Thermodynamic and kinetic analysis of heterogeneous photocatalysis for semiconductor systems
Abstract
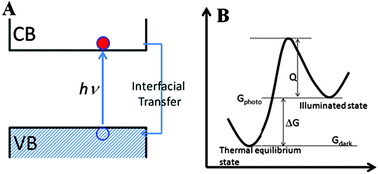
Since the report of the Honda–Fujishima effect, heterogeneous photocatalysis has attracted much attention around the world because of its potential energy and environmental applications. Although great progresses have been made in recent years, most were focused on preparing highly-active photocatalysts and investigating visible light utilization. In fact, we are still unclear on the thermodynamic and kinetic nature of photocatalysis to date, which sometimes leads to misunderstandings for experimental results. It is timely to give a review and discussion on the thermodynamics and kinetics of photocatalysis, so as to direct future researches. However, there is an absence of a detailed review on this topic until now. In this article, we tried to review and discuss the thermodynamics and kinetics of photocatalysis. We explained the thermodynamic driving force of photocatalysis, and distinguished the functions of light and heat in photocatalysis. The Langmuir–Hinshelwood kinetic model, the ˙OH oxidation mechanism, and the direct–indirect (D–I) kinetic model were reviewed and compared. Some applications of the D–I model to study photocatalytic kinetics were also discussed. The electron transport mode and its importance in photocatalysis were investigated. Finally, the intrinsic relation between the kinetics and the thermodynamics of photocatalytic reactions was discussed.

 Please wait while we load your content...
Please wait while we load your content...