Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis of copper sulphide-based hybrid nanostructures and their application in shape control of colloidal semiconductor nanocrystals
Joanna
Kolny-Olesiak
*
Energy and Semiconductor Research Laboratory, Department of Physics, Carl von Ossietzky University of Oldenburg, 26129 Oldenburg, Germany. E-mail: joanna.kolny@uni-oldenburg.de; Fax: +49 441 798 3326; Tel: +49 441 798 3261
First published on 12th May 2014
Abstract
Copper sulphide is a material with low toxicity and high application potential. Colloidal synthesis allows its incorporation into hybrid nanostructures, which not only combine the properties of different materials within one nanocrystal, but also exhibit new features due to the interaction of the building blocks connected on the nanometer scale. Starting with copper sulphide seeds, such hybrid nanocrystals composed of copper sulphide and semiconductors like CuInS2, CuInxGa1−xS2, Cu2ZnSnS4, ZnS, CdS, and PbS have been synthesized. In some of these reactions the main focus lies on the formation of new hybrid nanocrystals; in others copper sulphide plays the role of a catalyst, and copper sulphide containing hybrid nanoparticles are only intermediates in the synthesis and shape control of other semiconductor materials. Both possibilities and the underlying growth mechanisms are discussed in this article.
Introduction
The field of nanochemistry rapidly evolved during the last two decades. Synthetic procedures have been developed which allow for precise control of the size, shape, structure and composition of a variety of nanostructured materials.1–8 These paved the way for studying the size and shape dependent properties of nano-crystallites, and therefore, increased our understanding of the properties of matter on the nanometer scale, resulting in possible creation of custom-made materials for a variety of applications.9–12 The possibilities to manipulate and control the properties of nanocrystals can be greatly extended by generating nanoparticles composed of more than one material. These so-called hybrid nanocrystals not only combine different optical, magnetic or catalytic features within one nanostructure but also evolve with new characteristics.13–16The focus of this article lies on the synthesis of semiconductor–semiconductor hybrid nanostructures which can be generated from copper sulphide seeds. Copper sulphide nanoparticles, which attract scientific attention due to their interesting properties, low toxicity and, therefore, high application potential, can be synthesized with a high degree of control over their size and shape.17–22 Cu2−xS is a p-type semiconductor which can form several nonstoichiometric compounds with x ranging from 0 to 1 (hexagonal covellite CuS, orthorhombic anilite Cu1.75S, hexagonal digenite Cu1.8S, monoclinic djurleite Cu1.94–1.97S, monoclinic roxbyite Cu1.75S, hexagonal or orthorhombic chalcocite Cu2S). Depending on the composition, the bandgap of Cu2−xS lies between 1.2 eV (for Cu2S) and 2.0 eV (for CuS); thus, copper sulphide can absorb a large fraction of the solar spectrum, which makes it an interesting material for photovoltaic applications. Moreover, the large number of copper vacancies in the non-stoichiometric compounds and the resulting high concentration of free charge carriers lead to plasmonic absorption in the IR spectral region.23 Furthermore, the presence of cationic vacancies and the high mobility of the cations facilitate incorporation of other cations into the copper sulphide lattice.
Because of these properties, copper sulphide is not only a material with high application potential but also a suitable catalyst which facilitates growth and enables shape control of other semiconductor nanomaterials. Therefore, two aspects will be discussed here: the possibility to modify the properties of copper sulphide particles by combining them with other semiconductors and the potential to synthesize shape-controlled nanocrystals in procedures including copper sulphide containing intermediates. Recent advances in the synthetic methods using copper sulphide seeds are summarized, and an overview of the current understanding of the underlying reaction mechanisms is given.
Overview of synthetic procedures
Several materials have already been combined with copper sulphide to form hybrid nanostructures, e.g., CuInS2,24–28 CuInxGa1−xS2,29,30 Cu2ZnSnS4,31,32 ZnS,33–36 CdS,37 PbS,38 and MnS.39 In all cases the synthetic procedures start with the formation of copper sulphide seeds, which subsequently react with other cations and an additional sulphur source to form heterostructured nanocrystals. These two steps can follow each other in a one-pot (heat-up or hot-injection) reaction; differences in the reactivity of the cations forming both parts of the nanostructure are in many cases sufficient to separate these two stages of the synthesis. Other procedures use preformed seeds (usually in the original reaction solution without further purification), and the formation of the hybrid nanoparticles is induced by the addition of the precursors for the formation of the second semiconductor material.37–40 New educts can be added all at once, in several small injections or continuously using a syringe pump. The rate of the addition of the precursors can influence the morphology of the resulting nanocrystals; other parameters, which can be varied to control the size and the shape of the resulting nanocrystals are the ratio between copper and the other cations and the reaction temperature. Experimental details of the synthesis of different copper sulphide based heterostructured nanocrystals as well as information about the morphology of the resulting materials can be found in Table 1.| Precursors, ligands, and solvents | Method/conditions | Shape, size, and structure | Ref. |
|---|---|---|---|
| acac: acetylacetonate, dedt: diethyldithiocarbamate, 1-DDT: 1-dodecanethiol, t-DDT: tert-dodecanethiol, HDA: hexadecylamine, HNRs: heterostructured nanorods, OA: oleic acid, OAc: acetate, ODE: octadecene, OLAM: oleylamine, TOPO: trioctylphosphine oxide, WZ: wurtzite, and ZB: zincblende. | |||
| Cu–In–S | |||
| Cu-oleate, In-oleate, OLAM, 1-DDT | 230–250 °C, 10 min - 1 h, heat-up | Acorn-shape: 30 nm × 48 nm, bottle-shape: 40 nm × 110 nm, larva-shape: 45 nm × 185 nm | Choi et al. 2006 (ref. 46) |
| Cu(acac)2, In(acac)3, 1-DDT, | 200 °C, injection of In(acac)3 into preformed copper sulphide seeds | HNRs: 77 nm × 12.0 nm, tear-drops: 48 nm× 23 nm | Han et al. 2008 (ref. 40) |
| Cu-oleate, In-oleate, 1-DDT, OLAM | 215–300 °C, 20 min, heat-up | Nanorods, WZ | Connor et al. 2009 (ref. 24) |
| Cu(OAc)2, In(OAc)3, 1-DDT, t-DDT, TOPO, OLAM | 240 °C, 30 s - 1 h, hot-injection | Various sizes, HNRs, P-shaped, hexagonal plates, WZ | Kruszynska et al. 2010 (ref. 25) |
| Cu(OAc)2, In(OAc)3, 1-DDT, t-DDT, TOPO, ODE, OLAM | 240 °C, 50 s - 1 h, hot-injection | Triangular pyramids, WZ | Kruszynska et al. 2011 (ref. 27) |
| Cu(NO3)2, In(NO3)3, 1-DDT, OLAM, OA | 240°, 30 min, heat-up | Quasi-spherical, rods, WZ | Lu et al. 2011 (ref. 28) |
| Cu(dedc)2, Cu(acac)2, In(dedc)3, OLAM, 1-DDT | The mixture is rapidly heated to 180 °C by immersion in an oil bath, 60 min | Nanoribbons with a rectangular cross-section: 2–3 μm in length, 20–50 nm in width, 15 nm in thickness, WZ | Li et al. 2013 (ref. 29) |
| Cu–In–Zn–S | |||
| CuOAc, In(OAc)3, zinc stearate, zinc ethylxanthate, 1-DDT, ODE | 240 °C, slow precursor injection via a syringe pump | Various sizes, Cu2S–ZnS, Cu2S–CIS and Cu2S–(CIS–ZnS alloy) HNRs, chalcocite and WZ | Chang and Cheng 2011 (ref. 26) |
| Cu1.94S–ZnS seeds, In(OAc)3, 1-DDT, ODE | 200 °C, 4 h, heat-up | Burning torch-like CIS–ZnS HNRs, head: 33 nm × 24 nm, tail: 29 nm × 9 nm, WZ | Yi et al. 2013 (ref. 47) |
| 200 °C, 6 injections of In(ac)3, 4 h reaction after last injection | Long rod-like CIS–ZnS HNRs, WZ | ||
| Cu–Zn–S | |||
| Cu(acac)2, Zn(acac)2, 1-DDT | 200 °C, ca. 4–5 h, injection of Zn(acac)2 in 6 portions | HNRs | Yi et al. 2010 (ref. 33) |
| CuI, Zn(dedt)2, OLAM | 150 °C, 30 min, heat-up | Hexagonal prism length: 60 nm; diameter: 30 nm, monoclinic Cu1.94S, WZ ZnS | Han et al. 2012 (ref. 35) |
| Cu-oleate, 1-DDT, OLAM | 220 °C, 30 min, injection of Zn-oleate at 220 °C | 28 nm slab-like ZnS nanocrystals embedded in a Cu1.94S nanosphere, monoclinic Cu1.94S and WZ ZnS | Huang et al. 2012 (ref. 34) |
| Zn(acac)2, Cu(acac)2, 1-DDT, ODE | 240–270 °C, reaction times up to 360 min, heat-up | Taper-like, matchstick-like, tadpole-like, or rod-like hybrid nanostructures, monoclinic Cu1.94S, WZ ZnS | Ye et al. 2013 (ref. 36) |
| Cu–In–Ga–S | |||
| Cu(acac)2, In(acac)3, Ga(acac)3, TOPO, ODE, 1-DDT, t-DDT | Injection at 155 °C, reaction temperature: 250–270 °C, 15–30 min | Nanorods, diameter: 7 nm, WZ | Coughlan et al. 2013 (ref. 30) |
| Cu(dedc)2, Cu(acac)2, In(dedc)3, Ga(dedc)3, OLAM, 1-DDT | The mixture is rapidly heated to 180 °C by immersion in an oil bath, 60 min | Nanoribbons with a rectangular cross-section: 1–2 μm in length, 30–50 nm in width, 9 nm in thickness, variable composition between 20 and 80% Ga, roxbyite and WZ | Li et al. 2013 (ref. 29) |
| Cu–Zn–Sn–S | |||
| Cu(acac)2, Zn(OAc)2, SnCl2, 1-DDT, OLAM, | 200–260 °C, different reaction times: up to 24 h, heat-up | Bullet-shape, 13 nm, WZ | Liao et al. 2013 (ref. 32) |
| CuCl2, Zn(dedc)2, Sn(dedc)4, 1-DDT, OLAM | 200 °C, 60 min, fast heat-up by immersion in an oil bath | Nanorods, 200 nm in length, WZ | Zhang et al. 2013 (ref. 31) |
| Cu–Cd–Zn–S | |||
| Cu(dedt)2, Cd(dedt)2, Zn(dedt)2 hexanethiol, trioctylamine | 250 °C, injection of Cd(dedt)2 and Zn(dedt)2 into preformed Cu1.94S seeds | Hexagonal discs with regions of monoclinic Cu1.94S and WZ CdS (CdxZn1-xS) | Regulacio et al. 2011 (ref. 37) |
| Cu–Pb–S | |||
| Cu(acac)2, Pb(dedt)2, 1-DDT | 200 °C, 10 min, injection of Pb(dedt)2 into preformed Cu1.94S seeds | HNRs, chalcocite and rock salt | Zhuang et al. 2012 (ref. 38) |
| Cu–Mn–S | |||
| Cu(OAc)2, Mn(OAc)2 1-DDT, OLAM | 220 °C, 15 min, injection of Mn(ac)2 into preformed Cu1.94S seeds | HNRs, 16 nm in length, djurleite and WZ | Zhou et al. 2013 (ref. 39) |
Growth mechanisms
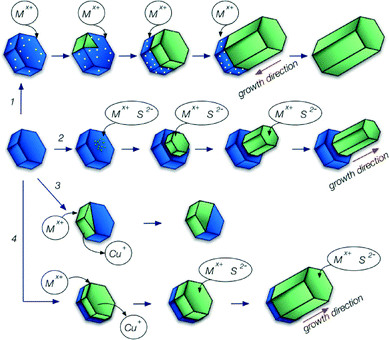
Although all of the synthetic methods described here follow a similar general pathway, they differ in the underlying growth mechanism (see Scheme 1). Most of the studies discussed in the next section describe the formation of one-dimensional hybrid nanocrystals. This kind of structure can be generated by two mechanisms: seeded growth or catalyst assisted growth. In both cases the synthesis starts with the formation of particles, which subsequently serve as starting points for the growth of another material. However, the role of these seeds in the growth process of the second material is different for both mechanisms.In seeded growth a second material nucleates on the surface of the first one. Its further growth takes place directly through accretion of monomers from solution, without any additional contribution from the original seed and the growth direction is away from the original seed (see Scheme 1 pathway 2).
In catalyst assisted growth, usually materials with a relatively low melting point (e.g., Au, Bi, In or Sn) are used as catalysts. The precursors of the second material dissolve in the liquid catalyst, but because of its limited solubility, above some critical concentration the second material forms a solid seed inside the first one, which subsequently grows through addition of monomers dissolved in the catalyst (see Scheme 1 pathway 1). This method has been effectively used to produce nanorods and nanowires of many different materials starting with precursors in the gas phase (vapour–liquid–solid procedure) or in solution (solution–liquid–solid procedure).41,42
Also copper sulphide can play the role of a catalyst in the growth of other materials, in spite of being solid at the reaction temperatures typically used in colloidal synthesis. Copper sulphide is a solid-state superionic conductor.43–45 At a relatively low temperature of 105 °C Cu2S undergoes phase transformation to a solid–liquid hybrid phase: the copper cations move like a liquid through the anion sublattice, which maintains its solid form.45
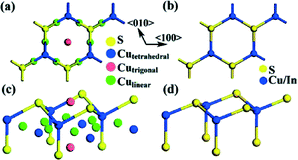
The sulphur anion sublattice of Cu2S is nearly identical to that of other metal sulphides with a wurtzite structure, e.g., CuInS2, CuGaS2, Cu2ZnSnS4, MnS or ZnS (see Fig. 1 for a comparison of the Cu2S and CuInS2 structures). Therefore, in most cases materials growing via a route involving hybrid structures with copper sulphide exhibit a wurtzite structure, even if this is not the thermodynamically stable modification at room temperature.
In some of the examples discussed below the formation of a hybrid nanostructure involves cation exchange.37–39 This method can be used when both materials involved contain the same kind of anion while the cations possess different chemical properties.
When hybrid nanostructures are formed through this mechanism, only part of the seed undergoes cation exchange (Scheme 1 pathway 3). Usually, during cation exchange the anion lattice does not change,48 therefore the resulting particles retain the shape of the original seeds.
The next growth mechanism, suggested for MnS and some of the ZnS containing hybrid nanostructures, also involves cation exchange but only in the initial step of the formation of the hybrid nanocrystal. After the formation of a hybrid nanostructure due to partial cation exchange the further growth follows the same pathway as that for the seeded growth described above (Scheme 1 pathway 4).
Overview of hybrid nanostructures with copper sulphide
Ternary and quaternary copper sulphides
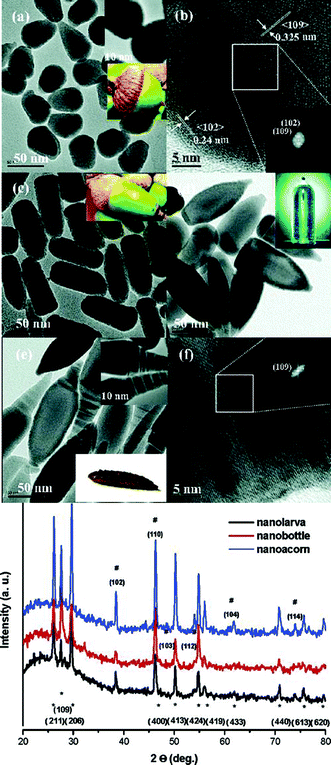
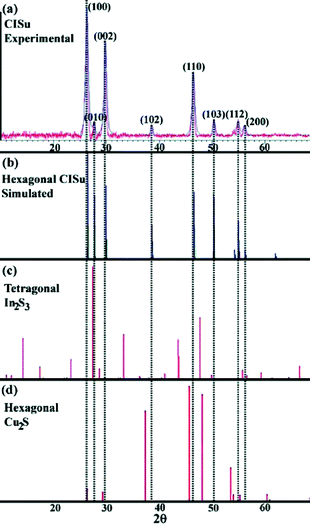
Nanoparticles composed of ternary and quaternary copper sulphides, such as copper indium sulphide, copper gallium sulphide and copper zinc tin sulphide, are of particular interest as absorber materials for solar energy conversion.7,8,49 Controlling the size, shape, structure and composition of these nanoparticles is essential to fully exploit their application potential. Therefore, there is a lot of activity in the field of colloidal synthesis of these nanocrystalline semiconductors. One possible way to control their shape is to use copper sulphide based intermediates in the synthesis. The following paragraphs give a short overview of the development of the synthetic methods for colloidal CuInS2, CuGaS2, CuInxGa1−xS2 and Cu2ZnSnS4 nanocrystals using this approach.The first example of shape-controlled hybrid nanostructures containing a copper sulphide phase was published in 2006 by Choi et al.46 (Fig. 2). Cu2S was generated in the beginning of a one-pot heat-up reaction in a decomposition process of copper oleate and dodecanethiol, yielding oleic acid and dodecene as by-products.
The subsequent formation of the hybrid nanoparticles was described as seed-mediated growth of a second phase on the surface of the Cu2S particles. The X-ray diffraction (XRD) pattern of the resulting particles was assigned to a mixture of the tetragonal structure of In2S3 and the hexagonal chalcocite-structured Cu2S. Spatially resolved energy dispersive X-ray analysis (EDX) measurements presented in the article show indeed the presence of copper sulphide regions within these particles; however the stoichiometry of the resulting particles is close to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (Cu
2 (Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In
In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
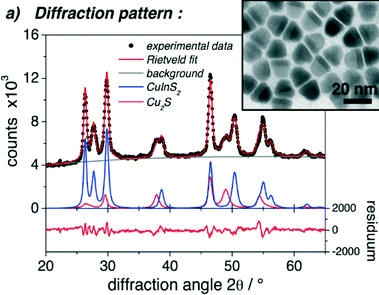
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S), which gives rise to the assumption that it is composed of copper indium disulphide and not indium sulphide. This is further supported by the comparison of the diffraction data presented within the article with the diffraction patterns of wurtzite copper indium disulphide (first described in 2008 by Pan et al.50). The reflections assigned to Cu2S–In2S3 match exactly the positions of the reflections of wurtzite copper indium disulphide. Also a comparison with the XRD data of other copper sulphide containing heterostructures26,27,31,38 shows that the intensity of the reflections, which can be assigned to the copper sulphide phase, is usually relatively low compared to that of, e.g., CuInS2 (see Fig. 3). Therefore, the interpretation of the data presented in the article, especially the assignment of the phases, should probably be partly reconsidered.
S), which gives rise to the assumption that it is composed of copper indium disulphide and not indium sulphide. This is further supported by the comparison of the diffraction data presented within the article with the diffraction patterns of wurtzite copper indium disulphide (first described in 2008 by Pan et al.50). The reflections assigned to Cu2S–In2S3 match exactly the positions of the reflections of wurtzite copper indium disulphide. Also a comparison with the XRD data of other copper sulphide containing heterostructures26,27,31,38 shows that the intensity of the reflections, which can be assigned to the copper sulphide phase, is usually relatively low compared to that of, e.g., CuInS2 (see Fig. 3). Therefore, the interpretation of the data presented in the article, especially the assignment of the phases, should probably be partly reconsidered.
A study presenting the formation of copper sulphide/indium sulphide hybrid structures was published in 2008 by Han et al.40 A high degree of shape control was achieved here by the addition of indium acetylacetonate to preformed copper sulphide seeds. By changing the time and the frequency of the addition of the indium precursor matchstick- and tear drop-like particles were synthesized. The underlying growth mechanism was investigated in this study, revealing that the formation of the hetero-nanostructures is based on a catalyst assisted mechanism rather than seed-mediated growth. However, here the assignment of the two phases comprising the hybrid nanoparticles to Cu2S–In2S3 based on the XRD data might also need reconsideration because the positions of the reflections exactly match those of wurtzite copper indium disulphide.
In 2009 Connor et al.24 described the synthesis of CuInS2 nanorods, which starts with the formation of copper sulphide nanoparticles and goes through an intermediate stage of heterostructured CuInS2–Cu2S nanorods (Fig. 4). The crystallographic structure of the CuInS2 part of the hybrid nanostructure was identified as wurtzite. A comparison with the reference XRD data for Cu2S and In2S3 clearly showed that the diffraction pattern of the particles described in the article cannot be satisfactorily explained as a superposition of the diffraction patterns of Cu2S and In2S3 (see Fig. 5). Connor et al.24 assumed an epitaxial growth of CuInS2 on the surface of Cu2S, followed by a phase conversion of the Cu2S part of the hybrid structure to CuInS2. The one dimensional growth of the CuInS2 phase was explained by the different reactivity of the two (001) faces of the Cu2S nanodiscs: one terminated with cations, the other with anions. At later stages of the reaction, when most of the copper precursor in the reaction solution was used up, the copper sulphide part of the nanostructure abruptly disappeared, which the authors attribute to a solid–solid seed based transformation due to the diffusion of indium ions from CuInS2 to Cu2S, facilitated by the high mobility of the cations in the copper sulphide phase at the reaction temperature. Thus, the reaction mechanism suggested here involves first epitaxial growth, while the high ionic conductivity of copper sulphide plays a role only during the last stage of the reaction.
 | ||
| Fig. 4 Growth mechanism proposed for the formation of CuInS2 nanorods. Reprinted with permission from J. Am. Chem. Soc., 2009, 131, 4962–4966. Copyright (2009) American Chemical Society. | ||
The following articles about the synthesis CuInS2 nanoparticles starting with copper sulphide seeds reported a higher degree of shape control and also contributed to a better understanding of the growth process of CuInS2–Cu2S hybrid nanocrystals. In 2010 Kruszynska et al.25,27 showed the formation of CuInS2–Cu2S and CuInS2 nanorods with different aspect ratios, dimeric nanorods as well as hexagonal discs and P-shaped particles in a hot-injection synthesis (Fig. 6). Also the formation of alloyed ZnS–CuInS2 nanorods was reported for the first time. The investigation of the growth process revealed that the formation of the CuInS2 phase takes place inside the copper sulphide seeds. The initial quasi-spherical hybrid nanostructure evolves into a Janus particle composed of two discs when the size of the CuInS2 phase exceeds a certain size. The further growth of the CuInS2 part of the hybrid nanostructure takes place only at the interface between the two phases, enabling efficient shape control of the evolving structure by influencing the size and the position of the copper sulphide phase within the hybrid structure. The copper sulphide phase disappears at the later stages of the reaction, which also stops the further growth of the CuInS2 phase; this additionally supports the catalytic role of copper sulphide in this synthesis.
Lu et al.28 published in 2011 a heat-up synthesis of CuInS2 nanorods allowing for precise shape control. Here, the formation of Cu2S in the beginning of the reaction was also assumed to play a major role in the size and shape evolution of the products of the reaction; however, no detailed investigation of the growth process was presented.
Chang and Cheng26 synthesized particularly uniform CuInS2–Cu2S, (ZnS)1−x(CuInS2)x–Cu2S and ZnS–Cu2S heterostructured nanorods by dropwise addition via a syringe pump of indium and/or zinc precursors to a solution of Cu2S seeds. The growth process and the shape evolution of the hybrid nanorods were monitored by taking aliquots from the reaction solution at different reaction times and studying them by TEM and XRD (see Fig. 7); however, no proof of the suggested seeded-growth mechanism was obtained.
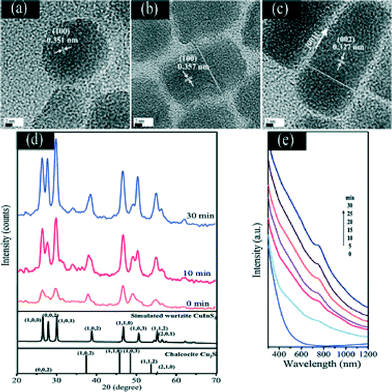 | ||
| Fig. 7 HRTEM images of as-prepared Cu2S–CuInS2 nanocrystals at different reaction times: (a) 0 min, (b) 10 min, and (c) 30 min. XRD patterns (d) and absorption spectra (e) of Cu2S–CuInS2 nanocrystals synthesized at different reaction times. Reproduced from ref. 26 with permission from The Royal Society of Chemistry. | ||
Not only isolated copper sulphide nanocrystals but also those already part of a heterostructure can be used to synthesize CuInS2. A reaction of ZnS–Cu2S nanorods with an indium precursor leads to the formation of CuInS2–ZnS hybrid nanorods.47 The morphology of the CuInS2 part of the resulting nanostructures depends on the rate of the addition of the indium salt.
Another example showing the potential of copper sulphide seeds to control the shape of CuInS2 and CuInxGa1−xS2 nanocrystals was demonstrated by Li et al. in 2013.29 2–3 μm long wurtzite CuInS2 and CuInxGa1−xS2 nanoribbons with a rectangular cross-section growing along the [110] direction and enclosed by (001) and (1–10) crystallographic faces were obtained by rapidly heating the reaction mixture (Fig. 8). The growth process starts with the formation of roxbyite nanocrystals, which are subsequently converted to bat-like heterostructures with a hemispherical copper sulphide head. The roxbyite head is about 10–15 nm in diameter in the beginning of the reaction; however, it grows to 20–40 nm within the first few minutes of the synthesis after which it remains stable in size. This behavior can be attributed to catalyst swelling, i.e., dissolution of In and/or Ga ions in the catalyst. Different lattice mismatch between roxbyite and wurtzite CuInS2 (or CuInxGa1−xS2) along different crystallographic directions leads to the formation of nanoribbons with a rectangular cross-section (20–50 nm: width; 15 nm: thickness). CuInxGa1−xS2 nanorods were synthesized by a hot-injection method by Coughlan et al.30 in 2013. The growth of the CuInxGa1−xS2 nanocrystals starts with the formation of copper sulphide particles (described as a mixture of different structures: chalcocite, covellite, anilite) which are converted to CuInS2 within the first 5 min of the reaction. Only during further heating do these particles react with the Ga ions present in the reaction solution to form CuInxGa1−xS2 nanocrystals. This difference in the reactivity between indium and gallium ions can be rationalized by taking into account that Ga3+ is a harder acid compared to In3+. The reaction conditions and stabilizers were systematically varied within this study, allowing the control of the shape of the resulting CuInxGa1−xS2 nanocrystals. For instance, the size of the copper sulphide nanocrystals was controlled by the addition of amines with different chain lengths. In this way the diameter of the resulting nanorods could also be controlled.
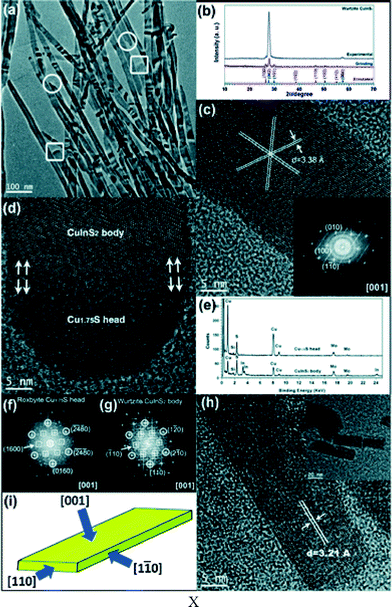 | ||
| Fig. 8 TEM images of CuInS2 nanoribbons. (a) The boxed parts refer to the head tip of the ribbons and the circled parts refer to the tail tip of ribbons. (b) XRD patterns of the CuInS2 nanoribbons. (c) A HRTEM image of CuInS2 nanoribbons. The inset shows the FFT pattern. (d) A HRTEM image of the head and body of a CuInS2 nanoribbon. (e) EDS spectra collected from the head and body of CuInS2 nanoribbons. (f and g) The two corresponding FFT patterns of the head and body of the nanoribbon in (d). The circled and boxed spots represent the relative orientation in Cu1.75S and CuInS2, respectively. (h) A cross-sectional HRTEM image of CuInS2 nanoribbons. The inset shows the low magnification TEM image displaying a rectangular cross section. (i) The schematic morphology of the nanoribbon with the crystallographic orientation. Reproduced from ref. 29 with permission from The Royal Society of Chemistry. | ||
Another quaternary material, which can be synthesized starting with copper sulphide seeds is wurtzite Cu2ZnSnS4. Liao et al.32 described the formation of bullet-like Cu2ZnSnS4 nanocrystals from djurleite copper sulphide nanoparticles. The characterization of the reaction products obtained after different reaction times revealed that the fraction of copper sulphide within the hybrid nanostructures gradually decreased and at longer reaction times pure Cu2ZnSnS4 nanocrystals were obtained. Zhang et al.31 synthesized leaf-shaped wurtzite Cu2ZnSnS4 nanoparticles 200 nm in length using cubic α-Cu2S nanocrystals as a catalyst in a heat-up reaction. Nanorods with heads composed of copper sulphide were observed as an intermediate state, but the final product was pure Cu2ZnSnS4 due to interdiffusion of Zn, Sn and Cu between Cu2ZnSnS4 and Cu2S.
Binary sulphides
This section is focused on the synthesis of hybrid nanostructures composed of copper sulphide and binary semiconductors, such as ZnS, CdS, PbS and MnS. The main motivation to generate this kind of hybrid nanomaterial is given by the possibility to modify the properties of copper sulphide nanocrystals in order to meet specific requirements of applications such as, photovoltaics, optoelectronics or theranostics. Copper sulphide–cadmium sulphide hetero-nanostructures have a type II band alignment, which facilitates the separation of light induced charge carriers. Therefore, this material combination is a potential absorber for nano-structured solar cells. Also the combination of copper sulphide with lead sulphide or the less toxic zinc sulphide is thought to be advantageous for applications in solar energy conversion.Copper sulphide particles exhibit localized surface plasmon resonance due to the large number of copper vacancies, and, consequently, high concentration of free charge carriers. This feature is of interest not only from the point of view of basic research, but also for a variety of applications, e.g., in photothermal therapy or as nano-antennas. The position of the plasmon band of copper sulphide particles can be influenced by combining copper sulphide with other materials, e.g., ZnS or MnS. In this way custom-made plasmonic materials can be obtained, e.g. absorbing within the so-called “therapeutic window” (700–1500 nm) or in the wavelength range, which is of interest for applications in telecommunication devices (1200–1600 nm). Using MnS for the modification of the plasmonic absorption of copper sulphide particles shifts the absorption to the “therapeutic window” and, thus, enables the use of the resulting hybrid nanostructures in photothermal therapy. Furthermore, MnS is a paramagnetic material which can be used as a contrast agent for nuclear magnetic resonance imaging. Therefore, copper sulphide–manganese sulphide heterostructures are potential theranostic agents.
The first example of heterostructured Cu1.94S–ZnS nanorods was presented in 2010 by Yi et al.33 A Zn precursor was injected in six portions within several hours into a solution containing djurleite seeds. The thickness of the nanorods could be controlled by the size of the copper sulphide particles. The shape evolution of the ZnS nanorods indicates that the copper sulphide particles do not only serve as a template but also play the role of a catalyst in the formation of the ZnS phase.
An impressive example of shape control in a one-pot heat-up reaction was demonstrated by Han et al.35 in 2012, who obtained hexagonal-prismatic Cu1.94S–ZnS, Cu1.94S–ZnS–Cu1.94S, and Cu1.94S–ZnS–Cu1.94S–ZnS–Cu1.94S heteronanostructures with screw-, dumbbell- and sandwich-like shapes (Fig. 9a). The ratio between copper and zinc in the reaction solution strongly influenced the growth process and, thus, the shape of these nanostructures. Their formation was a consequence of several different growth steps, such as partial cation exchange, growth through accretion of monomers from solution and restructuring. The reaction pathways leading to the generation of different shapes are summarized in Fig. 9b.
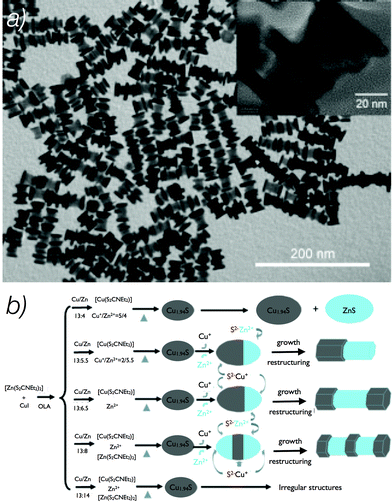 | ||
| Fig. 9 TEM image of Cu1.94S–ZnS heteronanostructures (a). Synthetic routes for preparation of different hexagonal prism Cu1.94S–ZnS heteronanostructures (b). Adapted with permission from ref. 35 Copyright© 2012 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
The first study of the applicability of a Cu1.94S–ZnS heterostructure as a nano-antenna was published in 2012 by Huang et al.34 The formation of the hybrid nanoparticles was induced by the injection of a zinc precursor into preformed copper sulphide seeds. The formation of the ZnS phase embedded in copper sulphide (Fig. 10) is assumed to be due to a partial cation exchange, which is followed by the further growth outside the copper sulphide seeds by accumulation of ions from solution.
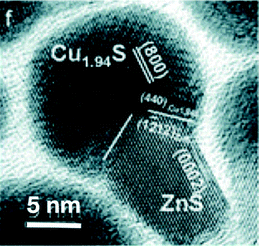 | ||
| Fig. 10 A Cu1.94S–ZnS heteronanostructure. Adapted from ref. 34 with permission from The Royal Society of Chemistry. | ||
Another example of a copper sulphide–zinc sulphide nanostructure was shown in 2013 by Ye et al.,36 who studied the influence of the copper to zinc ratio on the structure and the composition of the resulting particles. Copper sulphide particles, and, consequently, the hybrid structures with ZnS were formed only above a certain copper amount in the reaction solution. Below this amount, cubic copper-doped ZnS was formed whereas the ZnS phase which was found within the hybrid nanostructures had a hexagonal wurtzite structure and did not contain copper dopant atoms. The authors assume that the dissolution of Zn ions in Cu1.94S nanocrystals is the first step in the formation of the hybrid nanocrystals.
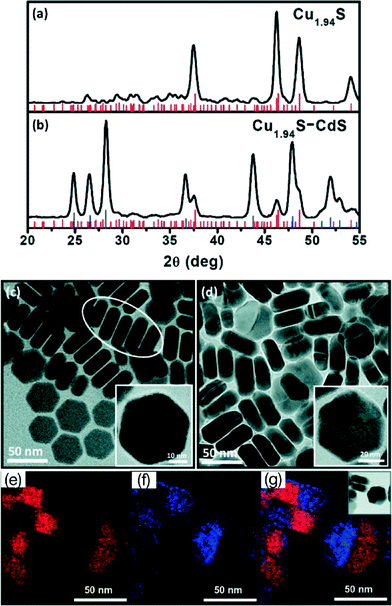
Cu1.94S–CdS and Cu1.94S–ZnxCd1−xS nanodisc heterostructures were synthesized by Regulacio et al. in 2011.37 The resulting nanodiscs exhibit a hexagonal shape and are composed of copper sulphide and CdS (or ZnxCd1−xS) regions (Fig. 11). Because of the conservation of the original hexagonal shape of the copper sulphide particles formed in the beginning of the synthesis the authors assume cation exchange to be the reaction mechanism for the generation of the nanostructures.
The formation of PbS–Cu2S hybrid nanorods was reported by Zhuang et al.38 in 2012. Addition of a Pb precursor to djurleite copper sulphide seeds resulted in the formation of cubic PbS nanorods attached to the hemispheres of copper sulphide with a chalcocite structure (Fig. 12). Thus, a phase transformation occurred in the copper sulphide particles during the formation of the PbS nanorods. The authors assume that the formation of PbS seeds is a consequence of partial cation exchange, while further growth results from the deposition of Pb and S monomers on the surface of these PbS seeds.
 | ||
| Fig. 12 TEM images of (a) Cu1.94S and (b) Cu2S–PbS heteronanostructures. Below the TEM images, a scanning TEM (STEM) image and the high resolution element mapping of Cu2S–PbS heteronanostructures are shown. The scale bars are all 40 nm. Adapted from ref. 38 with permission from The Royal Society of Chemistry. | ||
A similar growth mechanism based on partial cation exchange and subsequent growth on the surface of the newly formed seeds was reported for the Cu1.94S–MnS nanoheterostructures by Zhou et al.39 MnS formed in this reaction had a wurtzite structure, while control experiments without copper sulphide resulted in the formation of the cubic modification of MnS.
Conclusions
Copper sulphide based hybrid nanocrystals are an interesting class of materials with application potential in solar energy conversion, optoelectronics, and theranostics. Furthermore, copper sulphide seeds can be used to synthesize and control the shape of materials which are interesting for many applications, especially in solar energy conversion. Copper sulphide based hybrid nanocrystals can be synthesized starting with seeds exhibiting different crystallographic structures. The most commonly used are monoclinic djurleite seeds; however, roxbyite and chalcocite seeds were also reported. Further investigations will be necessary to find out if the crystallographic structure of the seeds can influence the growth process of the hybrid nanocrystals. Also the influence of the shape of the seeds has not been studied yet. Preparation of hybrid nanocrystals using seeds with different shapes could open up new possibilities of shape control of such heteronanostructures. Furthermore, the reaction mechanisms suggested in different studies differ from one another and need to be studied in more detail, e.g., by in situ techniques. A deeper understanding of the underlying growth process will enable more precise control of the properties of the hybrid nanostructures and a more efficient exploitation of their application potential.Notes and references
- C. B. Murray, C. R. Kagan and M. G. Bawendi, Annu. Rev. Mater. Sci., 2000, 30, 545–610 CrossRef CAS.
- K. L. Sowers, B. Swartz and T. D. Krauss, Chem. Mater., 2013, 25, 1351–1362 CrossRef CAS.
- J. H. Han, S. Lee and J. Cheon, Chem. Soc. Rev., 2013, 42, 2581–2591 RSC.
- J. Park, J. Joo, S. G. G. Kwon, Y. Jang and T. Hyeon, Angew. Chem., Int. Ed., 2007, 46, 4630–4660 CrossRef CAS PubMed.
- P. Samokhvalov, M. Artemyev and I. Nabiev, Chem. – Eur. J., 2013, 19, 1534–1546 CrossRef CAS PubMed.
- D. Aldakov, A. Lefrançois and P. Reiss, J. Mater. Chem. C, 2013, 1, 3756–3776 RSC.
- J. Kolny-Olesiak and H. Weller, ACS Appl. Mater. Interfaces, 2013, 5, 12221–12237 CAS.
- E. Witt and J. Kolny-Olesiak, Chem. – Eur. J., 2013, 19, 9746–9753 CrossRef CAS PubMed.
- D. V. Talapin, J.-S. Lee, M. V. Kovalenko and E. V. Shevchenko, Chem. Rev., 2010, 110, 389–458 CrossRef CAS PubMed.
- A. Chen and S. Chatterjee, Chem. Soc. Rev., 2013, 42, 5425–5438 RSC.
- D. Wang and Y. Li, Adv. Mater., 2011, 23, 1044–1060 CrossRef CAS PubMed.
- B. Kim, M. Hackett, J. Park and T. Hyeon, Chem. Mater., 2014, 26, 59–71 CrossRef CAS.
- L. Carbone and P. D. Cozzoli, Nano Today, 2010, 5, 449–493 CrossRef CAS PubMed.
- R. Costi, A. E. Saunders and U. Banin, Angew. Chem., Int. Ed., 2010, 49, 4878–4897 CrossRef CAS PubMed.
- P. D. Cozzoli, T. Pellegrino and L. Manna, Chem. Soc. Rev., 2006, 35, 1195–1208 RSC.
- C. de Mello Donegá, Chem. Soc. Rev., 2011, 40, 1512–1546 RSC.
- L. Chen, Y.-B. Chen and L.-M. Wu, J. Am. Chem. Soc., 2004, 126, 16334–16335 CrossRef CAS PubMed.
- Y. Wu, C. Wadia, W. Ma, B. Sadtler and P. Alivisatos, Nano Lett., 2008, 8, 2551–2555 CrossRef CAS PubMed.
- H. Zhang, Y. Zhang, J. Yu and D. Yang, J. Phys. Chem. C, 2008, 112, 13390–13394 CAS.
- W. P. Lim, C. T. Wong, S. L. Ang, H. Y. Low and W. S. Chin, Chem. Mater., 2006, 18, 6170–6177 CrossRef CAS.
- Y. Wang, Y. Hu, Q. Zhang, J. Ge, Z. Lu, Y. Hou and Y. Yin, Inorg. Chem., 2010, 6601–6608 CrossRef CAS PubMed.
- M. Kruszynska, H. Borchert, A. Bachmatiuk, M. H. Rümmeli, B. Buechner, J. Parisi and J. Kolny-Olesiak, ACS Nano, 2012, 6, 5889–5896 CrossRef CAS PubMed.
- I. Kriegel, J. Rodríguez-Fernández, E. Da Como, A. A. Lutich, J. M. Szeifert and J. Feldmann, Chem. Mater., 2011, 23, 1830–1834 CrossRef CAS.
- S. T. Connor, C.-M. Hsu, B. D. Weil, S. Aloni and Y. Cui, J. Am. Chem. Soc., 2009, 131, 4962–4966 CrossRef CAS PubMed.
- M. Kruszynska, H. Borchert, J. Parisi and J. Kolny-Olesiak, J. Am. Chem. Soc., 2010, 132, 15976–15986 CrossRef CAS PubMed.
- J.-Y. Chang and C.-Y. Cheng, Chem. Commun., 2011, 47, 9089–9091 RSC.
- M. Kruszynska, H. Borchert, J. Parisi and J. Kolny-Olesiak, J. Nanopart. Res., 2011, 13, 5815–5824 CrossRef CAS.
- X. Lu, Z. Zhuang, Q. Peng and Y. Li, CrystEngComm, 2011, 13, 4039–4045 RSC.
- Q. Li, L. Zhai, C. Zou, X. Huang, L. Zhang, Y. Yang, X. Chen and S. Huang, Nanoscale, 2013, 5, 1638–1648 RSC.
- C. Coughlan, A. Singh and K. Ryan, Chem. Mater., 2013, 25, 653–661 CrossRef CAS.
- W. Zhang, L. Zhai, N. He, C. Zou, X. Geng, L. Cheng, Y. Dong and S. Huang, Nanoscale, 2013, 5, 8114–8121 RSC.
- H.-C. Liao, M.-H. Jao, J.-J. Shyue, Y.-F. Chen and W.-F. Su, J. Mater. Chem. A, 2013, 1, 337 CAS.
- L. Yi, A. Tang, M. Niu, W. Han, Y. Hou and M. Gao, CrystEngComm, 2010, 12, 4124 RSC.
- F. Huang, X. Wang, J. Xu, D. Chen and Y. Wang, J. Mater. Chem., 2012, 22, 22614 RSC.
- S.-K. Han, M. Gong, H.-B. Yao, Z.-M. Wang and S.-H. Yu, Angew. Chem., Int. Ed., 2012, 51, 6365–6368 CrossRef CAS PubMed.
- H. Ye, A. Tang, L. Huang, Y. Wang, C. Yang, Y. Hou, H. Peng, F. Zhang and F. Teng, Langmuir, 2013, 29, 8728–8735 CrossRef CAS PubMed.
- M. D. Regulacio, C. Ye, S. H. Lim, M. Bosman, L. Polavarapu, W. L. Koh, J. Zhang, Q.-H. Xu and M.-Y. Han, J. Am. Chem. Soc., 2011, 133, 2052–2055 CrossRef CAS PubMed.
- T.-T. Zhuang, F.-J. Fan, M. Gong and S.-H. Yu, Chem. Commun., 2012, 48, 9762–9764 RSC.
- J. Zhou, F. Huang, J. Xu and Y. Wang, CrystEngComm, 2013, 15, 4217 RSC.
- W. Han, L. Yi, N. Zhao, A. Tang, M. Gao and Z. Tang, J. Am. Chem. Soc., 2008, 130, 13152–13161 CrossRef CAS PubMed.
- H. J. Fan, P. Werner and M. Zacharias, Small, 2006, 2, 700–717 CrossRef CAS PubMed.
- Y.-Z. Long, M. Yu, B. Sun, C.-Z. Gu and Z. Fan, Chem. Soc. Rev., 2012, 41, 4560–4580 RSC.
- S. Hoshino, Solid State Ionics, 1991, 48, 179–201 CrossRef CAS.
- K. Wakamura and I. Tsubota, Solid State Ionics, 2000, 130, 305–312 CrossRef CAS.
- L.-W. Wang, Phys. Rev. Lett., 2012, 108, 085703 CrossRef.
- S.-H. Choi, E.-G. Kim and T. Hyeon, J. Am. Chem. Soc., 2006, 128, 2520–2521 CrossRef CAS PubMed.
- L. Yi, Y. Liu, N. Yang, Z. Tang, H. Zhao, G. Ma, Z. Su and D. Wang, Energy Environ. Sci., 2013, 6, 835–840 CAS.
- P. K. Jain, L. Amirav, S. Aloni and P. Alivisatos, J. Am. Chem. Soc., 2010, 132, 9997–9999 CrossRef CAS PubMed.
- Y. Zhao and C. Burda, Energy Environ. Sci., 2012, 5, 5564–5576 CAS.
- D. Pan, L. An, Z. Sun, W. Hou, Y. Yang, Z. Yang and Y. Lu, J. Am. Chem. Soc., 2008, 130, 5620–5621 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |