Signaling mechanisms of LOV domains: new insights from molecular dynamics studies†
Abstract
Phototropins are one of several classes of photoreceptors used by plants and algae to respond to light. These
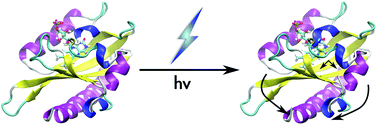
- This article is part of the themed collection: Blue-light photoreceptors

 Please wait while we load your content...
Please wait while we load your content...