Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Competition between excited state proton and OH− transport via a short water wire: solvent effects open the gate†
Gül
Bekçioğlu
ab,
Christoph
Allolio
b,
Maria
Ekimova
c,
Erik T. J.
Nibbering
c and
Daniel
Sebastiani
*b
aPhysics Department, Freie Universität Berlin, Arnimallee 14, 14195 Berlin, Germany
bInstitut für Chemie, Martin-Luther-Universität Halle-Wittenberg, Von-Danckelmann-Platz 4, 06120 Halle (Saale), Germany. E-mail: daniel.sebastiani@chemie.uni-halle.de
cMax Born Institut für Nichtlineare Optik und Kurzzeitspektroskopie, Max Born Strasse 2A, D-12489 Berlin, Germany
First published on 16th April 2014
Abstract
We investigate the acid–base proton exchange reaction in a microsolvated bifunctional chromophore by means of quantum chemical calculations. The UV/vis spectroscopy shows that equilibrium of the keto- and enol-forms in the electronic ground state is shifted to the keto conformation in the excited state. A previously unknown mechanism involving a hydroxide ion transport along a short water wire is characterized energetically, which turns out to be competitive with the commonly assumed proton transport. Both mechanisms are shown to have a concerted character, as opposed to a step-wise mechanism. The alternative mechanism of a hydrogen atom transport is critically examined, and evidence for strong solvent dependence is presented. Specifically, we observe electrostatic destabilization of the corresponding πσ* state by the aqueous solvent. As a consequence, no conical intersections are found along the reaction pathway.
Proton transfer reactions play a crucial role in a large variety of chemical reactions, as well as in biological systems.1–7 In aqueous solutions, where the hydrogen bond network is very extensive, these reactions can be enormously complicated due to the association of the structural reorganization of water molecules. In this context, relatively stable hydrogen bonded chains between series of water molecules have been discussed in the literature along which the proton transfer may occur.8–14 A “water wire” linked by hydrogen bonds may enable the transfer of protons in a concerted, where protons are transferred in a single step over the water molecules comprising the wire, or a sequential von-Grotthuss hopping fashion.12,14,15
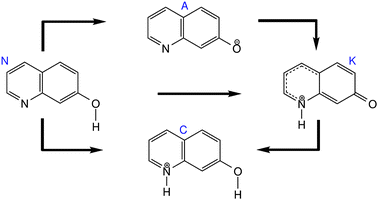
The mechanism of excited state proton transfer reactions in solutions may involve different pathways, depending on the reactivities of donor and acceptor sites of the compounds. In this regard, photoacid/-base molecules, known to perform proton transfer reactions after photoexcitation, have been extensively studied.16–28 7-Hydroxyquinoline (7HQ), a bifunctional chromophore, provides both an acidic site with proton donating functionality as well as a basic site that can accept a proton.29–40 In aqueous solutions, 7HQ can adopt four protropic equilibrium species, a neutral molecule (N), an enol-deprotonated anion (A), an imine protonated cation (C), and the equilibrium keto (K) form (see in Fig. 1).35–38,40 Upon excitation to the first excited state (S1), a proton transfer reaction is initiated, the hydroxyl group acts as a proton donor and the nitrogen atom acts as a proton acceptor. The keto form of 7HQ is found to be stable in the S1 state, and its fluorescence is strongly Stokes shifted relative to the enol.32,33 Hence, the occurrence of a proton transfer can be documented experimentally by the keto form.
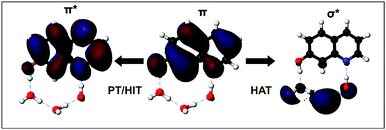
Several quantum chemical calculations can be found in the literature which have been successful in simulating particular features of the tautomerization of 7HQ starting with a pre-defined arrangement of a solvent cluster in the gas phase.39,41–46 The photoacidity of 7HQ has been discussed by Leutwyler and co-workers based on both configuration interaction singlets (CIS) and complete active space self-consistent field (CASSCF) methods along (mixed) hydrogen bonded wires consisting of NH3 and/or H2O molecules in the gas phase.47,48 According to their results, the enol–keto tautomerization in the excited state proceeds via hydrogen atom transfer instead of proton transfer in a step-wise fashion. From their calculations, a conical intersection between an excited ππ* state and a πσ* state drives the migration of an electron from the hydroxyl group to the solvent molecule (see in Fig. 2). Then, a net transfer of a hydrogen atom along the hydrogen bonded wire occurs by consecutive electron and proton transfer. As a last step, a reverse crossing brings the system back into the fluorescent ππ* state of the ketonic tautomer. Sobolewski and Domcke have also reported hydrogen atom transfer reactions in the excited state dynamics of phenol in the gas phase with NH3 and H2O molecules.49,50
In this work, we investigate proton and hydroxide ion (OH−) transfer reactions of the 7HQ·(H2O)3 complex with regard to both the release of a proton from the hydroxyl group to the water molecule and the cleaving of a proton from the water molecule to the nitrogen atom in the ground (S0), triplet (T1), and the singlet (S1) states in aqueous solution. Our excited state structure optimizations and transition state energetics shall clarify whether the OH− transport process can compete with the standard photoacid picture of proton release from the donor site. Hydroxide ion transfer based mechanisms have also been reported from experimental time-resolved and steady-state fluorescence studies.34,37
We have performed full structure optimizations and transition state calculations of 7HQ·(H2O)3 in aqueous solution using DFT/TD-DFT51 methods in the B3LYP/TZVP52,53 level in both ground and excited states using Gaussian 09.54 Solvation effects were captured by the conductor polarized continuum model.55 The relative energies of the optimized geometries are reported, including the zero point energy correction (see ESI†).
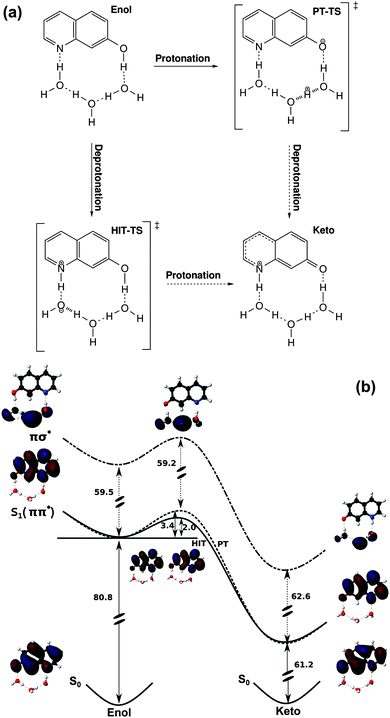
We have computed two different reaction pathways for the enol–keto tautomerization of 7HQ in the S1 state in aqueous solution. The first pathway starts with the deprotonation of the 7HQ hydroxyl group, (see Fig. 3a). Proton transport then occurs along the water wire to the nitrogen atom. In the quantum chemistry literature, only pathways starting with deprotonation of 7HQ have been considered.41–46 An alternative mechanism starts by protonation of the nitrogen site of 7HQ. This process corresponds to the protonation of the water-exposed nitrogen, then an effective charge will be transported along the water wire. Finally the transport terminates with deprotonation of the 7HQ hydroxyl group. We denote this mechanism by hydroxide ion transfer (HIT) (see Fig. 3). This mechanism has been discussed intensely in the literature, where quantum chemical calculations have led to the interesting hypothesis of “magic numbers” for the length of the water wire.56–59 At such specific numbers in the water chain, an enhanced stability of the wire has been predicted, leading to a potentially higher probability for the hydroxide ion transfer mechanism.
The transition states and reaction profiles for both mechanisms are depicted in Fig. 3b. The transition state of the proton transfer pathway contains a H5O2+ (Zundel cation), whereas the transition state of the hydroxide ion transfer occurs via H3O2− (Eigen-core hydrated proton). Moreover, the reaction path of hydroxide ion transfer and proton transfer was analyzed confirming that both hydroxide ion and proton transfer mechanisms happen in a concerted non-sequential way, where protons are transferred in a single step over the water wire. Compared to the study of Hassanali et al., we predict a somewhat reduced requirement of extended aqueous solvation effects for successful proton/OH− transfer processes.15 Nevertheless, the need for a stable arrangement of a certain number of water molecules as well as their stabilization by means of the surrounding solvent is a common finding.
The entire enol–keto tautomerization is exergonic by 16.7 kcal mol−1 and 11.3 kcal mol−1 in the S1 and T1 states, respectively (see in Table 1). In contrast to this in the ground state, the reaction is endergonic by 2.8 kcal mol−1. The resulting S1 state hydroxide ion and proton transfer pathways have similar reaction barriers of 2.0 kcal mol−1 and 3.4 kcal mol−1, respectively. When switching to gas phase cluster conditions, the hydroxide ion transfer pathway in the lowest S1 state exhibits a reaction barrier of 1.3 kcal mol−1 (see ESI†). For comparison, Leutwyler and co-workers reported an activation energy of ≈20 kcal mol−1 for the excited state hydrogen atom transfer in the essentially same system.47,48 We therefore believe that our hydroxide ion transfer must be considered at least as an alternative viable mechanism in the gas phase.
| State | ΔEr | ΔEa-HIT | ΔEa-PT |
|---|---|---|---|
| S0 | 2.8 | 10.2 | 15.1 |
| T1 | −11.3 | 6.3 | 8.5 |
| S1 | −16.7 | 2.0 | 3.4 |
To investigate the possibility of excited state hydrogen atom transfer mechanisms, we analyze the first two excited electronic states and the lowest πσ* state of enol, keto, and the transition states of the hydroxide ion and proton transfer pathways (see in Table 2). The lowest πσ* state corresponds to an excited state hydrogen atom transfer mechanism, because the σ* orbital is localized almost exclusively in the solvent region. This state is well-separated from the fluorescent S1 state, which is in contrast with gas phase studies by Leutwyler and co-worker47,48 where a conical intersection between ππ* and πσ* was reported. Moreover, we found no πσ* state excitation of the transition state of the hydroxide ion transfer in the electronic spectra but instead a σπ* state which might correspond to an analogous electron coupled hydroxyl radical transfer mechanism.
| Enol | TS-HIT | TS-PT | Keto | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| State | ΔE | f | State | ΔE | f | State | ΔE | f | State | ΔE | f |
| ππ* | 3.34 | 0.142 | ππ* | 3.02 | 0.133 | ππ* | 2.85 | 0.135 | ππ* | 2.47 | 0.15 |
| ππ* | 4.24 | 0.134 | ππ* | 4.1 | 0.213 | ππ* | 4.15 | 0.07 | ππ* | 3.94 | 0.158 |
| πσ* | 5.97 | 0.003 | πσ* | — | — | πσ* | 5.17 | 0.006 | πσ* | 5.5 | 0.002 |
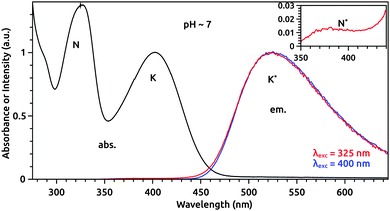
The absorption and fluorescence spectra at pH ∼ 7 are reported in Fig. 4. In neutral aqueous solution, 7HQ in the S0 state is present both in enol and keto forms labeled N and K in the steady-state absorption and fluorescence spectra. From the excited 7HQ, only a single fluorescence band is observed, regardless of excitation wavelength, which can be assigned to the keto configuration labeled K* in the emission spectrum in Fig. 4. Only a minor amount of N* emission is populated. The calculated fluorescence energies of 3.34 eV (N*) and 2.47 eV (K*) are in agreement with the experimental values of 3.35 eV and 2.37 eV, respectively. We also identified the lowest absorption bands of the enol-deprotonated (A) and the imine protonated cation (C) labeled in Fig. 1 at pH ∼ 13 and −1, respectively (see ESI†). Photoexcitation to A* results only in an eventual fluorescent photon emission of the A* form. No emission from the protonated form is observed. This shows that the proton capture process from the solvent has an effective timescales for beyond the lifetime of the excited state. At low pH ∼ −1, the predominant C form only emits an unshifted fluorescent photon. This emission of the C* form does not exhibit any decay characteristics for a proton dissociation process into the water. Hence, the OH group of 7HQ is not acidic enough to release its proton, and the acid–base equilibrium remains basically on the acid side.
In summary, quantum chemical calculations reveal a coexistence of intrinsically different tautomerization pathways in microsolvated photoacids along short water wires, which are significantly influenced by solvation effects. Besides the conventional proton (or Eigen/Zundel cation) transfer from the acidic to the basic site, we observe a novel tautomerization pathway, essentially consisting of an effective hydroxyl anion transport from base to acid. In contrast to previous reports on 7HQ, the existence of stable hydrogen atom species cannot be confirmed. Complexes involving the latter species are energetically destabilized by dielectric shielding effects due to the surrounding aqueous solvent, and may only exist in the gas phase. In particular, our calculations show that there is no conical intersection between ππ* and πσ* states along the reaction path in solution.
The energy profile of the novel OH− tautomerization pathway turns out to be virtually similar to the commonly assumed proton transfer. In this context, OH− transport means the initial capture of a proton by the basic site from the solvent, followed by a corresponding proton release of the acidic site into the solvent. The presence of extended solvation, however, is not required for this pathway, so that we expect this process to be observable experimentally both in solution and in the gas phase.
The energy profile of the reaction paths shows clear evidence that both transfer mechanisms happen in a concerted fashion, i.e. without stable intermediates along the water wire. While this observation does not exclude the possibility for intermediates under full solvation conditions at ambient temperatures, it illustrates that concerted proton and hydroxide ion transport mechanisms are indeed possible for short water wires.
We elucidate the coexistence of processes involving proton and hydroxide ion species. The latter is commonly partially discarded due to the unfavorable characteristics of the OH− in terms of its surprising hydrophobicity and slow diffusion properties. Our findings show that at least for short water wires, the hydroxyl anion transfer can energetically compete easily with its proton counterpart. This communication sheds new light on the molecular nature of acid–base proton exchange processes, some of the most fundamental chemical reactions in nature, and provides a novel interpretation of existing experimental observation.
Acknowledgements
This work was supported by a scholarship of the Leibniz Graduate School of Molecular Biophysics Berlin. The computations were performed with a grant of computer time provided by the NIC supercomputers of the Research Centre Jülich.References
- M. E. Tuckerman, D. Marx and M. Parrinello, Nature, 2002, 417, 925 CrossRef CAS PubMed.
- H. Chen, G. A. Voth and N. Agmon, J. Phys. Chem. B, 2010, 114, 333–339 CrossRef CAS PubMed.
- P. L. Geissler, C. Dellago, D. Chandler, J. Hutter and M. Parrinello, Science, 2001, 291, 2121–2124 CrossRef CAS PubMed.
- L. Vilciauskas, M. E. Tuckerman, G. Bester, S. J. Paddison and K.-D. Kreuer, Nature, 2012, 4, 461–466 CAS.
- D. Marx, M. Tuckerman, J. Hutter and M. Parrinello, Nature, 1999, 397, 601 CrossRef CAS PubMed.
- J. A. Morrone and M. E. Tuckerman, J. Chem. Phys., 2002, 117, 4403–4413 CrossRef CAS PubMed.
- C. Lao-ngam, P. Asawakun, S. Wannarat and K. Sagarik, Phys. Chem. Chem. Phys., 2011, 13, 4562–4575 RSC.
- V. R. I. Kaila and G. Hummer, J. Am. Chem. Soc., 2011, 133, 19040–19043 CrossRef CAS PubMed.
- J. Chen, X.-Z. Li, Q. Zhang, A. Michaelides and E. Wang, Phys. Chem. Chem. Phys., 2013, 15, 6344–6349 RSC.
- F. Garczarek and K. Gerwert, Nature, 2006, 439, 109–112 CrossRef CAS PubMed.
- Z. Cao, Y. Peng, T. Yan, S. Li, A. Li and G. A. Voth, J. Am. Chem. Soc., 2010, 132, 11395–11397 CrossRef CAS PubMed.
- B. K. Mai, K. Park, M. P. T. Duong and Y. Kim, J. Phys. Chem. B, 2013, 117, 307–315 CrossRef CAS PubMed.
- V. R. Kaila and G. Hummer, Phys. Chem. Chem. Phys., 2011, 13, 13207–13215 RSC.
- A. Hassanali, M. K. Prakash, H. Eshet and M. Parrinello, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 20410–20415 CrossRef CAS PubMed.
- A. Hassanali, F. Giberti, J. Cuny, T. D. Kühne and M. Parrinello, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 13723–13728 CrossRef CAS PubMed.
- N. Agmon, J. Phys. Chem. A, 2005, 109, 13–35 CrossRef CAS PubMed.
- Y. Erez and D. Hupper, J. Phys. Chem. A, 2010, 114, 8075–8082 CrossRef CAS PubMed.
- O. F. Mohammed, D. Pines, E. T. J. Nibbering and E. Pines, Angew. Chem., 2007, 119, 1480–1483 CrossRef.
- L. M. Tolbert and J. E. Haubricht, J. Am. Chem. Soc., 1994, 116, 10593–10600 CrossRef CAS.
- O. F. Mohammed, D. Pines, J. Dreyer, E. Pines and E. T. J. Nibbering, Science, 2005, 310, 83–86 CrossRef CAS PubMed.
- O. F. Mohammed, D. Pines, E. T. J. Nibbering and E. Pines, Angew. Chem., 2007, 119, 1480–1483 CrossRef.
- O. F. Mohammed, D. Pines, E. Pines and E. T. J. Nibbering, Chem. Phys., 2007, 341, 240–257 CrossRef CAS PubMed.
- M. Rini, B.-Z. Magnes, E. Pines and E. T. J. Nibbering, Science, 2003, 301, 249–352 CrossRef PubMed.
- M. Rini, D. Pines, B.-Z. Magnes, E. Pines and E. T. J. Nibbering, J. Phys. Chem. A, 2004, 122, 9593–9610 CrossRef PubMed.
- S. Luber, K. Adamczyk, E. T. J. Nibbering and V. S. Batista, J. Phys. Chem. A, 2013, 117, 5269–5279 CrossRef CAS PubMed.
- F. Messina, M. Prémont-Schwarz, O. Braem, D. Xiao, V. S. Batista, E. T. J. Nibbering and M. Chergui, Angew. Chem., Int. Ed., 2013, 52, 6871–6875 CrossRef CAS PubMed.
- I. Presiado, N. Karton-Lifshin, Y. Erez, R. Gepshtein, D. Shabat and D. Huppert, J. Phys. Chem. A, 2012, 116, 7353–7363 CrossRef CAS PubMed.
- S.-Y. Park and D.-J. Jang, J. Am. Chem. Soc., 2010, 132, 297–302 CrossRef CAS PubMed.
- J. L. Pérez-Lustres, F. Rodriguez-Prieto, M. Mosquera, T. A. Senyushkina, N. P. Ernsting and S. A. Kovalenko, J. Am. Chem. Soc., 2007, 129, 5408–5418 CrossRef PubMed.
- S.-Y. Park and D.-J. Jang, Phys. Chem. Chem. Phys., 2012, 14, 8885–8891 RSC.
- A. Csehi, L. Illes, G. J. Halasz and A. Vibok, Phys. Chem. Chem. Phys., 2013, 15, 18048–18054 RSC.
- E. Bardez, A. Chatelain, B. Larrey and B. Valeur, J. Chem. Phys., 1994, 98, 2357–2366 CrossRef CAS.
- O. Poizat, E. Bardez, G. Buntix and V. Alain, J. Phys. Chem. A, 2004, 108, 1873–1880 CrossRef CAS.
- S.-I. Lee and D.-J. Jang, J. Phys. Chem., 1995, 99, 7537–7541 CrossRef CAS.
- Y. S. Park, B. Kim, Y. S. Lee, O. H. Kwon and D.-J. Jang, Photochem. Photobiol. Sci., 2009, 8, 1611–1617 Search PubMed.
- S. Y. Park, Y. S. Lee, O. H. Kwon and D. J. Jang, Chem. Commun., 2009, 926–928 RSC.
- I. Presiado, Y. Erez, R. Gepshtein and D. Huppert, J. Phys. Chem. C, 2009, 113, 20066–20075 CAS.
- O. H. Kwon, T. G. Kim, Y. S. Lee and D. J. Jang, J. Phys. Chem. B, 2006, 110, 11997–12004 CrossRef CAS PubMed.
- N. Al-Lawatia, J. Husband, T. Steinbrecher and O. K. Abou-Zied, J. Phys. Chem. A, 2011, 115, 4195–4201 CrossRef CAS PubMed.
- B. Kang, K. C. Ko, S.-Y. Park, D.-J. Jang and J. Y. Lee, Phys. Chem. Chem. Phys., 2011, 13, 6332–6339 RSC.
- M. Thut, C. Tanner, A. Steinlin and S. Leutwyler, J. Phys. Chem. A, 2008, 112, 5566–5572 CrossRef CAS PubMed.
- C. Tanner, C. Manca and S. Leutwyler, J. Chem. Phys., 2005, 122, 204326 CrossRef PubMed.
- C. Manca, C. Tanner, S. Coussan, A. Bach and S. Leutwyler, J. Chem. Phys., 2004, 121, 2578–2590 CrossRef CAS PubMed.
- C. Tanner, C. Manca and S. Leutwyler, Science, 2003, 302, 1736–1739 CrossRef CAS PubMed.
- M. Guglielmi, I. Tavernelli and U. Rothlisberger, Phys. Chem. Chem. Phys., 2009, 11, 4549–4555 RSC.
- A. Fernández-Ramos, E. Martínez-Nunez, S. A. Vázquez, M. A. Ríos, C. Estévez, M. Merchán and L. Serrano-Andrés, J. Phys. Chem. A, 2007, 111, 5907–5912 CrossRef PubMed.
- C. Tanner, M. Thut, A. Steinlin, C. Manca and S. Leutwyler, Int. Rev. Phys. Chem., 2005, 24, 457–488 CrossRef.
- C. Tanner, M. Thut, A. Steinlin, C. Manca and S. Leutwyler, J. Phys. Chem. A, 2005, 110, 1758–1766 CrossRef PubMed.
- A. L. Sobolewski, W. Domcke, C. Dedonder-Lardeux and C. Jouvet, Phys. Chem. Chem. Phys., 2002, 4, 1093 RSC.
- W. Domcke and A. L. Sobolewski, Science, 2003, 302, 1693 CrossRef CAS PubMed.
- E. Runge and E. K. U. Gross, Phys. Rev. Lett., 1984, 52, 997–1000 CrossRef CAS.
- A. D. Becke, J. Chem. Phys., 1993, 8, 5648–5652 CrossRef PubMed.
- A. Schäfer, C. Huber and R. Ahlrichs, J. Chem. Phys., 1994, 100, 5829–5835 CrossRef PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09 Revision A.02.
- V. Barone and M. Cossi, J. Phys. Chem. A, 1998, 102, 1995–2001 CrossRef CAS.
- N. J. Singh, M. Park, S. K. Min, S. B. Suh and K. S. Kim, Angew. Chem., Int. Ed., 2006, 45, 3795–3800 CrossRef CAS PubMed.
- M. Park, I. Shin, N. J. Singh and K. S. Kim, J. Phys. Chem. A, 2007, 111, 10692–10702 CrossRef CAS PubMed.
- S. Odde, C. Pak, H. M. Lee, K. S. Kim and B. J. Mhin, J. Chem. Phys., 2004, 121, 204–208 CrossRef CAS PubMed.
- I. Shin, M. Park, S. K. Min, E. C. Lee, S. B. Suh and K. S. Kim, J. Chem. Phys., 2006, 125, 234305–234312 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Computational details, optimized geometries of the compounds in the S1 state, their relative energies and extensive fluorescence and absorption data. See DOI: 10.1039/c4cp00970c |
| This journal is © the Owner Societies 2014 |