Isomorphous substitution of divalent metal ions in layered double hydroxides through a soft chemical hydrothermal reaction†
Abstract
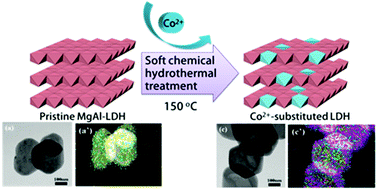
We have successfully incorporated Co2+ ions into layered double hydroxides (LDHs) comprising Mg and Al hydroxides via isomorphous substitution utilizing a soft chemical hydrothermal reaction. The inductively coupled plasma-atomic emission spectroscopy (ICP-AES) analysis showed that the incorporation of Co2+ into an LDH was highly dependent on the dissolution of Mg2+. The X-ray diffraction (XRD) patterns showed that the crystalline phase, as well as the crystallinity of pristine LDH, was well preserved without the evolution of impurities during the substitution reaction. It was notable that the size (∼250 nm) and hexagonal plate-like morphology of LDHs did not change significantly upon Co2+ substitution. Transmission electron microscopy-energy dispersive spectroscopy (TEM-EDS) exhibited homogeneous distribution of Co2+ in the LDH particles obtained by this substitution reaction. Solid-state UV-vis and X-ray absorption spectroscopy (XAS) verified that the incorporated Co2+ ions were well stabilized in the octahedral sites of an LDH, which were formerly occupied by Mg2+ ions.
- This article is part of the themed collection: Layered Inorganic Solids

 Please wait while we load your content...
Please wait while we load your content...