tBuO-Py)3Pb]− in the presence of Eu(II) (see ref. 5d). This reaction differs fundamentally from the current study in that no additional oxidising metal is required in the formation of 2a.
Footnote |
| † Electronic supplementary information (ESI) available: Additional figures and tables and NMR spectra. CCDC 1008866 and 1008867. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4dt02216e |
| This journal is © The Royal Society of Chemistry 2014 |