Asymmetric catalytic arylation of ethyl glyoxylate using organoboron reagents and Rh(i)–phosphane and phosphane–phosphite catalysts†
Abstract
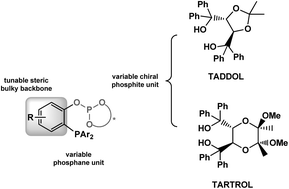
Herein we report the first application of Rh(I)–phosphane and phosphane–phosphite catalysts in the enantioselective catalytic arylation of ethyl glyoxylate with organoboron reagents, providing access to ethyl mandelate derivatives in high yield (up to 99%) and moderate to very good enantioselectivities (up to 75% ee). Commercial phosphane ligands, such as (R)-MonoPhos and (R)-Phanephos were tested, as well as non-commercial (R,R)-TADDOL-derived phosphane–phosphite ligands. Those ligands containing bulky substituents in the ortho-and para-positions of the chiral phosphite moiety were found to be the most selective.

 Please wait while we load your content...
Please wait while we load your content...