The role of the central Fe atom in the N4-macrocyclic structure for the enhancement of oxygen reduction reaction in a heteroatom nitrogen–carbon nanosphere†
Abstract
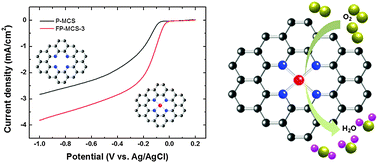
In the present work, we demonstrated the significance of a central transition metal, Fe, in a N4-macrocycle for the enhancement of ORR activity and other electrochemical properties. The catalysts were synthesized by a solution plasma process. Fe–phthalocyanine/benzene and phthalocyanine/benzene were chosen as the precursors of Fe–phthalocyanine based mesoporous carbon (FP-MCS) and phthalocyanine based mesoporous carbon (P-MCS) catalysts, respectively. The existence of Fe–N4 and N4 macrocyclic structures was confirmed by X-ray photoelectron spectroscopy. From the chemical bonding structure, FP-MCS demonstrated that the Me–N peaks increased as the amount of iron–phthalocyanine introduced in the experiment increased. The dominant active site was shifted from pyridinic nitrogen to Me–N when iron–phthalocyanine was present. The analysis of Tof-SIMS indicated that the relative intensity of FeN4Cy+ ions was approximately 50% of the total amount of ionized species of ∑FeNxCy+. Both XPS and Tof-SIMS results confirmed that the Fe–N4 site was the most favourable structure in the matrix. From CV measurements, the cathodic peak current corresponding to ORR activity slightly shifted from −0.19 V to −0.17 V when the active site changed from N4 to Fe–N4 macrocyclic structure. The current density increased more than 30% in the presence of iron. Based on the calculation of Koutecky–Levich plots, the electron transfer numbers for ORR reaction in P- and FP-MCSs were 3.25 and 3.98, respectively. These results clearly demonstrated that the presence of a Fe central ion in the N4-macrocyclic structure significantly enhanced the ORR and charge transfer number in ORR activities.

 Please wait while we load your content...
Please wait while we load your content...