Synthesis, H2PO4− and Pd2+ ion sensing and gold nanoparticle encapsulation of ferrocenyldendrimers by a green chemistry approach†
Perumal Rajakumar*a,
Ramasamy Anandhana,
Devaraj Manojb and
Jayadevan Santhanalakshmib
aDepartment of Organic Chemistry, University of Madras, Guindy Campus, Chennai 600 025, India. E-mail: perumalrajakumar@gmail.com; Fax: +91 044 22300488; Tel: +91 044 22351269 ext. 213
bDepartment of Physical Chemistry, University of Madras, Guindy Campus, Chennai 600 025, India
First published on 16th October 2013
Abstract
A green chemistry approach for the synthesis of three novel ferrocenyldendrimers containing a triazolyl ferrocenyl group at the periphery under microwave conditions using an ionic liquid (IL) mediated click reaction is described. The application of such ferrocenyldendrimers as sensors for H2PO4− and Pd2+ is also discussed. The ferrocenyldendrimers which are used for the synthesis of Au nanoparticles were characterized using UV-Vis, spectroscopy and high resolution transmission electron microscopy (HRTEM). From the HRTEM images it is shown that dendrimer 1 stabilizes the gold nanoparticle, whereas dendrimers 2 and 3 encapsulate the gold nanoparticle.
Introduction
Dendrimers with electrochemically active groups at the periphery can be potentially used in materials chemistry as biochips,1 nanoscience devices,2 electrode modifiers, biosensors3 and catalysts.4 In particular, ferrocenyl-terminated dendrimers provide a single wave in cyclic voltammetry, which facilitates their application as sensors, multielectron redox catalysts and electrochemical references.5 Anion recognition is an interesting topic in supramolecular chemistry and has become a very active research area due to the existence of anions in biological and environmental systems.6 In this context, 1,2,3-triazolyl ferrocenyl groups at the periphery of a dendrimer7 is an ideal choice to recognize and bind oxo anions and metal cations by redox monitors.8Dendrimer-entrapped metal nanoparticles are usually prepared by reducing metal ions that have been complexed with dendrimer templates. The application of metal nanoparticles in various fields of research is interesting because of their inherent size-dependent optical, electrical, catalytic, and magnetic properties.9 Moreover dendrimer encapsulated Au nanoparticles are important due to their biocompatibility10 and are also used in nanomedicine.11 Hence development in this area awaits better synthetic approaches for preparing nanomaterials. For example, Crooks et al.12 and Boyen et al.12 prepared stabilized Au nanoparticles with a size range 1–8 nm using dendrimers and block copolymers and showed that they self assembled onto a variety of substrates. More recently, Astruc and co-workers13 reported an interesting family of triazolyldendrimers by using a click reaction to form dendrimer-encapsulated gold nanoparticles and dendrimer-stabilized gold nanoparticles which are useful for catalytic and biomedical applications.
The Cu(I) catalysed Huisgen 1,3-dipolar cycloaddition of azides to alkynes, resulting in 1,4-disubstituted triazoles under thermal conditions, is one of the most powerful click reactions, which happens to be an efficient and rapid approach for the synthesis of novel dendrimers with desirable functions.14 Under microwave assisted click chemistry conditions 1,4-disubstituted-1,2,3-triazole is exclusively formed within a short time duration.15 Though efforts have been made in click chemistry in various directions, the issue regarding the green aspect remains unexplored. The lack of solubility of the reactants creates a serious obstacle for the use of a water medium in click chemistry and hence solid supports like SiO2, Al2O3, and Na2SO4 have been used.16 Further to microwave assistence,17 the use of ionic liquids (ILs)18 is another way to meet the principles of green chemistry in a click reaction. ILs can be excellent solvents for metal-catalyzed reactions.19 However, reports on the use of ILs for the synthesis of 1,4-disubstituted-1,2,3-triazole through click chemistry under microwave conditions is still a rare observation. We report herein the green chemistry approach for the synthesis of ferrocenyldendrimers 1, 2 and 3 (Fig. 1) with triazolylferrocene as a surface group under microwave assisted solid support reaction conditions and the application of the dendrimers as sensors for H2PO4− and Pd2+ ions. Furthermore, in the current investigation the tert-amine has a two fold application viz. it forms the ILs in situ and at the same it stabilizes the Cu(I) which is generated in situ by a complexation phenomenon and hence the present approach could be used as a better approach than the existing ones.
Results and discussion
The synthesis of dendrimers through a convergent approach is based on the formation of a triazole linkage between an azide and an acetylene unit. ILs are generated in situ by adding 1.0 equiv. of 3,5-bis(propargyloxy)benzyl chloride 5 to a tertiary amine base such as Et3N, N-methylpyrrolidine or N-methylmorpholine and then reacting with 2.1 equiv. of azidomethylferrocene 4 in the presence of 5 mol% CuSO4·5H2O and 10 mol% sodium ascorbate under microwave irradiation (80 W) for 1 min. Sodium azide is then added followed by microwave irradiation for 1 min in the presence of Na2SO4 as a solid support to give the first generation ferrocenyl dendritic azide 6 with a 93% yield (Scheme 1).The amine not only forms an ionic liquid by reacting with 5, but also stabilizes the Cu(I) intermediate formed during the course of the reaction. N-Methylmorpholine is a stronger base than N-methylpyrrolidine and triethylamine and hence it form ILs very effectively and at the same time it effectively stabilizes the Cu(I) intermediate and hence the best yields are obtained while using NMM as the base. The synthesis of second generation ferrocenyl dendritic azide 7 has been achieved from the 3,5-bis(propargyloxy)benzyl chloride 5 by repeating the same sequence of reactions. The reaction of the dendritic azide G1–N3 6 with 5 in the presence of the N-methylmorpholine and in the presence of copper sulphate under microwave irradiation (80 W) for 1 min was followed by the addition of sodium azide, and under microwave irradiation in the presence of Na2SO4 as a solid support gave the dendritic azide 7 in a 92% yield (Scheme 1).
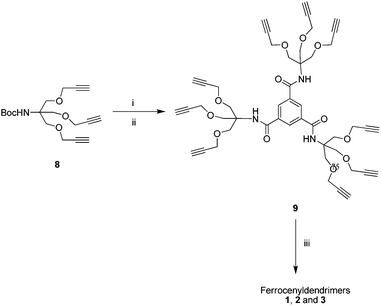
The tert-butyloxy carbonyl (t-BOC) protecting group was removed from the trispropargyl derivative 8 using trifluoroaceticacid (TFA) at 0 °C to room temperature to give a tris[(propargyloxy)methyl]aminomethanetrifluro acetate salt which was coupled without any purification with 0.3 equiv. of benzene-1,3,5-tri carboxylic acid chloride in the presence of Et3N in dry dichloromethane (DCM) at room temperature under N2 to give the TRIS acetylene terminated amide core 9 in a 58% yield. The reaction of 1.0 equiv. of 9 with 9.1 equiv. of azidomethylferrocene 4/dendritic azide 6/dendritic azide 7 under MW irradiation using Na2SO4 as a solid support for 1 min gave the ferrocenyldendrimers 1, 2 and 3 in 80%, 86% and 92% yields respectively (Scheme 2).
Cyclic voltammetry and electrochemical sensing
Cyclic voltammograms (CVs) of the three dendrimers 1, 2 and 3 were recorded with a glassy carbon (GC) electrode in CH2Cl2 using [nBu4N][PF6] as the supporting electrolyte.The ferrocenyldendrimers 1, 2 and 3 show a single and well defined redox wave. The cathodic peak potential (Epc) appeared at 460 mV, 505 mV and 558 mV, whereas the anodic peak potential was observed at 613 mV, 691 mV and 796 mV for dendrimers 1, 2 and 3 respectively. The cyclic voltammograms of dendrimer 1 at various scan rates and the variation of current intensity against scan rate are shown in ESI† as Fig. S1 and S2. Cyclic voltammogram studies were carried out in solution to examine whether the triazolyl ferrocenyl group present in dendrimers 1, 2 and 3 can function as a sensor for the oxo-anion like H2PO4− and the Pd(II) cation in Pd(PPh3)Cl2. On adding 1 equivalent of [n-Bu4N][H2PO4] per equivalent of triazole to a solution of dendrimer 1 in CH2Cl2 the CV showed a shift of the cathodic peak potential to the less positive side whereas on adding 1 equivalent of Pd(PPh3)Cl2 per equivalent of triazole to a solution of the dendrimer 1 in CH2Cl2 the shift was observed towards the more positive side (Fig. 2). Similarly, the synthesized dendrimers 2 and 3 reported here were also used for sensing the H2PO4− and Pd2+ ions (Table 1).
| Alkyne | Azide | tert-Amine base | Yield of dendritic azide 6 | Alkyne | Azide | tert-Amine base | Yield of dendritic azide 7 |
|---|---|---|---|---|---|---|---|
| 5 | 4 | Et3N | 83% | 5 | 6 | Et3N | 80% |
| 5 | 4 | N-Methyl pyrrolidine | 89% | 5 | 6 | N-Methyl pyrrolidine | 86% |
| 5 | 4 | N-Methyl morpholine | 93% | 5 | 6 | N-Methyl morpholine | 92% |
CV titration of dendrimers 1, 2 and 3 with a H2PO4− ion
The recognition of an oxo-anion (H2PO4−) by the ferrocenyldendrimers 1, 2 and 3 was investigated by adding (nBu4N)(H2PO4) to the electrochemical cell containing the ferrocenyldendrimers 1, 2 or 3 in CH2Cl2. Initially, before the addition of H2PO4− the dendrimer 1 showed a single and well defined redox wave with the cathodic and anodic peak potentials as mentioned earlier. While adding H2PO4− to the electrochemical cell, ferrocenyldendrimers 1, 2 and 3 show a splitting in the anodic CV wave which reflects the ion-pairing interaction between H2PO4− and the outer [Fe]+. A similar observation has been also reported by Geiger and co-workers20 in which the ion-pairing interactions have an influence on the separation between the two CV waves of binuclear or multinuclear redox systems. Further additions of H2PO4− show a significant decrease in the intensity of the ferrocenyl wave followed by the appearance of the new wave at the less positive cathodic peak potential. The appearance of the new wave is characteristic of relatively slow heterogeneous electron transfer which reveals the existence of strong interactions between the negatively charged oxygen atom of the phosphate groups (H2PO4−) and the ferrocenyl group (Fe center). Cyclic voltammograms were recorded after the periodic addition of [n-Bu4N][H2PO4] to dendrimer 1. At the end of the titration, the intensity of the new wave was further increased with a decrease in the intensity of the old wave (i.e., almost diminished) with the shift in the cathodic peak potential at 327 ± 5 mV, 413 ± 5 mV, 314 ± 5 mV respectively. The apparent shift signifies that the assembly of the dendrimer-oxoanion (with a FeIII state) is easier to oxidize than that of the dendrimer without the oxoanion assembly (with a FeII state), probably because the anion increases the electron density at the redox centre. The titration curve of dendrimers 1, 2 and 3 exhibited a similar behaviour as shown in the ESI† (Fig. S3a–c).Table 2 describes the recognition data which includes the differences in the potential observed between the initial and final CV wave (ΔE1/2 = before and after the titration of H2PO4−), the potential difference between the final anodic and cathodic waves (Epa − Epc), and the ratio of the apparent association constant (K(+)/K(0)). The potential difference between the anodic and cathodic waves (Epa − Epc = decreasing slightly with an increase in the dendrimers size) indicates that there is a reorganization of the dendrimer–guest assemblies upon slow heterogeneous electron transfer. The titration curve with respect to dendrimers 1 and 2 on adding various concentrations of H2PO4− is also shown in the ESI† (Fig. S4a and b).
| Dendrimers | E1/2a (Epa − Epc) | ipc/ipa | nb | Recognition of (nBu4N)(H2PO4) | Recognition of Pd(PPh3)2Cl2 | ||||
|---|---|---|---|---|---|---|---|---|---|
| E1/2a (Epa − Epc) | ΔE1/2c | K(+)/K(0)d | E1/2a (Epa − Epc) | ΔE1/2c | K(+)/K(0)d | ||||
a E1/2 = (Epa + Epc)/2 vs. [FeCp*2]. The peak potentials might be perturbed by some adsorption,21b thus K(+)/K(0) values must be considered with caution. Adsorption is weak, however, during the titration process and becomes important only at, and after, the equivalent point. The potential of the new wave does not vary significantly during the titration or at the equivalent point. Thus, errors in the E1/2 values are limited. Electrolyte: 0.1 M (nBu4N)(PF6); working electrode: GCE; counter electrode: Pt; quasi-reference electrode: Ag; internal reference: [FeCp*2]; scan rate: 0.100 V s−1; 20 °C.b Number of electrons involved calculated from the Anson–Bard equation21 using anodic intensities.c Difference between values of E1/2 before and after titration.d Ratios of apparent association constants; error < 10%; ΔE1/2 = 0.058![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log(K(+)/K(0))22 at 20 °C. log(K(+)/K(0))22 at 20 °C. |
|||||||||
| 1 | 0.499 (0.153) | 1.81 | 9 | 0.385 (0.198) | 0.114 | 92 | 0.601 (0.135) | −0.116 | 100 |
| 2 | 0.556 (0.186) | 1.85 | 27 | 0.454 (0.180) | 0.102 | 57 | 0.645 (0.184) | −0.089 | 34 |
| 3 | 0.646 (0.238) | 1.32 | 63 | 0.381 (0.279) | 0.265 | 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 065 065 |
0.643 (0.273) | −0.003 | 1 |
CV titration of dendrimers 1, 2 and 3 with bis(triphenylphosphine)Pd(II)dichloride
Cyclic voltammograms were recorded after the periodic addition of Pd(PPh3)2Cl2 to dendrimer 1. After adding Pd(PPh3)2Cl2 at various concentrations to dendrimer 1 a new redox wave appeared at the more positive side than the initial wave which signifies that the oxidation of the dendrimer becomes more difficult than with the dendrimer alone. This shows that each triazole group of the dendrimer effectively binds on the Pd(PPh3)2Cl2 unit and hence oxidation becomes difficult as seen from Table 2. The titration curve of the dendrimers 1, 2 and 3 with Pd(PPh3)2Cl2 are shown in the ESI† (Fig S5a–c).Synthesis and characterization of AuNPs stabilized by the 1,2,3-triazolyl dendrimers 1, 2 and 3
Dendrimers 1, 2 and 3 are also used to stabilize Au nanoparticles prepared in situ by the reduction of Au3+ using NaBH4. In the reaction mixture the Au3+ ions are protected well with the dendrimer in the presence of CHCl3 and MeOH, and after the addition of NaBH4 the Au3+ ions are reduced to Au0 nanoparticles with a significant change in color from yellow to wine red (pink), which was monitored by UV-Vis spectroscopy. Fig. 3 shows the UV-Vis spectra of the Au nanoparticles obtained using dendrimers 1, 2 and 3 respectively. Dendrimer 1 stabilized Au nanoparticles and showed a broad absorption band with a surface plasmon resonance (SPR) peak at 544 nm revealing the formation of Au nanoparticles. Dendrimer 3 shows a sharp absorption peak and the SPR value gets blue shifted confirming the effective reduction of nanoparticles with a decrease in particle size of less than 3 nm. Generally a distinctive shift in the SPR peak from 544 nm to 525 nm accompanied by a decrease in the absorbance as the generation of dendrimers increases from 1 to 3, emphasises the effective encapsulation of the Au nanoparticles by the dendrimer. | ||
| Fig. 3 UV-Vis spectra of dendrimer 1 stabilized, and dendrimers 2 and 3 encapsulated Au nanoparticles. | ||
Fig. 4 shows the typical HRTEM images of Au nanoparticles stabilized with dendrimers 1, 2 and 3. Dendrimer 1 has an open structure, and therefore a Au nanoparticle surrounded by dendrimer 1 will emerge and aggregate into Au nanoparticles of a larger size. Au nanoparticles cannot be encapsulated (inter-dendrimer stabilized) by a lower generation of dendrimer 1 that contains only nine triazole units. It is quite evident from Fig. 4A that dendrimer 1 stabilized Au nanoparticles show a wide particle size distribution with an average diameter of about 9.0 ± 0.5 nm. On the other hand the higher generation of dendrimers 2 and 3 show a narrow particle size distribution which are intra-dendrimer encapsulated (but not stabilized) Au nanoparticles.23 The HRTEM image shows that Au nanoparticles obtained by encapsulation with dendrimers 2 and 3 are smaller when compared to dendrimer 1. The average particle size was calculated to be 2.0 ± 0.3 nm and 1.5 ± 0.3 nm respectively. The decrease in particle size can be attributed to the increase in the number of triazole units in dendrimers 2 and 3 which contain twenty-seven and sixty-three triazole units, respectively, that provides the effective reduction in particle size. The mechanism for the encapsulation of Au nanoparticles can be explained as follows: after the complexation of the ferrocenyltriazole unit with Au3+ in the presence of MeOH, the Au acetate molecule gets dechelated and binds with the triazole ligand, and therefore the Au3+ ions get encapsulated into the dendrimer 3 interior which hinders the Au atoms emerging from the dendrimer 3. During the addition of NaBH4, the Au3+ ions are reduced into Au0 nanoparticles with smaller size nanoparticles compared to dendrimers 1 and 2.
 | ||
| Fig. 4 HRTEM image of (A) dendrimer 1 stabilized, (B) dendrimer 2 and (C) dendrimer 3 encapsulated Au nanoparticles. | ||
Binding energies of the Au 4f peaks can be determined by referencing with the adventitious C 1s peak at 284.6 eV. The Au 4f line peaks are fitted using a Gaussian fitting procedure from which the Au 4f5/2 and Au 4f7/2 peaks were observed. The deconvolution of the two main core-level Au 4f peak regions was resolved and the peak positions are obtained at 84.19 eV and 87.9 eV respectively (ESI Fig. S6†). Moreover, the main peak–peak difference of the core-level peak positions which is ∼3.7 eV reveals the formation of pure Au only and agrees well with earlier reported methods.24
Conclusion
In conclusion, a microwave assisted, solid supported IL catalyzed click reaction has been successfully used for the synthesis of ferrocenyldendrimers 1, 2 and 3, and all the dendrimers exhibited an excellent electrochemical sensing property for H2PO4− and Pd2+. Furthermore, Au nanoparticles are successfully synthesized and stabilized by dendrimer 1 and encapsulated by dendrimers 2 and 3 respectively. With an increase in the generation of the dendrimers the Au nanoparticle size was found to decrease rapidly with the increase in the stability of the nanoparticles. Future work will evaluate and explore the development of dendrimer encapsulated Au nanoparticles for electrochemical biosensors.Acknowledgements
The authors thank CSIR, New Delhi, India, for financial assistance and DST-FIST for providing NMR facilities to the department. RA thanks CSIR, New Delhi, for a fellowship, and SAIF for MALDI-TOF mass spectra.Notes and references
- (a) J. Satija, V. V. R. Sai and S. Mukherji, J. Mater. Chem., 2011, 21, 14367 RSC; (b) M. Y. Hong, Y. J. Kim, J. W. Lee, K. Kim, J. H. Lee, J. S. Yoo, S. H. Bae, B. S. Choi and H. S. Kim, J. Colloid Interface Sci., 2004, 274, 41 CrossRef CAS PubMed; (c) M. Y. Hong, H. C. Yoon and H. S. Kim, Langmuir, 2003, 19, 416 CrossRef CAS.
- (a) C. Amatore, Y. Bouret, E. Maisonhaute, J. I. Goldsmith and H. D. Abruna, Chem.–Eur. J., 2001, 7, 2206 CrossRef CAS; (b) M. C. Daniel, J. Ruiz, S. Nlate, J. C. Blais and D. Astruc, J. Am. Chem. Soc., 2003, 125, 2617 CrossRef CAS PubMed.
- (a) J. Losada, M. P. G. Armada, M. Zamora, B. Alonso, I. Cuadrado and C. M. Casado, Bioelectrochemistry, 2006, 69, 65 CrossRef PubMed; (b) J. Losada, M. Zamora, P. G. Armada, I. Cuadrado, B. Alonso and C. M. Casado, Anal. Bioanal. Chem., 2006, 385, 1209 CrossRef CAS PubMed; (c) F. J. Martinez, B. Gonzalez, B. Alonso, J. Losada, M. P. Garcia-Armada and C. M. Casado, J. Inorg. Organomet. Polym. Mater., 2008, 18, 51 CrossRef CAS.
- (a) D. Astruc and F. Chardac, Chem. Rev., 2001, 101, 2991 CrossRef CAS PubMed; (b) R. van Heerbeeck, P. C. J. Kamer, P. W. N. M. van Leeuwen and J. N. H. Reek, Chem. Rev., 2002, 102, 3717 CrossRef PubMed; (c) R. W. Scott, A. K. Datye and R. M. Crooks, J. Am. Chem. Soc., 2003, 125, 3708 CrossRef CAS PubMed; (d) Y. Niu and R. M. Crooks, Chem. Mater., 2003, 15, 3463 CrossRef CAS; (e) O. M. Wilson, S.-K. Oh, Y. Niu and R. M. Crooks, Langmuir, 2005, 21, 10209 CrossRef PubMed; (f) M. R. Knecht, J. C. Garcia-Martinez and R. M. Crooks, J. Am. Chem. Soc., 2006, 128, 4510 CrossRef PubMed; (g) Y. Jiang and Q. Gao, J. Am. Chem. Soc., 2006, 128, 716 CrossRef CAS PubMed.
- (a) D. Astruc, New J. Chem., 2009, 33, 1191 RSC; (b) C. M. Casado, I. Cuadrado, M. Moron, B. Alonso, B. Garcia, B. Gonzales and J. Losada, Coord. Chem. Rev., 1999, 53, 185 Search PubMed.
- (a) P. D. Beer, Adv. Inorg. Chem., 1993, 39, 79 Search PubMed; (b) P. D. Beer, Chem. Commun., 1996, 689 RSC (Feature Article); (c) P. D. Beer, Acc. Chem. Res., 1998, 31, 71 CrossRef CAS; (d) P. D. Beer, P. A. Gale and Z. Chen, Adv. Phys. Org. Chem., 1998, 31, 1 CAS; (e) P. D. Beer, Angew. Chem., Int. Ed., 2001, 40, 486 CrossRef CAS; (f) J. H. R. Tucker and S. R. Collison, Chem. Soc. Rev., 2002, 31, 147 RSC; (g) P. D. Beer and E. J. Hayes, Coord. Chem. Rev., 2003, 240, 167 CrossRef CAS; (h) A. Labande and D. Astruc, Chem. Commun., 2000, 1007 RSC; (i) A. Labande, J. Ruiz and D. Astruc, J. Am. Chem. Soc., 2002, 124, 1782 CrossRef CAS PubMed.
- (a) C. Villena, J. Losada, P. Garcıa-Armada, C. M. Casado and B. Alonso, Organometallics, 2012, 31, 3284 CrossRef CAS; (b) B. Gonzalez, B. Alonso, J. Losada, M. P. Garcıa-Armada and C. M. Casado, Organometallics, 2006, 25, 3558 CrossRef CAS; (c) R. Villoslada, B. Alonso, C. M. Casado, M. P. Garcıa-Armada and J. Losada, Organometallics, 2009, 28, 727 CrossRef CAS.
- (a) C. Ornelas, J. R. Aranzaes, E. Cloutet, S. Alves and D. Astruc, Angew. Chem., Int. Ed., 2007, 46, 872 CrossRef CAS PubMed; (b) R. Djeda, A. Rapakousiou, L. Liang, N. Guidolin, J. Ruiz and D. Astruc, Angew. Chem., Int. Ed., 2010, 49, 8152 CrossRef CAS PubMed; (c) M. Yamada and H. Nishihara, Langmuir, 2003, 19, 8050 CrossRef CAS; (d) C. Ornelas, J. R. Aranzaes, L. Salmon and D. Astruc, Chem.–Eur. J., 2008, 14, 50 CrossRef CAS PubMed.
- (a) C. N. R. Rao, G. U. Kulkarni, P. J. Thomas and P. P. Edwards, Chem.–Eur. J., 2002, 8, 29 CrossRef; (b) M. Valden, X. Lai and D. W. Goodman, Science, 1998, 281, 1647 CrossRef CAS.
- (a) M. Liu, K. Kono and J. M. J. Frechet, J. Controlled Release, 2000, 65, 121 CrossRef CAS; (b) R. Duncan, Nat. Rev. Drug Discovery, 2003, 2, 347 CrossRef CAS PubMed.
- (a) S. L. Lal, S. E. Clare and N. J. Halas, Acc. Chem. Res., 2008, 41, 1842 CrossRef CAS PubMed; (b) E. Boisselier and D. Astruc, Chem. Soc. Rev., 2009, 38, 1759 RSC.
- (a) H. G. Boyen, G. Kastle, F. Weigl, B. Koslowski, C. Dietrich, P. Ziemann, J. P. Spatz, S. Riethmuller, C. Hartmann, M. Moller, G. Schmid, M. G. Garnier and P. Oelhafen, Science, 2002, 297, 1533 CrossRef CAS PubMed; (b) Y.-G. Kim, S.-K. Oh and R. M. Crooks, Chem. Mater., 2004, 16, 167 CrossRef CAS; (c) D. F. Yancey, E. V. Carino and R. M. Crooks, J. Am. Chem. Soc., 2010, 132, 2942 CrossRef PubMed.
- (a) E. Boisselier, A. K. Diallo, L. Salmon, J. Ruiz and D. Astruc, Chem. Commun., 2008, 4819 RSC; (b) E. Boisselier, A. K. Diallo, L. Salmon, C. Ornelas, J. Ruiz and D. Astruc, J. Am. Chem. Soc., 2010, 132, 2729 CrossRef CAS PubMed; (c) C. Ornelas, L. Salmon, J. Ruiz and D. Astruc, Chem.–Eur. J., 2008, 14, 50 CrossRef CAS PubMed.
- (a) P. Rajakumar, R. Anandhan and V. Kalpana, Synlett, 2009, 1417 CrossRef CAS PubMed; (b) P. Rajakumar and R. Anandhan, Eur. J. Med. Chem., 2011, 46, 4687 CrossRef CAS PubMed; (c) P. Rajakumar and R. Anandhan, Aust. J. Chem., 2012, 65, 1457 CrossRef CAS; (d) M. Meldal and C. W. Tornøe, Chem. Rev., 2008, 108, 2952 CrossRef CAS PubMed.
- (a) C. O. Kappe and E. V. Eycken, Chem. Soc. Rev., 2010, 39, 1280 RSC; (b) M. Munteanu, S. Choi and H. Ritter, Macromolecules, 2008, 41, 9619 CrossRef CAS.
- (a) P. Rajakumar and V. Murali, Chem. Commun., 2001, 2710 RSC; (b) A. Megia-Fernandez, M. Ortega-MuÇoz, J. Lopez-Jaramillo, F. Hernandez-Mateo and F. Santoyo-Gonzalez, Adv. Synth. Catal., 2010, 352, 3306 CrossRef CAS.
- (a) F. V. Rantwijk and R. A. Sheldon, Chem. Rev., 2007, 107, 2757 CrossRef PubMed; (b) H. Weingartner, Angew. Chem., Int. Ed., 2008, 47, 6543 CrossRef PubMed.
- (a) G. Machado, J. Scholten, T. de Vargas, S. R. Teixera, L. H. Ronchi and J. Dupont, Int. J. Nanotechnol., 2007, 4, 541 CrossRef CAS; (b) K. Biswas and C. N. R. Rao, Chem.–Eur. J., 2007, 13, 6123 CrossRef CAS PubMed.
- (a) B. Ni, Q. Zhang and A. D. Headley, J. Org. Chem., 2006, 71, 9857 CrossRef CAS PubMed; (b) P. S. Schulz, N. Muller, A. Bosmann and P. Wasserscheid, Angew. Chem., Int. Ed., 2007, 46, 1293 CrossRef CAS PubMed.
- (a) N. Camine, U.-T. Mueller-Westerhoff and W. E. Geiger, J. Organomet. Chem., 2001, 637–639, 823 Search PubMed; (b) F. Barriere, N. Camine and W. E. Geiger, J. Am. Chem. Soc., 2002, 124, 7262 CrossRef CAS PubMed; (c) F. Barriere and W. E. Geiger, J. Am. Chem. Soc., 2006, 128, 3980 CrossRef CAS PubMed; (d) F. Barriere and W. E. Geiger, Acc. Chem. Res., 2010, 43, 1030 CrossRef PubMed.
- (a) J. B. Flanagan, S. Margel, A. J. Bard and F. C. Anson, J. Am. Chem. Soc., 1978, 100, 4248 CrossRef CAS; (b) A. J. Bard and R. L. Faulkner, Electrochemical Methods, Wiley, New York, 2nd edn, 2001 Search PubMed.
- S. R. Miller, D. A. Gustowski, Z.-H. Chen, G. W. Gokel, L. Echegoyen and A. E. Kaifer, Anal. Chem., 1988, 60, 2021 CrossRef CAS.
- C. Ornelas, L. Salmon, J. R. Aranzaesa and D. Astruc, Chem. Commun., 2007, 4946 RSC.
- L. Fabris, S. Antonello, L. Armelao, R. L. Donkers, F. Polo, C. Toniolo and F. Maran, J. Am. Chem. Soc., 2006, 128, 326 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra43238f |
| This journal is © The Royal Society of Chemistry 2014 |