Novel selective catalyst derived from uniform clustered NaY zeolite microspheres†
Wei Gang Lina, Mi Mi Wana, Yu Zhoua, Hui Cheng Gua, Shi Lu Zhoub and Jian Hua Zhu*a
aKey Laboratory of Mesosopic Chemistry, College of Chemistry and Chemical Engineering, Nanjing University, Nanjing 210093, China. E-mail: jhzhu@nju.edu.cn; Fax: +86-25-83317761; Tel: +86-25-83595848
bChina Tobacco Shandong Industrial Co. Ltd, Tsinan 250000, China
First published on 26th March 2013
Abstract
The design and preparation of a new “self-protected” zeolite catalyst are reported in this paper for the first time, in order to face the challenges arising from environmental protection, in selective catalytic degrading of tobacco specific nitrosamine (TSNA) carcinogens in environmental tobacco smoke (ETS). A new method to synthesize NaY zeolite with uniform microsphere morphology via an organic amine TMAOH as bifunctional template is reported in this study. The spherical zeolite is prepared by a one-step hydrothermal crystallization without any post-treatment or other shape-directing agent. It has a rugged surface like a cluster of crystal grains and its crystallization process is monitored in detail by means of XRD and SEM. To assess how the zeolite morphology affects its catalytic performance in complex systems, this material was directly mixed with tobacco shred and added into cigarettes to in situ degrade TSNA in smoke. As a result, the clustered zeolite microsphere can avoid the interference of particulate matter in smoke and shows a high selectivity in the removal of TSNA. This synthetic method offers a novel way of preparing zeolites with a spherical morphology, which has great potential application in the fields of catalysis, adsorption and separation.
Introduction
Special morphology endows heterogeneous catalysts with a new performance, especially for zeolite materials. With the ability of shape-selectivity, high thermal and hydrothermal stability, low toxicity and fine biocompatibility, zeolites have been used in various fields such as drug release, environmental protection and human health care,1–6 apart from the traditional applications of adsorption, separation, heterogeneous catalysis and ion exchange. And the strict requirement of new potential applications such as environmental catalysis spurs the preparation of new zeolite materials. One noteworthy example is the catalysis in tobacco smoke to selectively reduce the carcinogen tobacco specific nitrosamines (TSNA), protecting human health from the risk of environmental tobacco smoke.7–9 In principle these zeolite catalysts can be put into the tobacco shred of a cigarette, and in situ eliminate various carcinogens such as TSNA in smoke when the tobacco is burnt.7–10 However, this objective becomes challenging for zeolites, not only because of the existence and interference of thousands of compounds in smoke but also the special location of TSNA that adheres on the particle matter. To avoid unnecessary trapping of particles that may cover the surface of the catalyst causing active site wastage,11 it is crucial to design and prepare new “self-protected” zeolite catalysts, through controlling the morphology of the zeolite to change the contacting model with the particulate matter in tobacco smoke, rebounding these particles but capturing the TSNA adhered them.Faujasite (FAU) zeolite, type X or Y with a well-defined structure, large pore volume, high Al content and high ion-exchange capacity, is a versatile catalyst or adsorbent in many processes such as oil refining and aromatics separation.1,12,13 Moreover, zeolite NaY can be a candidate for degrading TSNA in tobacco smoke due to its large micropores and high catalytic activity.7,8,10 However, the tiny crystal grains of zeolite NaY always gather together on tobacco leaf to form aggregations with many cavities in the micrometer scale,7 whose size is close to that of particulate matter in smoke.14 Consequently, these cavities often trap the particles in mainstream smoke, lowering the value of total particulate matter (TPM) and deactivating the catalyst. Thus, the morphology of the catalyst seriously affects the selective removal of TSNA in tobacco smoke. Zeolite NaA with the large crystal grains can avoid such unwanted capture of TPM, but its activity is limited by its small pore size.7 Therefore, construction of NaY with large crystal grains sized at the micrometer level is urgent to fabricate the “self-protected” zeolite with higher selectivity towards TSNA, not only for controlling environmental pollution caused by smoking that is well known to be a global problem, but also for the development of a new efficient catalyst in such extreme complex systems.
Microspheres are the most popular configuration for the preparation of zeolites with large particles, mostly via assembly of zeolite nano-grains. Various types of zeolites with microsphere morphology have been prepared not only for their advantages of limited attrition, easy handling and recycling in catalysis,15 but also for their potential applications in chromatographic separation which requires the carrier to be, for the best, spherical in morphology with a uniform size in the micrometer range.16–18 Usually, hard-templating methods are preferable in preparing spherical zeolites. Tosheva et al. synthesized zeolites silicate-1 and Beta via anion exchanged resin beads as shape-directing templates.19–21 The zeolite crystallized within the pore of the resins and was finally obtained with the size and shape similar to the initial resin beads. Similarly, Kang et al. applied the polymerization-induced colloidal aggregation (PICA) method to synthesize the zeolite Beta and ZSM-5 by utilizing urea–formaldehyde (UF) resin as co-polymerization caster.18 The final sphere was assembled by nanozeolite grains and had a high adsorption capacity and a rapid adsorption rate towards large proteins. Afterwards, Shi et al. simplified this method by directly adding urea and formaldehyde precursor into the as-obtained solution of colloidal zeolite after hydrothermal crystallization.22 However, all of the spherical zeolites were obtained with the assistance of shape-directing template and they had low or no content of aluminum; the high Al content Y type zeolites with uniform microsphere morphology have not yet been reported, because of the difficulty in assembling the primary crystal grains with high Al content.
In this article, we successfully synthesized the spherical NaY zeolite with adjustable uniform size at the micrometer level for the first time. The zeolite sphere with the Si/Al of about 1.8 was assembled by small crystal grains, like a cluster. The organic amine TMAOH was used here as a bifunctional template acting both as the template of zeolite Y and the splicing agent for the formation of microspheres. The clustered zeolite microsphere (CZM) was obtained by only one step of hydrothermal crystallization without any post-treatment or other shape-directing agent. The ingredient and the addition sequence were investigated in detail to search the optimal synthetic conditions. Moreover, this material was directly mixed with tobacco shred to check its actual performance of selectively reducing TSNA in smoke that is flooded with thousands of compounds and numerous particulate matters, compared to the common NaY zeolite with tiny crystal grains.
Experimental section
Reagents
Zeolite NaY with the Si/Al ratio of 2.33 is commercially available powder sample bought from Catalyst Plant of Nankai University (China). Amorphous silica (400 meshes) was the product of Qingdao Haiyang (China). Tetramethyl ammonium hydroxide (TMAOH, 25% aqueous solution) and NaAlO2 were purchased from Sinopharm Chemical. Other regents with A.R. purity were used as received. Both commercially available Burley type tobacco and cigarette pipe were provided by Tobacco Shandong (China).Synthesis of samples
To synthesize the sample of clustered zeolite microspheres (CZM), sodium silicate and sodium aluminate were used as the silica and aluminum source, respectively. Sodium silicate solution was prepared by dissolving 11.04 g SiO2 and 8.48 g NaOH into 32.4 g deionized water, denoted as solution A; meanwhile 3.28 g NaAlO2 was dissolved into deionized water to get solution B. Then, solution A was added dropwise into solution B under vigorous stirring at 293 K to obtain a milky gel. After the mixture was stirred for 0.5 h, the calculated amount of TMAOH (25%) aqueous solution was added accompanied with vigorous stirring for another 0.5 h. The final composition of the synthetic gel was 0.684Na2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xTMA+
xTMA+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1SiO2
1SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.108Al2O3
0.108Al2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22.6H2O, which was aged at 363 K for a period of time (in most cases for 20 h). Finally the solid product was collected by filtering and washing until the filtrate was neutral, followed by drying and calcination at 823 K for 5 h. The calcined sample was denoted as CYx where x presents the ratio of TMA+ to SiO2 in the synthetic gel. For comparison, the amount of NaAlO2 additive was varied in the synthesis to adjust the Si/Al ratio, and these obtained samples were named as CYx(y) where y refers to different Si/Al ratios in the synthetic gel.
22.6H2O, which was aged at 363 K for a period of time (in most cases for 20 h). Finally the solid product was collected by filtering and washing until the filtrate was neutral, followed by drying and calcination at 823 K for 5 h. The calcined sample was denoted as CYx where x presents the ratio of TMA+ to SiO2 in the synthetic gel. For comparison, the amount of NaAlO2 additive was varied in the synthesis to adjust the Si/Al ratio, and these obtained samples were named as CYx(y) where y refers to different Si/Al ratios in the synthetic gel.Characterization
X-ray diffraction (XRD) patterns of samples were recorded on an ARL XTRA diffractometer with Cu Kα radiation in the 2-theta range of 5–90°. Nitrogen adsorption of the sample at 77 K was measured on a Micromeritics ASAP 2020 volumetric adsorption analyzer, and the sample was outgassed at 573 K before the test. The BET specific surface area of sample was calculated using adsorption data acquired at a relative pressure (p/p0) range of 0.05–0.22 and the total pore volume determined from the amount adsorbed at a relative pressure of 0.99. The pore size distribution of sample was calculated from the desorption branch using the Barret–Joyner–Halenda (BJH) method, and its chemical composition was detected by the X-ray Fluorescence (XRF) method using ARL-9800 X-ray fluorescence spectrometer. The morphology of the sample was observed by scanning electron microscopy (SEM) using a Hitachi S4800 FE-SEM system with 10 kV accelerating voltage and 0.01 mA of beam current for imaging. TG-DSC analysis was performed on a Netzsch STA449C TG/DSC analyzer from 303 K to 1123 K in air with a heating rate of 10 K min−1.Smoking test
The smoking experiment was carried out as reported previously:7,8 750 mg Burley type fine cut tobacco and 30 mg powder zeolite were mixed manually until homogeneous, and then hand-made into a cigarette by use of a commercial cigarette pipe with a filter tip of cellulose acetate fiber. For comparison, the control cigarette was prepared using the same procedure without zeolite. These hand-made cigarettes were selected to ensure their error of weight and pressure drop below ±2% and ±5%, respectively, and then conditioned for at least 48 h at 295 K and 60% relative humidity (R. H.) before the smoking test. The smoking process was performed on a SM450 smoking machine (Borgwaldt) under ISO standard conditions (35 cm3 puff, 2 s duration every 60 s) with 10 cigarettes for each sample, the mainstream smoke passed through one Cambridge filter pad where the particle phase in smoke was captured. This filter pad was extracted by 20 mL ammonium acetate solution and the obtained extractive solution was further cleaned-up by using the Agela PCX extraction column and finally cleaned-up with a filter membrane (0.22 μm). An Agilent 1200 LC system along with the Applied Biosystems API 5000 mass spectrometer were used to determine the TSNA content in each sample, with the assistance of internal standard method.23Results and discussion
The formation of clustered zeolite microspheres
Fig. 1 reveals the wide-angle XRD patterns of the CZM samples synthesized with different TMAOH amounts before calcination. There was no crystalline phase in the absence of TMAOH under this synthetic procedure. When a relative low ratio of TMAOH to silicon was used (0.025), nepheline was the primary phase in the XRD pattern.24 The (1 0 0) diffraction peak of FAU zeolite appeared subsequently when the TMAOH/Si ratio elevated. Further increasing this ratio generated more obvious phases of FAU zeolite, and the nepheline structure disappeared when the ratio reached 0.2. Fig. 2 depicts the influence of crystallization time on zeolite formation with a TMAOH/Si ratio of 0.2 in the synthetic gel. Only amorphous aluminosilicate species were observed in the XRD pattern below 6 h of crystallization, and the nepheline structure began to appear when the synthetic composition was aged for 8 h. The nepheline phase was maintained on the sample crystallized for 12 h but disappeared on that for 15 h or more; meanwhile the FAU structure started to emerge on the sample aged for 12 h, and the crystallinity kept increasing as the aging time was prolonged up to 20 h. Scheme 1 illustrates the strange formation of FAU structure in these synthetic conditions, which is characterized with nepheline as an intermediate phase. Actually this phase quickly transforms to FAU zeolite which consists of sodalite units connected by double six rings. The 12th hour of crystallization seems to be a transition state from nepheline to FAU zeolite because of the coexistence of two phases in the XRD patterns. | ||
| Fig. 1 The wide-angle XRD patterns of clustered zeolite microspheres (before calcination) synthesized using different amounts of TMAOH. | ||
 | ||
Fig. 2 The wide-angle XRD patterns of clustered zeolite microspheres (before calcination) synthesized at the TMAOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Si ratio of 0.2 with different crystallization times. Si ratio of 0.2 with different crystallization times. | ||
 | ||
| Scheme 1 Formation process of FAU structure in CY sample. | ||
The relative crystallinities of the samples synthesized under different conditions are shown in Fig. 3, and the degree of crystallinity is indicated by the strength of (1 0 0) the peak of FAU zeolite in the XRD patterns.25 The sample with the TMAOH/Si ratio of 0.4 exhibited the strongest (1 0 0) diffraction peak, which is regarded as 100% relative crystallinity in this case. It is clear that using more organic template TMAOH in the synthesis led to a more intact crystallization (Fig. 3A). Especially, the relationship between the TMAOH/Si ratio and the relative crystallinity is almost linear when the ratio was below 0.2, mirroring the importance of organic amine template to the formation of the zeolite phase in the synthesis. Fig. 3B displays the crystallization curve of the zeolite in the synthesis with a TMAOH/Si ratio of 0.2. It followed the typical ‘S’ shape of zeolite crystallization curve with an inducing time of about 8 h and the crystallinity reached a summit after 20 h, implying the completion of crystallization.
 | ||
| Fig. 3 Relative crystallinity of the zeolite microsphere samples synthesized by (A) using different amounts of TMAOH and (B) crystallizing for different times (crystallization curve). | ||
Fig. 4 illustrates the SEM images of uncalcined CZM samples synthesized with different amounts of TMAOH addition, in comparison with that of normal NaY zeolite (Fig. S1a†). Only irregular amorphous particles were observed in the absence of TMAOH (Fig. 4a). When a little bit of TMAOH (0.025![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to Si) was added, a few spheres could be found by careful observation (Fig. S1b†). The regular microsphere morphology emerged at the TMAOH/Si ratio of 0.05 (Fig. 4b) although there was no intact FAU phase in the XRD pattern (Fig. 1). By close inspection it can be seen that the microspheres were quite uniform and constructed by many small crystal grains, just like clusters. As the amount of organic amine template increased in the synthesis, these crystal grains grew bigger and angular, making the mellow surface of the sphere become rugged (Fig. 4c–f). The amount of TMAOH also influenced the size of the clustered microspheres. When the ratio of TMAOH/Si was 0.05–0.2, the diameter of the spheres was about 4–6 μm; as this ratio elevated to 0.3 and 0.4, the diameter obviously decreased to 2–3 μm and 1.5 μm, respectively. It means that the size of CZM sample can be adjusted according to the amount of organic amine template added to the synthetic gel.
1 to Si) was added, a few spheres could be found by careful observation (Fig. S1b†). The regular microsphere morphology emerged at the TMAOH/Si ratio of 0.05 (Fig. 4b) although there was no intact FAU phase in the XRD pattern (Fig. 1). By close inspection it can be seen that the microspheres were quite uniform and constructed by many small crystal grains, just like clusters. As the amount of organic amine template increased in the synthesis, these crystal grains grew bigger and angular, making the mellow surface of the sphere become rugged (Fig. 4c–f). The amount of TMAOH also influenced the size of the clustered microspheres. When the ratio of TMAOH/Si was 0.05–0.2, the diameter of the spheres was about 4–6 μm; as this ratio elevated to 0.3 and 0.4, the diameter obviously decreased to 2–3 μm and 1.5 μm, respectively. It means that the size of CZM sample can be adjusted according to the amount of organic amine template added to the synthetic gel.
 | ||
| Fig. 4 SEM images of the clustered zeolite microspheres (before calcination) synthesized using TMAOH/Si ratios of (a) 0, (b) 0.05, (c) 0.1, (d) 0.2, (e) 0.3 and (f) 0.4. (The crystallization time is 20 h). | ||
Fig. 5 displays the SEM images of uncalcined CZM samples with different crystallization times, which reveals the formation procedure of the special morphology. Before 6 h of crystallization, only irregular fragments can be observed (Fig. 5a). The intact spheres with mellow and the smooth surfaces firstly emerged at the 8th hour and they were surrounded by large amount of amorphous particles (Fig. 5b). When the aging time was prolonged from 8 h to 12 h, the sphere grew bigger from about 1.5 μm to 3 μm (Fig. 5c). Further prolonging the aging time to 15 h resulted in microspheres of 4–5 μm (Fig. 5d), meanwhile the mellow sphere began to become angular and rugged, like a cluster of grains. At the same time, the amorphous species scattering around the microsphere were consumed during this process of crystallization, remaining much less on the sample aged for 18 h than that for 15 h (Fig. 5e). Amorphous particles completely disappeared in the SEM image of the sample with a crystallization of 20 h (Fig. 5f), leaving only microspheres that were identified as NaY zeolite.
 | ||
Fig. 5 The SEM images of clustered zeolite microspheres (before calcination) synthesized at the TMAOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Si ratio of 0.2 with different crystallization times. Si ratio of 0.2 with different crystallization times. | ||
Textural properties
Fig. 6 shows the TG-DTA profiles of the as-synthesized samples with different TMAOH/Si ratios in the synthesis. The profiles had two steps in which the first one appeared below 573 K was assigned to desorption of water while the second step from 623 to 923 K corresponded to the decomposition of the organic template TMAOH.18 Sample CY0.1 lost less water than others during the heating process, probably due to its incomplete crystallization of zeolite structure. The weight loss in the 623–923 K stage rests with the amount of organic amine template added into the initial gel: 0.79% and 0.84% for CY0.1 and CY0.2 while 1.01% and 1.67% for CY0.3 and CY0.4, respectively. After removal of the template, CY samples kept the same structure and morphology as that before calcination (Fig. S2 and S3†), which proves the stability of this morphology during calcination. Table 1 lists the textural properties of calcined CY samples, in which all of them (CY0.05 ∼ CY0.4) have the Si/Al ratio around 1.8 which is characteristic of zeolite Y. Sample CY0.05 had very small surface area and pore volume due to its poor crystallization; and these parameters were slightly enlarged on the CY0.1 sample, but still much lower than that of the common zeolite NaY, owing to its partial transformation of the zeolite phase. Both surface area and pore volume of the spherical zeolites CY0.2–0.4 exceeded 700 m2 g−1 and 0.33 cm3 g−1 respectively, which were similar to that of normal NaY to confirm the relative completion of crystallization. To explore the influence of the Si/Al ratio in the synthetic gel on the structure and morphology of the final products, a series of samples with different Si/Al ratios in the synthesis were characterized. Sample CY0.2(9.2) generated the Si/Al ratio of 2.0, but its zeolitic structure was quite poor (Fig. S4†) and its morphology was also untidy (Fig. S5†). The Si/Al ratio of CY0.2(2.3) sample was about 1.7, and its XRD pattern showed the well structure of FAU zeolite but it formed an irregular sphere morphology with many amorphous particles around. When the Si/Al ratio in the synthetic gel decreased to 1 for the CY0.2(1) sample, LTA zeolite formed with a Si/Al ratio of about 1 (Fig. S4,† and Table 1). It is clear that the initial Si/Al ratio in the synthetic gel is very important for the formation of microsphere morphology and the value of 4.6 is appropriate; but the Si/Al ratio in the synthesis has little influence on the final composition of the CY sample whose Si/Al ratio was kept in a small range of 1.7 to 2.0 although that in the synthetic gel varied from 2.3 to 9.2. | ||
| Fig. 6 DG-DTA curves of the clustered zeolite microsphere samples. | ||
| Sample | SBET (m2 g−1) | Vtotal (cm3 g−1) | Smicroa (m2 g−1) | Vmicroa (cm3 g−1) | Si/Al |
|---|---|---|---|---|---|
| a t-plot micropore area and micropore volume. | |||||
| CY0.05 | 41 | 0.051 | 21 | 0.010 | 1.86 |
| CY0.1 | 229 | 0.124 | 204 | 0.095 | 1.83 |
| CY0.2 | 715 | 0.340 | 673 | 0.314 | 1.79 |
| CY0.3 | 701 | 0.338 | 656 | 0.306 | 1.81 |
| CY0.4 | 740 | 0.363 | 690 | 0.322 | 1.85 |
| CY0.2 (9.2) | — | — | — | — | 2.06 |
| CY0.2 (2.3) | — | — | — | — | 1.68 |
| CY0.2 (1) | — | — | — | — | 1.06 |
| NaY | 734 | 0.355 | 690 | 0.322 | 2.33 |
Fig. 7 exhibits the nitrogen adsorption isotherms and the BJH pore distribution of CY samples. An obvious uptake appeared on CY0.05 at high relative pressure (0.85–1.0), indicating macropores formed in this sample by packing of aggregated particles;26 and which was confirmed by the wide band from 20 to 120 nm in the pore distribution. Sample CY0.1, CY0.2 and zeolite NaY showed the typical type I isotherm of microporous materials. However, sample CY0.3 and CY0.4 revealed the IV type isotherms with a slight H4 hysteresis loop from the relative pressure of 0.45 to 0.9. This phenomenon implies the relative uniform slit-shaped intercrystal mesopores with a narrow distribution at 3.9 nm, which was probably generated from the aggregation of zeolite nanocrystals.26 Even so, the proportion of mesopore in these samples was fairly low.
 | ||
| Fig. 7 N2 adsorption–desorption isotherms (A) and BJH pore size distribution (B) of CY samples. | ||
Selective degradation of TSNA in smoke
In order to assess the specificity of clustered NaY microspheres in the catalytic degradation of carcinogen TSNA in tobacco smoke, a smoking experiment is performed by separately mixing CY and normal NaY zeolite with tobacco shred as catalysts,7,8 in order to test their ability of in situ TSNA degradation. The basic parameters of the smoking experiment are listed in Table S1.† The dispersion of composites on tobacco shred is displayed in Fig. 8, in which the surface of the tobacco shred itself is relatively smooth with some wrinkles (Fig. 8a). The spherical particles of the CY sample are dispersed individually and as monolayers on the surface of tobacco shred, without overlap and accumulation (Fig. 8b and c); while the tiny grains of common NaY always form aggregations and construct many micrometer cavities which can trap the particulate matter in an airflow when mainstream smoke passes7 (Fig. 8d). | ||
| Fig. 8 SEM images of (a) tobacco shred surface and (b) CY0.2, (c) CY0.3, (d) NaY samples dispersed on tobacco shred. | ||
Table 2 lists the TPM value and TSNA content in the mainstream smoke of sample cigarettes collected in the smoking experiment. As a result, the TPM amount of the cigarette containing the CY sample seems similar to that of the control, but that with zeolite NaY is lowered by 13%, from 13.8 to 12.0 mg cig−1. This difference mainly resulted from the morphologic diversity between the CY sample and NaY zeolite, because the cavities caused by the aggregation of zeolite will capture the particulate matter in mainstream smoke.7 Moreover, the ability of CY samples to selectively degrade TSNA in smoke is still prominent. The data of TSNA/TPM in Table 2 represents the TSNA amount in unit weight of particulate matter and the reduction of this value judges the selectivity of removing TSNA from the particle phase in smoke.8 All of the cigarettes containing zeolite catalyst could reduce the TSNA content in smoke, whereas CY0.1 performed the worst (only from 10.29 to 9.21 ng mg−1) due to its incomplete crystallization of the zeolite structure; while CY0.2 was the best in selectively removing TSNA (to 8.16 ng mg−1) in tobacco smoke. The function of CY0.3 (to 8.34 ng mg−1) and CY0.4 (to 8.47 ng mg−1) was also good, both higher than that of common NaY (to 8.53 ng mg−1). Since the specific morphology of CY samples is believed to have reduced the interference of particulate matter in smoke that usually causes the deactivation of catalyst or adsorbent,11 these composites exhibit a highly selective reduction of TSNA (for CY0.2–0.4 samples) compared to zeolite NaY although they all have the same FAU framework. This advantage of CY samples on selectively degrading TSNA in mainstream smoke makes them candidates for catalysts for controlling the pollution caused by smoking.
| Sample | CY0.1 | CY0.2 | CY0.3 | CY0.4 | NaY | Control |
|---|---|---|---|---|---|---|
| TSNA (ng/cig) | 123.5 | 108.53 | 110.1 | 113.47 | 102.41 | 141.95 |
| TPM (mg/cig) | 13.4 | 13.3 | 13.2 | 13.4 | 12.0 | 13.8 |
| TSNA/TPM (ng mg−1) | 9.21 | 8.16 | 8.34 | 8.47 | 8.53 | 10.29 |
| Reduction of TPM (%) | 2.90 | 3.62 | 4.35 | 2.90 | 13.04 | — |
| Reduction of TSNA (%) | 13.01 | 23.54 | 22.44 | 20.06 | 27.85 | — |
| Reduction of [TSNA/TPM] (%) | 10.45 | 20.70 | 18.94 | 17.71 | 17.0 | — |
Discussion
This new preparation route of the zeolite microsphere with high Al-content is developed with the absence of post treatment and other shape-directing agents. In the synthesis, the organic amine TMAOH plays a very important role not limited solely as the template of zeolite Y. The abundant charge of the organic amine enables it to be an adhesive gathering small particles together to form large spherical aggregations, similar to the im-PICA process.22 The same synthesis process but without TMAOH gave only the irregular particle products (Fig. 4a); also, a thimbleful of TMAOH additive (0.025 to Si) only led to very few spheres among large amounts of amorphous particles (Fig. S1b†). Accordingly, the organic amine functions as a splicing agent in the formation of this special morphology. And the interaction between TMAOH and Al species is the main bond to assemble the amorphous substances together, since halving the amount of the Al source in the synthetic gel of CY0.2 (9.2) resulted in nothing with a sphere morphology (Fig. S5b†).Intact and uniform spheres can be observed in the SEM images of CY0.05 and CY0.1 samples, but their crystallization degree is relatively low, with an impure phase of zeolite mixed with nepheline (Fig. 1). It means that the crystallization of zeolite does not synchronize with the formation of the microspheres, but lags behind by a period of time. The crystallizing process of CY0.2 observed by SEM image also proved it: the zeolite phase firstly emerges on the sample aged for 12 h (Fig. 2), but the spheres appeared at the 8th hour (Fig. 5b). The spheres grow bigger and bigger as the aging time is prolonged, from about 2 μm at the 8th hour to 3 μm at the 12th hour, then to the final size of 4–6 μm after the 15th hour (Fig. 5). Actually the growth of the microsphere wraps the amorphous substance scattering around it, and these amorphous particles containing Al and Si species become less and less as observed in SEM images with prolonged aging time until they finally disappear after 20 h. Furthermore, the sphere surface is mellow before the 12th hour but become rugged after that, indicating the formation of zeolite crystals. It coincides with the crystallization curve in Fig. 3b where the crystallization degree of zeolite starts to burst after aging for about 12 h.
The schematic formation mechanism of clustered zeolite microsphere is illustrated in Scheme 2, taking CY0.2 as an example. The Si and Al species firstly form amorphous aluminosilicate particles in the colloidal solution via the formation of Si–O–Al bonds immediately after being mixed;27 and one amorphous particle contains one nucleus of zeolite crystal.2,28 These amorphous particles are gradually gathered, attached and assembled under the attraction of TMAOH as a splice agent; and when the synthetic gel has been aged at 363 K for 8 h, the spheres start to appear. These spheres grow bigger by wrapping more and more amorphous particles around them as the aging time is prolonged. The crystallization of the zeolite happens a bit later than the formation of the sphere morphology because of the hysteresis caused by the induction period,28 and it starts at about the 12th hour when the intact spheres have already existed, in which both nucleation and crystallization take place on the surface of the amorphous particles.28 As a result, the crystal growth consumes the amorphous aluminosilicate gel close to the sites of spontaneous mass crystallization,29 which can perform in two ways: the first one is gathering the scattering aluminosilicate particle around the microsphere and the second one is digesting the adjacent substance within the sphere, making a group of amorphous particles fuse into one piece of a relatively large crystal. The process continues until the amorphous nutrient is exhausted at about the 20th hour for CY0.2 samples, resulting in the morphology of microspheres with a rugged surface, like a cluster of NaY zeolite grains.
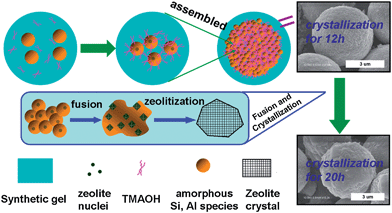 | ||
| Scheme 2 Possible formation mechanism of clustered zeolite microsphere sample. | ||
Critically, the formation of FAU structure in this case is a bit different from the common situation in which the sodalite precursor is preferred.30 The gel phase firstly causes tetrahedral single six-rings (S6R) built by units of TO4 that may be the precursor for the nucleation.31,32 And the S6R can gradually construct to different types of topologies: ‘…AA…’ as double six-rings (D6R), ‘…ABAB…’ as nepheline and ‘…ABCABC…’ as sodalite. As aforementioned, the aluminosilicate retains the structure of nepheline as an intermediate state in our situation, and then, with the assistance of the template TMAOH, it quickly transforms to the FAU structure which is connected by sodalite cages via D6R rings (Scheme 1). Since the assembly of primary amorphous particles in the synthetic gel relates to the attraction between TMAOH and Al species, the newly formed sphere-like aggregation consists of Al-rich particles, which can explain why the Si/Al ratio in the product is much lower than that in the initial gel. The stoichiometric sodium nepheline (Na[SiAlO4]) is rooted from the Al-rich aggregation at relatively high temperature which contains only six-rings of 3Si3Al.32 Although the possible ring structural arrangement can change the ring members to 4Si2Al, 5SiAl or even 6Si for the FAU structure32 by digesting some Si nutrition in the gel, the variation is kept to a small extent and the overall Si/Al ratio still holds at a relatively low degree (about 1.8). The Si/Al ratio is also stabilized at this level when halving or doubling the Al source: the Si/Al ratio only changes a little, to 1.7 and 2.0 for the sample CY0.2(9.2) and CY0.2(2.3), respectively.
The timing of TMAOH addition is also important for the formation of microsphere morphology. Generally, we added the organic amine after the mixing of solution A (Al source) and B (Si source), the sphere morphology is obtained. For comparison, we also did the synthesis by adding TMAOH into solution B before mixing with solution A. The XRD pattern and SEM image of this resulting sample are displayed in Fig. S6,† which shows irregular morphology without any spheres although the NaY structure exists. It indicates that the role of bifunctional template can only take effect after the mixing of Si and Al source when these two species have already spontaneously formed amorphous particles; otherwise the TMAOH will lose the function of splicing the scattering aluminosilicate gel into large spheres. Moreover, the amount of TMAOH influences the degree of relative crystallinity at the low content level (TMAOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Si = 0–0.1), and adjusts the size of microspheres at the high content level (TMAOH
Si = 0–0.1), and adjusts the size of microspheres at the high content level (TMAOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Si = 0.2–0.4). The reason is when the template is enough for the zeolite formation, the excess organic amine controls the alkalinity of the solution; and increasing the TMAOH amount will considerably elevate the alkalinity of the synthetic solution and result in the reduction of the crystal size.33
Si = 0.2–0.4). The reason is when the template is enough for the zeolite formation, the excess organic amine controls the alkalinity of the solution; and increasing the TMAOH amount will considerably elevate the alkalinity of the synthetic solution and result in the reduction of the crystal size.33
The special morphology of the CY zeolite endows the sample with the ability of self-protection in tobacco smoke containing lots of particulate matter and dramatically improves the performance of the catalyst. The “self-protection” means this CY sample can effectively resist the interference of particulate matter in tobacco smoke. As reported previously,7 NaA zeolite with isolated large crystals can rebound the aerosols in smoke (sized at the micrometer level) rather than capture them, but its weak catalytic activity deprives its morphology advantage. In contrast, NaY zeolite shows an excellent performance in catalysis but it has the inherent drawback, as illustrated in Scheme 3, the tiny grains of zeolite often construct some cavities to trap the particulate matter in tobacco smoke hence the catalyst is quickly covered by the oily substance and deactivated, meanwhile the total particulate matter (TPM) value of smoke is lowered significantly. With the special isolated large crystals such as that of zeolite NaA, CY sample can also rebound the particulate matter in smoke to avoid unnecessary capture, so it is able to reduce the TSNA level of smoke with only a minor effect on the TPM value. Spherical morphology endows CY zeolite the ability to exclude the unnecessary capture of particulate matter in smoke, as if it was self-protected. Moreover, the rugged surface of clustered spheres increases the probability of collision with the particles in smoke, which is beneficial for the removal of TSNA adhered on these particles.7,8 As expected, CY composites slightly reduced the TPM value in the smoking experiment (Table 2), but kept a high activity in removal of TSNA, performing better than commercial NaY to selectively remove TSNA in unit weight of particulate matter. These results allow CY sample to be a novel candidate added into tobacco shred to selectively reduce TSNA in smoke.
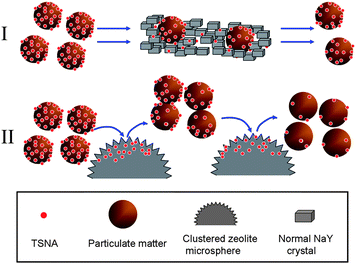 | ||
| Scheme 3 Removal of TSNA by (I) normal NaY and (II) clustered zeolite microsphere in tobacco smoke. | ||
This process of synthesizing zeolite with sphere morphology is not limited in the preparation of NaY zeolite using TMAOH as bifunctional template. And the effect of assembling microspheres by an organic amine template is applicable for other amines such as TPAOH, with the same procedure but different ingredients. It is hoped that this method can be used to develop a simple and fast universal approach for the synthesis of uniform spherical zeolites at the micrometer level, which has great potential not only as a “self-protected” zeolite catalyst, but also for the wider application in chromatographic separation and biochemistry.
Conclusions
NaY zeolite with uniform microsphere morphology was one-step synthesized via an organic amine TMAOH as bifunctional template. TMAOH acts as not only as the template of zeolite Y, but also as the splicing agent to assemble amorphous aluminosilicate particles to spheres. The size of microspheres can be adjusted in the range from 1.5 to 6 μm by varying the amount of TMAOH addition.The formation of sphere morphology was anterior to the crystallization of zeolite Y. And the process of crystallization went through the phase of nepheline as an intermediate state and finally transformed to the NaY structure as the hydrothermal aging time was prolonged. The nucleation and crystallization occurred on the amorphous species within or around the microspheres and made the mellow surface of the spheres become rugged like a cluster of crystal grains.
This CY material can be a substitute for zeolite NaY as catalyst added into tobacco shred because its highly dispersed distribution results in much less capture of TPM than the common NaY which has tiny crystal grains; and it exceeds the catalytic activity of common NaY in degrading strong carcinogen TSNA in tobacco smoke, offering a novel “self-protected” zeolite catalyst candidate for controlling the environmental pollution cause by smoking.
This study is our first approach to synthesize spherical NaY zeolite due to its difficulty of preparation and prominent catalytic activity in tobacco smoke. It is hoped that this method can be a simple and universal way for the synthesis of zeolite with uniform microsphere morphology.
Acknowledgements
Financial support from NSF of China (21173117 and 21273106), the Scientific Research Foundation of Graduate School, and Analysis Center of Nanjing University is gratefully acknowledged.Notes and references
- A. Corma, Chem. Rev., 1997, 97, 2373 CrossRef CAS.
- L. Tosheva and V. P. Valtchev, Chem. Mater., 2005, 17, 2494 CrossRef CAS.
- M. E. Argun, J. Hazard. Mater., 2008, 150, 587 CrossRef CAS.
- V. J. Inglezakis, M. A. Stylianou, D. Gkantzou and M. D. Loizidou, Desalination, 2007, 210, 248 CrossRef CAS.
- S. Fox, T. S. Wilkinson, P. S. Wheatley, B. Xiao, R. E. Morris, A. Sutherland, A. J. Simpson, P. G. Barlow, A. R. Butler, I. L. Megson and A. G. Rossi, Acta Biomater., 2010, 6, 1515 CrossRef CAS.
- F. Wei, J. Y. Yang, Q. Hou and J. H. Zhu, New J. Chem., 2010, 34, 2897 RSC.
- W. G. Lin, Y. Zhou, Y. Cao, M. M. Wan and J. H. Zhu, Catal. Today, 2012 DOI:10.1016/j.cattod.2012.07.045.
- W. G. Lin, Y. Zhou, F. N. Gu, S. L. Zhou and J. H. Zhu, Appl. Catal., B, 2013, 129, 301 CrossRef CAS.
- L. Gao, Y. Cao, S. L. Zhou, T. T. Zhuang, Y. Wang and J. H. Zhu, J. Hazard. Mater., 2009, 169, 1034 CrossRef CAS.
- W. M. Meier and K. Siegmann, Microporous Mesoporous Mater., 1999, 33, 307 CrossRef CAS.
- Y. Zhou, W. G. Lin, M. M. Wan, J. Yang and J. H. Zhu, J. Mater. Chem., 2012, 22, 23633 RSC.
- S. I. Zones, Microporous Mesoporous Mater., 2011, 144, 1 CrossRef CAS.
- D. Zhai, L. Zhao, J. S. Gao and C. M. Xu, Phys. Chem. Chem. Phys., 2012, 14, 7296 RSC.
- C. H. Keith, Beitr. Tabakforsch. Int., 1982, 11, 123 Search PubMed.
- G. Ertl, H. Knözinger and J. Weitkamp, Preparation of Solid Catalysis, Wiley-VCH, Weinheim, 1999, p. 4 Search PubMed.
- M. Matsui, Y. Kiyozumi, T. Yamamoto, Y. Mizushina, F. Mizukami and K. Sakaguchi, Chem.–Eur. J., 2001, 7, 1555 CrossRef CAS.
- F. Xu, Y. J. Wang, X. D. Wang, Y. H. Zhang, Y. Tang and P. Y. Yang, Adv. Mater., 2003, 15, 1751 CrossRef CAS.
- Y. J. Kang, W. Shan, J. Y. Wu, Y. H. Zhang, X. Y. Wang, W. L. Yan and Y. Tang, Chem. Mater., 2006, 18, 1861 CrossRef CAS.
- L. Tosheva, V. P. Valtchev and J. Sterte, Microporous Mesoporous Mater., 2000, 35–36, 621 CrossRef CAS.
- L. Tosheva, B. Mihailova, V. P. Valchev and J. Sterte, Microporous Mesoporous Mater., 2001, 48, 31 CrossRef CAS.
- V. Naydenov, L. Tosheva and J. Sterte, Chem. Mater., 2002, 14, 4881 CrossRef CAS.
- Y. Shi, X. Li, J. K. Hu, J. H. Lu, Y. C. Ma, Y. H. Zhang and Y. Tang, J. Mater. Chem., 2011, 21, 16223 RSC.
- K. A. Wagner, N. H. Finkel, J. E. Fossett and I. G. Gillman, Anal. Chem., 2005, 77, 1001 CrossRef CAS.
- A. Pasculli and D. Novembre, Comput. Geosci., 2012, 42, 110 CrossRef CAS.
- J. Gu, Y. J. Wu, J. Wang, Y. D. Lu and X. Q. Ren, J. Mater. Sci., 2009, 44, 3777 CrossRef CAS.
- Y. Huang, K. Wang, D. H. Dong, D. Li, M. R. Hill, A. J. Hill and H. T. Wang, Microporous Mesoporous Mater., 2010, 127, 167 CrossRef CAS.
- G. Harvey and L. S. Dent Glasser, ACS Symp. Ser., 1989, 398, 49 CrossRef CAS.
- S. Mintova, N. H. Olson and T. Bein, Angew. Chem., Int. Ed., 1999, 38, 3201 CrossRef CAS.
- V. P. Valtchev and K. N. Bozhilov, J. Phys. Chem. B, 2004, 108, 15587 CrossRef CAS.
- F. N. Gu, F. Wei, J. Y. Yang, N. Lin, W. G. Lin, Y. Wang and J. H. Zhu, Chem. Mater., 2010, 22, 2442 CrossRef CAS.
- H. Lechert and H. Kacirek, Zeolites, 1993, 13, 192 CrossRef CAS.
- S. Markovic, V. Dondur and R. Dimitrijevic, J. Mol. Struct., 2003, 654, 223 CrossRef CAS.
- G. S. Zhu, S. L. Qiu, J. H. Yu, Y. Sakamoto, F. S. Xiao, R. R. Xu and O. Terasaki, Chem. Mater., 1998, 10, 1483 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: XRD patterns and SEM images of calcined CY sample, CY samples synthesized using different amounts of Al source and different timings of TMAOH addition along with basic parameters of the smoking experiment. See DOI: 10.1039/c3ta10483d |
| This journal is © The Royal Society of Chemistry 2013 |
