Open Access Article
Open Access ArticleModular chiral gold(I) phosphite complexes†
Nicolas Delponta,
Imma Escofeta,
Patricia Pérez-Galána,
Dirk Spiegla,
Mihai Raducana,
Christophe Boura,
Riccardo Sinisia and
Antonio M. Echavarren*ab
aInstitute of Chemical Research of Catalonia (ICIQ), Av. Països Catalans 16, 43007 Tarragona, Spain. E-mail: aechavarren@iciq.es
bDepartament de Química Analítica i Química Orgànica, Universitat Rovira i Virgili, C/Marcel·li Domingo s/n, 43007 Tarragona, Spain
First published on 2nd July 2013
Abstract
Chiral gold(I) phosphite complexes are readily prepared modularly from 3,3′-bis(triphenylsilyl)-1,1′-bi-2-naphthol. These chiral gold(I) phosphite complexes are very reactive precatalysts for the [4+2] cycloaddition of aryl-substituted 1,6-enynes with enantiomeric ratios ranging from 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 up to 94
14 up to 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.
6.
Introduction
Homogeneous gold catalysis provides efficient solutions for the construction of complex carbon skeletons under mild conditions.1–4 Much of the progress in the enantioselective C–C multiple bond activation catalysed by gold has been achieved in the last few years in intramolecular reactions.5–15 However, wide-scope enantioselective gold-catalysed transformations are still relatively scarce.In 2005 we reported the first gold(I)-catalysed enantioselective alkoxycyclization of 1,6-enynes with a cationic catalyst generated in situ from [(R)-Tol-BINAP(AuCl)2] and AgSbF6.16 Related enantioselective cyclizations of 1,6-enynes have been carried out more recently with chiral NHC–gold(I)17 and phosphine–gold18 complexes, or using platinum catalysts.19
We have developed a general gold(I)-catalysed cycloisomerization of substrates 1 by formal [4+2] cycloaddition of arylalkynes with alkenes to form stereospecific cycloadducts 2,20 with the core structure of pycnanthuquinones (Scheme 1).21–23
![Gold(i)-catalyzed [4+2] cycloaddition of 1,6-enynes 1 and the structures of pycnanthuquinones A–C.](/image/article/2013/CY/c3cy00250k/c3cy00250k-s1.gif) | ||
| Scheme 1 Gold(I)-catalyzed [4+2] cycloaddition of 1,6-enynes 1 and the structures of pycnanthuquinones A–C. | ||
As part of a program on the development of general strategies for the synthesis of these terpenoid quinones, we examined an alternative pathway based on the gold-catalysed cyclization of benzyl-substituted 1,5-enynes.24 In parallel, we also studied the enantioselective cycloaddition of aryl-substituted 1,6-enynes 1 using a variety of gold(I) catalysts with chiral phosphine ligands. Whereas we obtained modest enantioselectivities in most cases,25 the group of Genêt and Michelet reported good results in the cyclization of two substrates 1a–b in the presence of a gold(I) catalyst generated in situ from DTBM-MeOBIPHEP and AgOTf,26 although in the case of 1b the yield was significantly lower than that obtained with achiral catalysts20 (Scheme 2).
![Enantioselective gold(i)-catalysed [4+2] cycloaddition of 1,6-enynes 1a–b.](/image/article/2013/CY/c3cy00250k/c3cy00250k-s2.gif) | ||
| Scheme 2 Enantioselective gold(I)-catalysed [4+2] cycloaddition of 1,6-enynes 1a–b. | ||
In an effort at developing general and practical methods for the screening of a large variety of chiral ligands in gold-catalysed reactions, we recently reported a procedure that allows performing enantioselective processes from catalysts prepared in situ from a cationic complex [Au(tmbn)2](SbF6) (tmbn = 2,4,6-trimethoxybenzonitrile) and the corresponding chiral ligand.27 As an alternative, we prepared a series of complexes bearing chiral phosphite ligands based on the BINOL motive using a relatively simple, modular approach from a commercially available 1,1′-bi-2-naphthol. We focused on phosphite ligands over phosphines because of their lower sensitivity to air and other oxidizing agents,28 and because phosphite gold(I) complexes are the most reactive catalysts for the activation of alkynes.29,30 Herein we report our efforts towards the development of chiral BINOL-derived phosphite gold(I) complexes. Chiral BINOL-derived phosphites have been used as building blocks for synthesis of chiral palladacycles, bis(phosphite) and mixed phosphite–phosphinite PCP-palladium pincer complexes.31,32 Monodentate phosphite gold(I) complexes with C3-symmetry33 and chiral gold phosphoramidite-based catalysts have also been used in a number of gold-catalysed reactions.10–12,34
Results and discussion
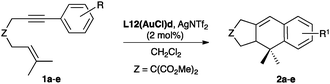
We initially examined the gold(I)-catalysed cyclization of enyne 1a to form adduct 2a using a wide range of complexes as precatalysts (Fig. 1). The structures of complexes L8(AuCl) (Fig. 2), L9(AuCl), L10(AuCl) (Fig. 3), L11(AuCl) (Fig. 4), and L12(AuCl)a (Fig. 5) and L12(AuCl)e were determined using X-ray diffraction.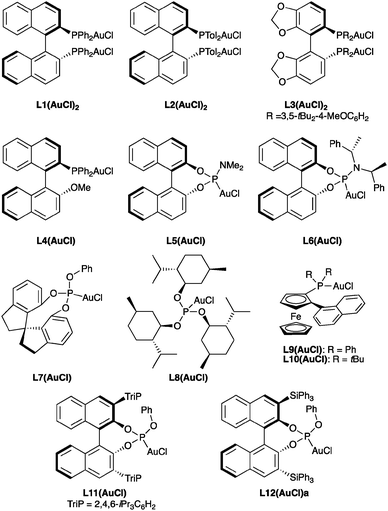 | ||
| Fig. 1 Chiral gold(I) complexes of the cyclization of 1,6-enyne 1a. | ||
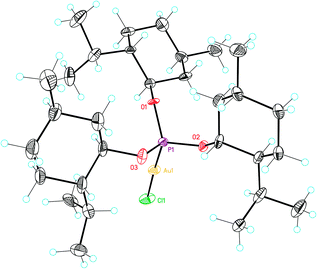 | ||
| Fig. 2 X-Ray crystal structure of gold complex L8(AuCl). ORTEP plot (50% thermal ellipsoids). | ||
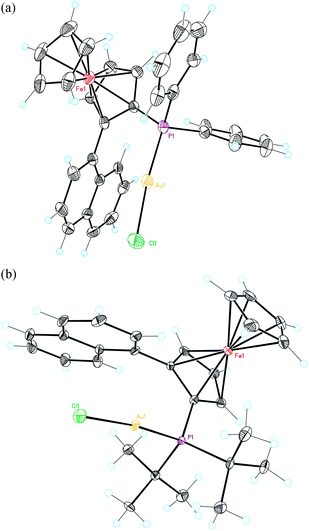 | ||
| Fig. 3 X-Ray crystal structures of ferrocenylphosphine gold complexes (a) L9(AuCl) and (b) L10(AuCl). ORTEP plot (50% thermal ellipsoids). | ||
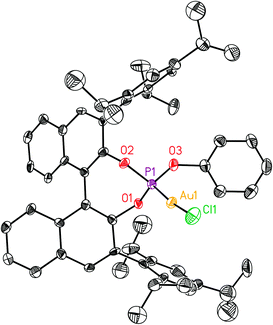 | ||
| Fig. 4 X-Ray crystal structure of gold complexes L11(AuCl) and L10(AuCl). ORTEP plot (50% thermal ellipsoids). Hydrogens are omitted for clarity. | ||
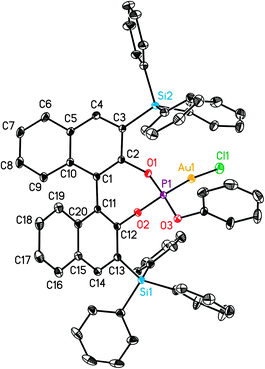 | ||
| Fig. 5 X-Ray crystal structure of gold complex L12(AuCl)a. ORTEP plot (50% thermal ellipsoids). Hydrogens are omitted for clarity. | ||
The cycloadditions were performed either at room temperature (condition A) or under microwave heating (condition B) (Table 1). Diphosphine–digold complexes L1(AuCl)2, L2(AuCl)2, and L3(AuCl)2 were investigated first (Table 1, entries 1–9). Cycloadduct 2a was obtained in all cases in good to excellent yield but only with low to moderate enantioselectivities. The best results with these diphosphine–digold complexes (56% ee) were obtained with L2(AuCl)2 in CHCl3 using AgPF6 under both conditions A and B (Table 1, entries 7 and 8). Using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of the digold complex to silver salt, under conditions in which the monocationic species are presumably formed, low enantioselectivities were observed. Biaryl gold–phosphine complex L4(AuCl) with the (R)-MOP ligand gave low enantiomeric excesses (Table 1, entries 11–13). BINOL-derived phosphoramidite complexes L5(AuCl) and L6(AuCl) also led to 2a in excellent yield but very poor enantioselectivities (Table 1, entries 14–19). Whereas reactions of complexes L7(AuCl)–L11(AuCl) led to poor to moderate enantioselectivities (Table 1, entries 20–27), results with phosphite gold complex L12(AuCl) were more promising (Table 1, entries 28 and 29). Although the enantiomeric excess was only marginally better than that obtained with L2(AuCl)2, phosphite gold complex L12(AuCl) was a significantly more reactive catalyst, leading to 2a in nearly quantitative yield in 12 h reaction time (vs. 24 h required with L2(AuCl)2).
1 ratio of the digold complex to silver salt, under conditions in which the monocationic species are presumably formed, low enantioselectivities were observed. Biaryl gold–phosphine complex L4(AuCl) with the (R)-MOP ligand gave low enantiomeric excesses (Table 1, entries 11–13). BINOL-derived phosphoramidite complexes L5(AuCl) and L6(AuCl) also led to 2a in excellent yield but very poor enantioselectivities (Table 1, entries 14–19). Whereas reactions of complexes L7(AuCl)–L11(AuCl) led to poor to moderate enantioselectivities (Table 1, entries 20–27), results with phosphite gold complex L12(AuCl) were more promising (Table 1, entries 28 and 29). Although the enantiomeric excess was only marginally better than that obtained with L2(AuCl)2, phosphite gold complex L12(AuCl) was a significantly more reactive catalyst, leading to 2a in nearly quantitative yield in 12 h reaction time (vs. 24 h required with L2(AuCl)2).
| Entry | Au complex | AgX | Conditions | Time | Yield (%) | ee (%) |
|---|---|---|---|---|---|---|
| a Au complex (2.5 mol%) and AgX (2.5 or 5 mol% for mono and digold complexes, respectively). Conditions A: 23 °C, CH2Cl2. Conditions B: microwave heating at 80 °C, CH2Cl2.b Reaction in CHCl3.c Conversion determined using 1H NMR.d Reaction in benzene.e Reaction at −20 °C. | ||||||
| 1 | L1(AuCl)2 | AgSbF6 | A | 24 h | 71 | 24 |
| 2 | L1(AuCl)2 | AgSbF6 | B | 18 min | 92 | 7 |
| 3 | L1(AuCl)2 | AgPF6 | A | 24 h | 81 | 31 |
| 4 | L1(AuCl)2 | AgPF6 | B | 18 min | 90 | 39 |
| 5 | L2(AuCl)2 | AgSbF6 | A | 30 h | 90 | 25 |
| 6 | L2(AuCl)2 | AgSbF6 | Ab | 18 min | 80 | 38 |
| 7 | L2(AuCl)2 | AgPF6 | Ab | 24 h | 89 | 56 |
| 8 | L2(AuCl)2 | AgPF6 | Bb | 15 min | 89 | 56 |
| 9 | L3(AuCl)2 | AgBF4 | A | 16 h | 91 | 25 |
| 10 | L4(AuCl) | AgSbF6 | A | 78 h | 56 | 18 |
| 11 | L4(AuCl) | AgSbF6 | B | 18 min | 78 | 20 |
| 12 | L4(AuCl) | AgPF6 | A | 78 h | 67 | 23 |
| 13 | L4(AuCl) | AgPF6 | B | 18 min | 84 | 25 |
| 14 | L5(AuCl) | AgSbF6 | A | 24 h | 91 | 8 |
| 15 | L5(AuCl) | AgSbF6 | B | 18 min | 95 | 12 |
| 16 | L5(AuCl) | AgPF6 | A | 24 h | 88 | 9 |
| 17 | L5(AuCl) | AgPF6 | B | 18 min | 94 | 14 |
| 18 | L6(AuCl) | AgSbF6 | B | 18 min | 95 | 5 |
| 19 | L6(AuCl) | AgPF6 | B | 18 min | 94 | 4 |
| 20 | L7(AuCl) | AgSbF6 | A | 12 h | 92 | 26 |
| 21 | L8(AuCl) | AgSbF6 | A | 2 h | 98 | <1 |
| 22 | L9(AuCl) | AgSbF6 | A | 24 h | >99c | 35 |
| 23 | L9(AuCl) | OTf | Ad | 24 h | >99c | 46 |
| 24 | L9(AuCl) | NTf2 | Ad | 24 h | 60c | 50 |
| 25 | L10(AuCl) | AgSbF6 | Ae | 24 h | >99c | 50 |
| 26 | L10(AuCl) | AgSbF6 | Ad | 24 h | >99c | 39 |
| 27 | L11(AuCl) | AgSbF6 | A | 12 h | 92 | 26 |
| 28 | L12(AuCl) | AgSbF6 | A | 12 h | 99 | 57 |
| 29 | L12(AuCl) | AgBF4 | A | 16 h | 90 | 57 |
Overall, the structures of Au(I) complexes L11(AuCl) and L12(AuCl)a in the solid state are similar (Fig. 4 and 5), although the Au–P–OPh angle in L12(AuCl)a (102.90°) is significantly more acute than that of L11(AuCl) (114.98°). Complex L12(AuCl)a shows a cone-shaped binding pocket surrounding with a closest distance of 3.304 Å between the gold centre and a phenyl ring of one of the SiPh3 groups, which is within the range (3.0–3.2 Å) observed in gold(I) complexes in bulky biaryl Buchwald phosphines.35 This weak Au(I)–arene interaction is not present in complex L11(AuCl).
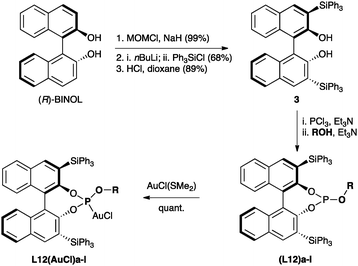
The preparation of a series of phosphite ligands (L12)a–n with different OR groups can be easily carried out using known methods31,32 from commercially available (R)-BINOL36 by known procedures via 3,3′-bis(triphenylsilyl)-1,1′-bi-2-naphthol (3) (Scheme 3),37 which is also commercially available. Ligands (L12)a–n were routinely purified by chromatography on silica gel under an inert atmosphere and the corresponding gold(I) complexes L12(AuCl)a–n were prepared in quantitative yields by reaction with [AuCl(SMe2)].
We assayed the catalytic activity of gold(I) complexes L12(AuCl)a–l (5 mol%) by mixing with AgSbF6 (5 mol%) at 0 °C in CH2Cl2, followed by addition of substrate 1a and slowly warming the reaction mixture to 23 °C over 2 h (Table 2).
| Entry | Au complex | R | ee (%) |
|---|---|---|---|
| a Au complex (5 mol%) and AgSbF6 (5 mol%), 0 to 23 °C, 2 h, CH2Cl2.b Reaction at −20 °C for 4 h.c Reaction at −20 °C for 16 h.d Reaction at −25 °C for 36 h.e Reaction at 0 °C for 7 h. | |||
| 1 | L12(AuCl)a | Ph | 70 |
| 2 | L12(AuCl)b | m-Tol | 72 |
| 3 | L12(AuCl)c | p-Tol | 80 |
| 4b | L12(AuCl)c | p-Tol | 83 |
| 5 | L12(AuCl)d | 4-tBuC6H4 | 82 |
| 6c | L12(AuCl)d | 4-tBuC6H4 | 88 |
| 7 | L12(AuCl)e | 4-MeOC6H4 | 60 |
| 8 | L12(AuCl)f | 2,4-Me2C6H3 | 74 |
| 9 | L12(AuCl)g | 3,5-Me2C6H3 | 81 |
| 10 | L12(AuCl)h | 2,4,6-Cl3C6H2 | 46 |
| 11 | L12(AuCl)i | 2-Napht | 70 |
| 12 | L12(AuCl)j | Me | 5 |
| 13d | L12(AuCl)k | PhCH2 | 81 |
| 14e | L12(AuCl)l | 3,5-tBu2C6H3CH2 | 74 |
Under these conditions, L12(AuCl)a led to 2a in 70% ee (Table 2, entry 1). The enantioselectivity was raised further by using phosphite ligands L12 derived from p-alkylsubstituted phenols (Table 2, entries 3–6). The best result (88% ee) was achieved with L12(AuCl)d derived from the tert-butylphenol group when the reaction was performed at −20 °C (Table 2, entry 6).38 Satisfactory results were also obtained with L12(AuCl)g and L12(AuCl)k (Table 2, entries 9 and 13).
The reactions with the best catalyst L12(AuCl)d were slower (16–24 h) in 1,2-dichloroethane, ethyl ether, or acetone as solvent (63–82% ee), whereas no reaction was observed in toluene or 1,4-dioxane after 1–2 days. On the other hand, changing the silver salt from AgSbF6 to AgOTf or AgNTf2 did not significantly affect the reactivity and enantioselectivity, while slightly lower enantiomeric excesses were obtained with AgPF6.39
Finally, the optimized phosphite gold(I) catalyst L12(AuCl)d was applied for the cyclization of 1,6-enynes 1a–e using 2 mol% catalyst loadings (Table 3). Substrate 1b with a p-OMe group gave the corresponding cycloadduct 2b in good yield and enantioselectivity, although a longer reaction time was required (Table 3, entry 2). Good enantioselectivity was also obtained with enyne 2c bearing a p-Me group (Table 3, entry 3). Sterically more demanding substrate 1d could also be cyclized in 70% yield and 79% ee (Table 3, entry 4). Finally, cyclization of 2e with a strong electron-withdrawing p-NO2 group at the phenyl ring gave cycloadduct 1d in 80% yield and 73% ee at 0 °C (Table 3, entry 5).
|
||||||
|---|---|---|---|---|---|---|
| Entry | Enyne | R | T (°C) | Time (h) | Product (yield, %) | ee (%) |
| 1 | 1a | H | −20 | 18 | 95 | 88 |
| 2 | 1b | p-MeO | −20 | 30 | 85 | 86 |
| 3 | 1c | p-Me | −20 | 15 | 98 | 87 |
| 4 | 1d | o-Me | −20 | 30 | 70 | 79 |
| 5 | 1e | p-O2N | 0 | 15 | 80 | 73 |
Conclusions
We have developed a series of chiral phosphite gold(I) complexes L12(AuCl)a–n that are easily prepared in a modular manner from BINOL. Cyclization of aryl-substituted 1,6-enynes with these complexes in the presence of a silver salt occurs with enantiomeric ratios ranging from 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 up to 94
14 up to 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6. It is also important to note that these chiral catalysts rival in reactivity with the most active catalysts for the cyclization of this more challenging class of compounds bearing a disubstituted alkyne.
6. It is also important to note that these chiral catalysts rival in reactivity with the most active catalysts for the cyclization of this more challenging class of compounds bearing a disubstituted alkyne.
Acknowledgements
We thank the MICINN (CTQ2010-16088/BQU), the AGAUR (2009 SGR 47), the European Research Council (Advanced Grant No. 321066), and the ICIQ Foundation for financial support. We also thank the ICIQ X-ray diffraction unit for the X-ray diffraction structures.Notes and references
- E. Jiménez-Núñez and A. M. Echavarren, Chem. Rev., 2008, 108, 3326–3350 CrossRef.
- D. J. Gorin, B. D. Sherry and F. D. Toste, Chem. Rev., 2008, 108, 3351–3378 CrossRef CAS.
- (a) A. Fürstner and P. W. Davies, Angew. Chem., Int. Ed., 2007, 46, 3410–3449 CrossRef; (b) A. Fürstner, Chem. Soc. Rev., 2009, 38, 3208–3221 RSC.
- V. Michelet, P. Y. Toulleca and J.-P. Genêt, Angew. Chem., Int. Ed., 2008, 47, 4268–4315 CrossRef CAS.
- (a) N. Huguet and A. M. Echavarren, in Asymmetric Synthesis II, ed. M. Christmann and S. Bräse, Wiley-VCH Verlag, 2012, ch. 26, pp. 205–212 Search PubMed; (b) R. A. Widenhoefer, Chem.–Eur. J., 2008, 14, 5382–5391 CrossRef CAS; (c) N. Bongers and N. Krause, Angew. Chem., Int. Ed., 2008, 47, 2178–2181 CrossRef CAS; (d) S. Sengupta and X. Shi, ChemCatChem, 2010, 2, 609–619 CrossRef CAS; (e) P. Pradal, P. Y. Toullec and V. Michelet, Synthesis, 2011, 1501–1514 Search PubMed; (f) A. Marinetti, H. Jullien and A. Voituriez, Chem. Soc. Rev., 2012, 41, 4884–4908 RSC.
- (a) M. J. Johansson, D. J. Gorin, S. T. Staben and F. D. Toste, J. Am. Chem. Soc., 2005, 127, 18002–18003 CrossRef CAS; (b) F. Kleinbeck and F. D. Toste, J. Am. Chem. Soc., 2009, 131, 9178–9179 CrossRef CAS; (c) G. L. Hamilton, E. J. Kang, M. Mba and F. D. Toste, Science, 2007, 317, 496–499 CrossRef CAS.
- (a) C. Liu and R. A. Widenhoefer, Org. Lett., 2007, 9, 1935–1938 CrossRef CAS; (b) P. Mukherjee and R. A. Widenhoefer, Angew. Chem., Int. Ed., 2012, 51, 1405–1407 CrossRef CAS.
- C.-M. Chao, D. Beltrami, P. Y. Toullec and V. Michelet, Chem. Commun., 2009, 6988–6990 RSC.
- A. Martínez, P. García-García, M. A. Fernández-Rodríguez, F. Rodríguez and R. Sanz, Angew. Chem., Int. Ed., 2010, 49, 4633–4637 CrossRef.
- I. Alonso, B. Trillo, F. López, S. Montserrat, G. Ujaque, L. Castedo, A. Lledós and J. L. Mascareñas, J. Am. Chem. Soc., 2009, 131, 13020–13030 CrossRef CAS.
- A. Z. González, D. Benitez, E. Tkatchouk, W. A. Goddard III and F. D. Toste, J. Am. Chem. Soc., 2011, 133, 5500–5507 CrossRef.
- S. Suárez-Pantiga, C. Hernández-Díaz, E. Rubio and J. M. González, Angew. Chem., Int. Ed., 2012, 51, 11552–11555 CrossRef.
- L.-I. Rodríguez, T. Roth, J. L. Fillol, H. Wadepohl and L. H. Gade, Chem.–Eur. J., 2012, 18, 3721–3728 CrossRef.
- S. Handa and L. M. Slaughter, Angew. Chem., Int. Ed., 2012, 51, 2912–2915 CrossRef CAS.
- S. G. Sethofer, T. Mayer and F. D. Toste, J. Am. Chem. Soc., 2010, 132, 8276–8277 CrossRef CAS.
- M. P. Muñoz, J. Adrio, J. C. Carretero and A. M. Echavarren, Organometallics, 2005, 24, 1293–1300 CrossRef.
- (a) Y. Matsumoto, K. B. Selim, H. Nakanishi, K. Yamada, Y. Yamamoto and K. Tomioka, Tetrahedron Lett., 2010, 51, 404–406 CrossRef CAS; (b) W. Wang, J. Yang, F. Wang and M. Shi, Organometallics, 2011, 30, 3859–3869 CrossRef CAS.
- C.-M. Chao, E. Genin, P. Y. Toullec, J.-P. Genêt and V. Michelet, J. Organomet. Chem., 2009, 694, 538–545 CrossRef CAS.
- (a) D. Brissy, M. Skander, P. Retailleau and A. Marinetti, Organometallics, 2007, 26, 5782–5785 CrossRef CAS; (b) P. Y. Toullec, C.-M. Chao, Q. Chen, S. Gladiali, J.-P. Genêt and V. Michelet, Adv. Synth. Catal., 2008, 350, 2401–2408 CrossRef CAS; (c) D. Brissy, M. Skander, H. Jullien, P. Retailleau and A. Marinetti, Org. Lett., 2009, 11, 2137–2139 CrossRef CAS; (d) H. Jullien, D. Brissy, P. Retailleau and A. Marinetti, Eur. J. Inorg. Chem., 2011, 5083–5086 CrossRef CAS; (e) H. Jullien, D. Brissy, R. Sylvain, P. Retailleau, J.-V. Naubron, S. Gladiali and A. Marinetti, Adv. Synth. Catal., 2011, 353, 1109–1124 CrossRef CAS.
- (a) C. Nieto-Oberhuber, S. López and A. M. Echavarren, J. Am. Chem. Soc., 2005, 127, 6178–6179 CrossRef CAS; (b) C. Nieto-Oberhuber, P. Pérez-Galán, E. Herrero-Gómez, T. Lauterbach, C. Rodríguez, S. López, C. Bour, A. Rosellón, D. J. Cárdenas and A. M. Echavarren, J. Am. Chem. Soc., 2008, 130, 269–279 CrossRef CAS.
- D. M. Fort, R. P. Ubillas, C. D. Mendez, S. D. Jolad, W. D. Inman, J. R. Carney, J. L. Chen, T. T. Ianiro, C. Hasbun, R. C. Bruening, J. Luo, M. J. Reed, M. Iwu, T. J. Carlson, S. R. King, D. E. Bierer and R. Cooper, J. Org. Chem., 2000, 65, 6534–6539 CrossRef CAS.
- D. W. Laird, R. Poole, M. Wikström and I. A. van Altena, J. Nat. Prod., 2007, 70, 671–674 CrossRef CAS.
- Total synthesis of (−)-pycnanthuquinone C: F. Löbermann, P. Mayer and D. Trauner, Angew. Chem., Int. Ed., 2010, 49, 6199–6202 CrossRef.
- V. López-Carrillo, N. Huguet, Á. Mosquera and A. M. Echavarren, Chem.–Eur. J., 2011, 17, 10972–10978 CrossRef.
- P. Pérez-Galán, PhD thesis, ICIQ-URV, 2005–2010; N. Delpont, PhD thesis, ICIQ-URV, 2007–2011.
- C.-M. Chao, M. R. Vitale, P. Y. Toullec, J.-P. Genêt and V. Michelet, Chem.–Eur. J., 2009, 15, 1319–1323 CrossRef CAS.
- M. Raducan, C. Rodríguez-Escrich, X. C. Cambeiro, E. Escudero-Adán, M. A. Pericàs and A. M. Echavarren, Chem. Commun., 2011, 47, 4893–4895 RSC.
- P. W. N. M. van Leeuwen, P. C. J. Kamer, C. Claver, O. Pàmies and M. Diéguez, Chem. Rev., 2011, 111, 2077–2118 CrossRef CAS.
- H. M. Amijs, V. López-Carrillo, M. Raducan, P. Pérez-Galán, C. Ferrer and A. M. Echavarren, J. Org. Chem., 2008, 73, 7721–7730 CrossRef.
- D. Benitez, N. D. Shapiro, E. Tkatchouk, Y. Wang, W. A. Goddard III and D. F. Toste, Nat. Chem., 2009, 1, 482–486 CrossRef CAS.
- (a) R. B. Bedford, Y.-N. Chang, M. F. Haddow and C. L. McMullin, Dalton Trans., 2011, 40, 9034–9041 RSC; (b) R. B. Bedford, Y.-N. Chang, M. F. Haddow and C. L. McMullin, Dalton Trans., 2011, 40, 9042–9050 RSC.
- (a) Phopshite L11: M. Kawasaki, P. Li and H. Yamamoto, Angew. Chem., Int. Ed., 2008, 47, 3795–3597 CrossRef CAS; (b) Phopshite L12a: A. Sakakura, M. Sakuma and K. Ishihara, Org. Lett., 2011, 13, 3130–3797 CrossRef CAS.
- A. Z. González and F. D. Toste, Org. Lett., 2009, 12, 200–203 CrossRef.
- (a) H. Teller, S. Flügge, R. Goddard and A. Fürstner, Angew. Chem., Int. Ed., 2010, 49, 1949–1953 CrossRef CAS; (b) H. Teller, M. Corbet, L. Mantilli, G. Gopakumar, R. Goddard, W. Thiel and A. Fürstner, J. Am. Chem. Soc., 2012, 134, 15331–15342 CrossRef CAS.
- (a) E. Herrero-Gómez, C. Nieto-Oberhuber, S. López, J. Benet-Buchholz and A. M. Echavarren, Angew. Chem., Int. Ed., 2006, 45, 5455–5459 CrossRef; (b) P. Pérez-Galán, N. Delpont, E. Herrero-Gómez, F. Maseras and A. M. Echavarren, Chem.–Eur. J., 2010, 16, 5324–5332 CrossRef.
- J. M. Brunel, Chem. Rev., 2005, 105, 4233 CrossRef CAS.
- V. E. Albrow, A. J. Blake, R. Fryatt, C. Wilson and S. Woodward, Eur. J. Org. Chem., 2006, 2549–2557 CrossRef CAS.
-
(a) Adduct 2a of 88% ee (determined by HPLC) has [α]20D − 25.0 ± 2.0 (c = 0.11, CHCl3). This value contrasts with that reported for 2a of 93% ee, [α]21D + 14.8, (c = 0.93, CHCl3) in ref. 26. When the cyclization of 2a with L(AuCl)2 (L = (R)-4-MeO-3,5-(tBu)2MeOBIPHEP = DTBM-MeO-BIPHEP) (3 mol%) and AgOTf (6 mol%) in Et2O, in addition to 2a (74 ee, estimated by chiral HPLC), known 4 (ref. 38b) was also obtained (72
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 ratio). We could not find conditions that would allow the full resolution of 4 and the enantiomers of 2a by chiral HPLC;
(b) S. Porcel and A. M. Echavarren, Angew. Chem., Int. Ed., 2007, 46, 2672–2676 CrossRef CAS
28 ratio). We could not find conditions that would allow the full resolution of 4 and the enantiomers of 2a by chiral HPLC;
(b) S. Porcel and A. M. Echavarren, Angew. Chem., Int. Ed., 2007, 46, 2672–2676 CrossRef CAS
. - See ESI† for details.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental results and NMR data. CCDC 933751 (L11(AuCl)), 933752 (L12(AuCl)a), 933753 (L9(AuCl)), 933754 (L10(AuCl)), 933756 (L8(AuCl)), 933757 (L12(AuCl)e). For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c3cy00250k |
| This journal is © The Royal Society of Chemistry 2013 |