Gas-sensing properties of p-type α-Fe2O3 polyhedral particles synthesized via a modified polyol method
Nguyen Viet Long*abdef,
Yong Yang*b,
Masayoshi Yuasab,
Cao Minh Thif,
Yanqin Caob,
Thomas Nanng and
Masayuki Nogamibc
aState Key Laboratory of High Performance Ceramics and Superfine Microstructure, Shanghai Institute of Ceramics, Chinese Academy of Science, 1295, Dingxi Road, Shanghai 200050, China. E-mail: nguyenviet_long@yahoo.com; Fax: +86-21-52414219; Tel: +86-21-52414321
bDepartment of Materials Science, Faculty of Engineering Sciences, Kyushu University, Kasuga-koen 6-1, Kasuga-shi Fukuoka, 816-8580, Japan
cDepartment of Materials Science and Engineering, Nagoya Institute of Technology, Gokiso-cho, Showa-ku, Nagoya 466-8555, Japan
dPosts and Telecommunications Institute of Technology, km 10 Nguyen Trai, Hanoi, Vietnam
eLaboratory for Nanotechnology, Ho Chi Minh Vietnam National University, LinhTrung, Thu Duc, Ho Chi Minh, Vietnam
fHo Chi Minh City University of Technology, 144/24 Dien Bien Phu, Ward 25, BinhThach, Ho Chi Minh City, Vietnam
gIan Wark Research Institute, ARC Special Research Centre, University of South Australia, Australia
First published on 19th November 2013
Abstract
The gas sensing properties of polyhedral α-Fe2O3 particles were investigated. The polyhedral α-Fe2O3 particles were synthesized via a modified polyol method with the addition of an extra amount of NaBH4 in ethylene glycol, and fabricated to a thick gas sensing film after calcination at 500 °C and 900 °C. The polyhedral α-Fe2O3 particles exhibited a p-type nature, which is a new result opposite to other results of recent reports of semiconductor gas sensors using α-Fe2O3. In particular, we suggest that the difference between structure and morphology of α-Fe2O3 particles can lead to p-type and n-type characterization. In addition, we suggested that Na ions from NaBH4 were incorporated into α-Fe2O3 oxide. They are the cause of the generation of holes in α-Fe2O3 oxide that led to the p-type nature of α-Fe2O3 oxide. The measurement of the sensor response to hydrogen, carbon monoxide, toluene, propane and ethanol revealed that the sensor device synthesized using polyhedral α-Fe2O3 particles is sensitive to hydrocarbons and ethanol. Importantly, the sensor device using polyhedral α-Fe2O3 particles calcined at 900 °C was strongly sensitive to ethanol due to the porous structure of the α-Fe2O3 particles.
1. Introduction
So far, oxide semiconductor gas sensors have been practically used for detecting gases such as inflammable gases, dangerous hazardous gases and environmental gases because of their high sensor response and stability as well as simplification in electric circuits.1 The gas sensing mechanism for reducing gases of oxide semiconductor gas sensors has been considered that target gases diffused into the porous gas sensing films and are oxidized by oxygen species (O− and O2−) absorbed on the oxide surface, leading to the change in electric resistance. In the case of n-type semiconductors, the electric resistance of the sensing film decreases when the target gases react with the sensing film, because the majority carrier, e.g. electrons, move from absorbed oxygen species to the semiconductor. In contrast, the electric resistance of the sensor film using p-type semiconductor increases when the target gases react with the sensing film, because the concentration of the majority carrier, e.g. holes in p-type semiconductors, decreases when electrons move from absorbed oxygen species to the semiconductor.2Various typical materials have been reported for use in semiconductor gas sensors, such as SnO2 (n-type),3 TiO2 (n-type),4 ZnO (n-type),5 In2O3 (n-type),6 WO3 (n-type),7 α-Fe2O3 (n-type),8 γ-Fe2O3 (n-type),9 carbon nanotubes (p-type),10 Co3O4 (p-type),11 V2O5 (p-type),12 and CuO (p-type).13 Among them, α-Fe2O3 has the potential for practical use due to its high sensitivity, high stability and insensitivity to humidity.14,15 It has been reported that shape and size of semiconductor oxides strongly affects the gas sensing performance of the semiconductor gas sensor. When the grain size of semiconductor oxide decreases, the volume ratio of the space charge layer to the grain increases, leading to the increase in the sensor response.16,17 The controlling methods of the shape and morphology can lead to improve the facile diffusion of target gases into the gas sensing films.18,19 Therefore, morphology and size of α-Fe2O3 has been controlled to nanosphere,20 nanorod,21,22 nanotube,23 hollow sphere24–26 and porous structure27–29 in order to improve the gas sensing properties of α-Fe2O3.
In this study, we have synthesized polyhedral α-Fe2O3 particles for use in sensor materials. Herein, polyhedral α-Fe2O3 particles were successfully synthesized by a modified polyol method with the addition of an extra amount of NaBH4 in ethylene glycol (EG). The large polyhedral α-Fe2O3 particles were fabricated for use in a gas sensing film and its gas sensing properties were investigated in detection of hydrogen (H2), carbon monoxide (CO), toluene (C6H5CH3), propane (C3H8), and ethanol (C2H5OH) as target gases.
2. Experimental section
2.1. Chemical
Poly(vinylpyrrolidone) (PVP) (FW = 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), FeCl3·4H2O, sodium borohydride (NaBH4) and ethylene glycol (EG) were purchased from Sigma-Aldrich Co. and used for the synthesis of α-Fe2O3 particles. Ethanol, acetone and hexane were purchased from Wako Pure Chemical Industries, Ltd. and used for the dispersal of α-Fe2O3 particles synthesized. α-Terpineol purchased from Kishida chemical Co. Ltd. was used for the binder of gas sensing films. All chemicals were used as received without further purification.
000), FeCl3·4H2O, sodium borohydride (NaBH4) and ethylene glycol (EG) were purchased from Sigma-Aldrich Co. and used for the synthesis of α-Fe2O3 particles. Ethanol, acetone and hexane were purchased from Wako Pure Chemical Industries, Ltd. and used for the dispersal of α-Fe2O3 particles synthesized. α-Terpineol purchased from Kishida chemical Co. Ltd. was used for the binder of gas sensing films. All chemicals were used as received without further purification.
2.2. Materials synthesis
The details and steps of the procedures for the controlled synthesis of polyhedral α-Fe2O3 particles were previously presented in the controlled synthesis of Pt nanoparticles.30 Briefly, 3 mL of EG, 1.5 mL of 0.0625 M FeCl3, 3 mL of 0.375 M PVP, and 0.028 g NaBH4 were used for making Sample 1 in a typical process of the controlled synthesis of the polyhedral α-Fe2O3 oxide particles. The known polyol processes were previously presented.30,42 In general, FeCl3 was completely reduced with the extra amount of NaBH4 in EG at 200–230 °C for 30 min. As a result, the dark-brown solutions containing polyhedral α-Fe2O3 oxide particles with large sizes, shapes and morphologies were obtained as the final product. They have the particle size of 1–5 μm and with polyhedral shape and morphology. Similar to the synthetic process for Sample 1, we have used the same processes for Samples 2 and 3 for XRD and SEM measurements. Then, Sample 1 was also used for XRD and SEM measurement and analysis. Sample 2 was heated at 500 °C for 1 h for SEM measurement and analysis (Fig. 1(a)). Sample 3 was heated at 900 °C for 1 h for SEM analysis (Fig. 1(b)). The crystal phase of samples was identified by means of an X-ray diffractometer (XRD; RINT2100, Rigaku Co., Ltd. Japan) with Cu Kα radiation (1.54056 Å) as an X-ray source. The morphology of samples was observed by field emission scanning electron microscopy (FE-SEM, JSM-6340F, JEOL, Japan). The samples for the SEM observation were obtained by dropping the suspension of α-Fe2O3 particles on copper grids or copper brass. | ||
| Fig. 1 SEM images of the samples of the as-prepared Fe-based particles calcined at (a) 500 and (b) 900 °C. | ||
3. Results and discussion
3.1. Materials characterizations
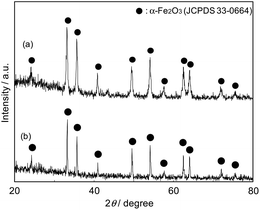
Prior to gas sensing measurements, the crystal phase and the morphology of samples were evaluated by means of XRD and FE-SEM, respectively. Fig. 3 shows the two XRD patterns of the samples after calcination at 500 and 900 °C. The pure crystal phases of both samples were assigned to α-Fe2O3 (JCPDS 33-0664) without any other impurity phases. Fig. 1(a) and (b) shows the FE-SEM image of α-Fe2O3 obtained at 500 °C and 900 °C, respectively. As shown in Fig. 1(a), α-Fe2O3 particles after the calcination at 500 °C were dense polyhedral particles with side lengths of 1–3 μm. Considering the crystalline size of α-Fe2O3 particles calculated by XRD, it seems that small size α-Fe2O3 nanocrystals was found in the dense polyhedral particles. On the other hand, in the case of α-Fe2O3 particles after the calcination at 900 °C, although the particle size is almost the same as α-Fe2O3 particles after calcination at 500 °C, many grain boundaries and small pores can be seen in the particles. It seems that small crystallites in one polyhedral particle were sintered, resulting in the formation of grain boundaries and small pores. However, the reason why the morphology of polyhedral particles is changed by the calcination temperature will be discussed in another paper.3.2. Gas sensing properties
In a semiconductor gas sensor, target gases react with oxygen species absorbed on the surface of the oxide semiconductor, resulting in the change in the electric resistance of the gas sensing film. Therefore, the dependence of the electric resistance on the oxygen partial pressure is an important factor for the operation of a semiconductor gas sensor. Fig. 4 shows the dependence of the electric resistance on the oxygen partial pressure (PO2) for the sensor device calcined at (a) 500 °C and (b) 900 °C. The electric resistance of both sensors decreased with an increase in the oxygen partial pressure, indicating the p-type nature of the α-Fe2O3 gas sensing film.31–33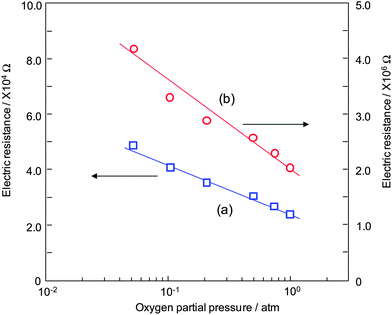 | ||
| Fig. 4 Dependence of the electric resistance on the oxygen partial pressure for the sensor device calcined at (a) 500 and (b) 900 °C. Operating temperature is 300 °C. | ||
In the case of p-type oxide semiconductors, oxygen attracts electrons in the valence band of the oxide, resulting in an increase in the hole concentrations on the large crystal surfaces of oxides. Therefore, the electric resistance of p-type α-Fe2O3 obtained in this study decreased with increasing the oxygen partial pressure.34 The main reason for the relatively large difference in the electric resistance between the two samples may be due to the possible effect of the crystalline size of α-Fe2O3. In the case of the p-type oxide semiconductor, the grain boundaries have lower electric resistance than the bulk oxide due to the formation of Schottky barriers. Hence, the larger number of grain boundaries tends to result in a lower electric resistance.
As shown in Fig. 3, the crystalline size of α-Fe2O3 calcined at 500 °C is smaller than α-Fe2O3 calcined at 900 °C. Therefore, it seems that the number of the grain boundaries in the sensor device calcined at 500 °C is larger than that of the device calcined at 900 °C, leading to a large decrease in the electric resistance of the sensor device calcined at 500 °C. Fig. 5 shows the response transients to 500 ppm H2 at various temperatures for the sensor device calcined at (a) 500 °C and (b) 900 °C. As shown in Fig. 5, the electric resistance increased when switching the flow gas from air to 500 ppm H2 attributed to the p-type nature of α-Fe2O3 obtained in this study. In the previous reports about semiconductor gas sensors with the use of α-Fe2O3, α-Fe2O3 oxide material works as an n-type oxide semiconductor, meaning that the electric resistance of α-Fe2O3 decreased under exposure to the reducing gases.35–38 However, our gas sensor exhibited the opposite tendency to the above reports. The origin of the p-type nature of α-Fe2O3 obtained in our research has not been elucidated yet. However, it seems that low concentration of Na ions from NaBH4 affected the major carrier of α-Fe2O3 obtained in our research. When Na ions from NaBH4 for the synthesis of samples are doped into α-Fe2O3 bulk, Na ions in the α-Fe2O3 lattice can generate holes as follows:39
| Na2O → 2NaFe′′ + OO + 2VO +4h˙ |
 | ||
| Fig. 5 The response transients to 500 ppm H2 for the sensor device calcined at (a) 500, and (b) 900 °C. | ||
The holes generated by the above formula can act as a source of p-type nature of α-Fe2O3 obtained in our research.
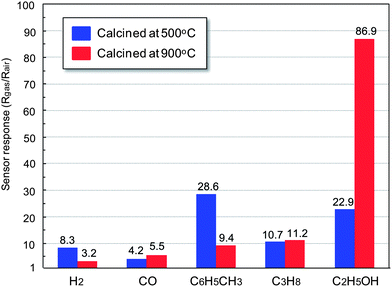
The sensor response to 500 ppm H2 was calculated from the response transients shown in Fig. 5, and summarized in Fig. 6. The sensor response was defined as the ratio of the electric resistance in air containing target gas (Rgas) to in air (Rair). As shown in Fig. 6, the sensor response increased with a decrease in the operating temperature. When the operating temperature is too high, H2 can be catalytically oxidized to water (H2O) without electron transfer between absorbed oxygen and α-Fe2O3. Therefore, lower operating temperature is preferable for high sensor response. However, as shown in Fig. 5, both the response and recovery time for H2 sensing decreased with a decrease in the operating temperature of our gas sensor. We suggested that the desorption of H2O produced by the reaction between H2 and the absorbed oxygen on α-Fe2O3 oxide surfaces became slow at lower temperature. In particular, the slow desorption of H2O prevented the adsorption of oxygen species on the surface of α-Fe2O3, resulting in the slow recovery of sensor response. Consequently, the optimal operating temperature of the sensor was defined as 300 °C. When the calcination temperature of the sensor devices was compared, the sensor response of the sensor device calcined at 500 °C exhibited a higher sensor response as compared with the sensor device calcined at 900 °C. It seems that the larger number of grain boundaries in the sensor device calcined at 500 °C gave the higher sensor response because the change in the electric resistance at the grain boundaries of oxides is one of the most important factors for a significant change in the electric resistance of the gas sensing film. Fig. 7 shows the dependence of the sensor response on the H2 concentration. As shown in Fig. 7, the sensor response to hydrogen gas (H2) was increased with an increase in the H2 concentration without saturation at high concentrations of H2. In particular, the sensor device calcined at 500 °C becomes more sensitive to a change in the H2 concentration due to the small crystalline size of α-Fe2O3. The sensor devices were exposed to various reducing gases, and compared with their sensor response. Typically, H2, CO, C6H5CH3, C3H8, and C2H5OH were chosen for the gas sensing measurements. Fig. 8 shows the response transients to various gases of the sensor device calcined at (a) 500 and (b) 900 °C. The value of the sensor response to various gases was summarized in Fig. 9. It was found that sensor devices using polyhedral α-Fe2O3 were sensitive to hydrocarbons and C2H5OH as compared with H2 and CO. However, as shown in Fig. 8, the recovery time for the hydrocarbons and C2H5OH was rather slow due to the slow desorption speed of oxidized intermediates of target gases. Such a slow recovery speed of α-Fe2O3 will be improved by doping foreign element or loading catalysts.40,41 As shown in Fig. 9, the sensor device calcined at 500 °C was most sensitive to C6H5CH3. However, in the case of the sensor device calcined at 900 °C, the highest sensor response was obtained by C2H5OH detection. This tendency can be explained by the difference in the porosity of the two sensor devices. As shown in Fig. 1(a), particles calcined at 500 °C are dense polyhedral particles with a side length of around 1–3 μm. In contrast, α-Fe2O3 particles calcined at 900 °C are also polyhedral particles but contains small pores in the polyhedral particles. It is likely that C2H5OH molecules can diffuse into the small pores in each polyhedral α-Fe2O3 particle. However, C6H5CH3 molecules were too large to diffuse into the pores facilely. Therefore, the sensor device using polyhedral α-Fe2O3 particles calcined at 900 °C was more sensitive to C2H5OH than C6H5CH3. As shown in the above, sensor devices obtained by calcination at 900 °C in this study are more sensitive and selective to ethanol as compared with semiconductor gas sensors using α-Fe2O3 reported in the other literature.21,25,36,38 There is a new phenomenon found in large particles with grain and boundary structure in Fig. 1(b), which is particle and structure deformation on the particle surface and inside the particle structure under heat treatment at 900 °C.43,44 We have suggested that phenomena of both plastic and elastic deformation on surface and inside α-Fe2O3 particles under nanoparticle heat treatments at 900 °C were identified for better gas sensing properties of the sensor device to ethanol. Therefore, the gas sensor using α-Fe2O3 particles with grain and boundary structures “new micro-nano structures” can be a good candidate for an ethanol-selective gas sensor without any other additives for high selectivity and sensitivity.45
 | ||
| Fig. 6 The dependence of the sensor response to 500 ppm H2 on the operating temperature for sensor devices calcined at (a) 500 and (b) 900 °C. | ||
 | ||
| Fig. 7 (a) The response transients to H2 at 300 °C and (b) dependence of the sensor response on the H2 concentration at 300 °C. | ||
 | ||
| Fig. 8 The response transients to various gases of sensor devices calcined at (a) 500 °C and (b) 900 °C. | ||
4. Conclusion
In this study, polyhedral α-Fe2O3 particles was successfully synthesized via a modified polyol method with the addition of an extra amount of NaBH4 in ethylene glycol, and its gas sensing characteristics were investigated. The measurement of the dependence of the electric resistance on the O2 concentration revealed that polyhedral α-Fe2O3 particles synthesized in this study exhibited a p-type nature; the electric resistance of the sensor device increased with an increase in the O2 concentration. This tendency is opposite to other reports of gas sensors using α-Fe2O3. The measurement of the sensor response to various reducing gases revealed that α-Fe2O3 particles prepared in this study are sensitive to hydrocarbons and alcohols as compared with H2 and CO gases. In particular, a sensor device calcined at 900 °C has sensitively responded to C2H5OH due to its porous structure. This result indicates that our sensor device obtained by calcination at 900 °C can be a good candidate for an ethanol-selective gas sensor.Acknowledgements
In this research, we are very grateful to the precious supports from Structural Ceramics Engineering Center, Shanghai Institute of Ceramics, Chinese Academy of Science, Dingxi Road 1295, Shanghai 200050, China. This study was also supported in part by a fund from the National Natural Science Foundation of China (NSFC, contract nos. 51071167 and 51102266).References
- K. Ihokura and J. Watson, The Stanic Oxide Gas Sensor – Principles and Applications, CRC Press, Boca Raton, FL, 1994 Search PubMed.
- P. T. Moseley, Sens. Actuators, B, 1992, 6, 149 CrossRef CAS.
- N. Yamazoe, Y. Kurokawa and T. Seiyama, Sens. Actuators, 1983, 4, 283 CrossRef CAS.
- G. S. Devi, T. Hyodo, Y. Shimizu and M. Egashira, Sens. Actuators, B, 2002, 87, 122 CrossRef CAS.
- L. Liao, H. B. Lu, J. C. Li, H. He, D. F. Wang, D. J. Fu and C. Liu, J. Phys. Chem. C, 2007, 111, 1900 CAS.
- S. N. Malchenko, Y. N. Lychkovsky and M. V. Baykov, Sens. Actuators, B, 1993, 13, 159 CrossRef CAS.
- J. L. Solisa, S. Saukko, L. Kish, C. G. Granqvist and V. Lantto, Thin Solid Films, 2001, 391, 255 CrossRef.
- V. V. Malyshev, A. V. Eryshkin, E. A. Koltypin, A. E. Varfolomeev and A. A. Vasiliev, Sens. Actuators, B, 1994, 19, 434 CrossRef CAS.
- C. V. Gopal Reddy, K. Kalyana Seela and S. V. Manorama, Int. J. Inorg. Mater., 2000, 2, 301 CrossRef.
- L. Valentini, C. Cantalini, I. Armentano, J. M. Kenny, L. Lozzi and S. Santucci, Diamond Relat. Mater., 2004, 13, 1301 CrossRef CAS PubMed.
- W. Y. Li, L. N. Xu and J. Chen, Adv. Funct. Mater., 2005, 15, 851 CrossRef CAS.
- M. Yu, X. Liu, Y. Wang, Y. Zheng, J. Zhang, M. Li, W. Lan and Q. Su, Appl. Surf. Sci., 2012, 258, 9554 CrossRef CAS PubMed.
- C. Yang, X. Su, J. Wang, X. Cao, S. Wang and L. Zhang, Sens. Actuators, B, 2013, 185, 159 CrossRef CAS PubMed.
- J. Peng and C. C. Chai, Sens. Actuators, B, 1993, 14, 591 CrossRef CAS.
- K. Song, Q. Wang, Q. Liu, H. Zhang and Y. Cheng, Sensors, 2011, 11, 485 CrossRef CAS PubMed.
- C. Xu, J. Tamaki, N. Miura and N. Yamazoe, Sens. Actuators, B, 1991, 3, 147 CrossRef CAS.
- A. Gurlo, M. Ivanovskaya, N. Bârsan, M. Schweizer-Berberich, U. Weimar, W. Göpel and A. Diéguez, Sens. Actuators, B, 1997, 44, 327 CrossRef.
- M. Tiemann, Chem.–Eur. J., 2007, 13, 8376 CrossRef CAS PubMed.
- M. M. Arafat, B. Dinan, S. A. Akbar and A. S. M. A. Haseeb, Sensors, 2012, 12, 7207 CrossRef CAS PubMed.
- Y. Cao, H. Luo and D. Jia, Sens. Actuators, B, 2013, 176, 618 CrossRef CAS PubMed.
- D. Patil, V. Patil and P. Patil, Sens. Actuators, B, 2011, 152, 299 CrossRef CAS PubMed.
- P. Sun, L. You, D. Wang, Y. Sun, J. Ma and G. Lu, Sens. Actuators, B, 2011, 156, 368 CrossRef CAS PubMed.
- H. Shan, C. Liu, L. Liu, S. Li, L. Wang, X. Zhang, X. Bo and X. Chi, Sens. Actuators, B, 2013, 184, 243 CrossRef CAS PubMed.
- J. Huang, M. Yang, C. Gu, M. Zhai, Y. Sun and J. Liu, Mater. Res. Bull., 2011, 46, 1211 CrossRef CAS PubMed.
- G. Fan, Y. Wang, M. Hu, Z. Luo, K. Zhang and G. Li, Sensors, 2012, 12, 4594 CrossRef CAS PubMed.
- G. Shi, K. Zhang, S. Zhou and Q. Zhang, Mater. Lett., 2013, 107, 228 CrossRef CAS PubMed.
- B. Sun, J. Horvat, H. S. Kim, W. S. Kim, J. Ahn and G. Wang, J. Phys. Chem. C, 2010, 114, 18753 CAS.
- Q. Hao, S. Liu, X. Yin, Z. Du, M. Zhang, L. Li, Y. Wang and Q. Li, CrystEngComm, 2011, 13, 806 RSC.
- D. Su, H. S. Kim, W. S. Kim and G. Wang, Microporous Mesoporous Mater., 2012, 149, 36 CrossRef CAS PubMed.
- N. V. Long, M. Ohtaki, M. Uchida, R. Jalem, H. Hirata, N. D. Chien and M. Nogami, J. Colloid Interface Sci., 2011, 359, 339 CrossRef PubMed.
- S. P. Mitoff, J. Chem. Phys., 1961, 35, 882 CrossRef CAS PubMed.
- Y. Li, W. Wlodarski, K. Galatsis, S. H. Moslih, J. Cole, S. Russo and N. Rockelmann, Sens. Actuators, B, 2002, 83, 160 CrossRef CAS.
- F. Hossein-Babaei, M. Keshmiri, M. Kakavand and T. Troczynski, Sens. Actuators, B, 2005, 110, 28 CrossRef CAS PubMed.
- P. T. Moseley, Sens. Actuators, B, 1991, 3, 167 CrossRef CAS.
- C. Cantalini, M. Faccio, G. Ferri and M. Pelino, Sens. Actuators, B, 1994, 19, 437 CrossRef CAS.
- N. D. Cuong, T. T. Hoa, D. Q. Khieu, T. D. Lam, N. D. Hoa and N. V. Hieu, J. Alloys Compd., 2012, 523, 120 CrossRef PubMed.
- Z. F. Dou, C. Y. Cao, Q. Wang, J. Qu, Y. Yu and W. G. Song, ACS Appl. Mater. Interfaces, 2012, 4, 5698 CAS.
- Y. Kang, L. Wang, Y. Wang, H. Zhang, Y. Wang, D. Hong, Y. Qv and S. Wang, Sens. Actuators, B, 2013, 177, 570 CrossRef CAS PubMed.
- C. V. Santilli, J. P. Bonnet, P. Dordor, M. Onillon and P. Hagenmuller, Ceram. Int., 1990, 16, 25 CrossRef CAS.
- Z. Jing, Mater. Lett., 2006, 60, 3315 CrossRef CAS PubMed.
- S. C. Lee, B. W. Hwang, S. J. Lee, H. Y. Choi, S. Y. Kim, S. Y. Jung, D. Ragupathy, D. D. Lee and J. C. Kim, Sens. Actuators, B, 2011, 160, 1328 CrossRef CAS PubMed.
- D. L. Huber, Small, 2005, 1(5), 482 CrossRef CAS PubMed.
- F. J. Humphreys and M. Hatherly, Recrystallization and related annealing phenomena, Elsevier Ltd, 2nd edn, 2004 Search PubMed.
- G. E. Totten, Steel Heat Treatment Handbook CRC Press, Taylor & Fracis Group, LLC, 2nd edn, 2007 Search PubMed.
- N. V. Long, Y. Yang, M. Yuasa, T. M. Cao, Y. Cao, T. Nann and M. Nogami, RSC Adv., 2014, 4, 6383–6390 RSC.
| This journal is © The Royal Society of Chemistry 2014 |