 Open Access Article
Open Access ArticleMultivalent glycoconjugates as anti-pathogenic agents†
Anna
Bernardi
a,
Jesus
Jiménez-Barbero
b,
Alessandro
Casnati
c,
Cristina
De Castro
d,
Tamis
Darbre
e,
Franck
Fieschi
f,
Jukka
Finne
g,
Horst
Funken
h,
Karl-Erich
Jaeger
h,
Martina
Lahmann
i,
Thisbe K.
Lindhorst
j,
Marco
Marradi
k,
Paul
Messner
l,
Antonio
Molinaro
d,
Paul V.
Murphy
m,
Cristina
Nativi
n,
Stefan
Oscarson
o,
Soledad
Penadés
k,
Francesco
Peri
p,
Roland J.
Pieters
q,
Olivier
Renaudet
r,
Jean-Louis
Reymond
e,
Barbara
Richichi
n,
Javier
Rojo
s,
Francesco
Sansone
c,
Christina
Schäffer
l,
W. Bruce
Turnbull
t,
Trinidad
Velasco-Torrijos
u,
Sébastien
Vidal
v,
Stéphane
Vincent
w,
Tom
Wennekes
x,
Han
Zuilhof
xy and
Anne
Imberty
*z
aUniversità di Milano, Dipartimento di Chimica Organica e Industriale and Centro di Eccellenza CISI, via Venezian 21, 20133 Milano, Italy
bCentro de Investigaciones Biológicas, CSIC, 28040 Madrid, Spain
cUniversità degli Studi di Parma, Dipartimento di Chimica, Parco Area delle Scienze 17/a, 43100 Parma, Italy
dDepartment of Chemical Sciences, Università di Napoli Federico II, Complesso Universitario Monte Santangelo, Via Cintia 4, I-80126 Napoli, Italy
eDepartment of Chemistry and Biochemistry, University of Berne, Freiestrasse 3, CH-3012, Berne, Switzerland
fInstitut de Biologie Structurale, 41 rue Jules Horowitz, 38027 Grenoble Cedex 1, France
gDepartment of Biosciences, University of Helsinki, P. O. Box 56, FI-00014 Helsinki, Finland
hInstitute of Molecular Enzyme Technology, Heinrich-Heine-University Düsseldorf, Forschungszentrum Jülich, D-42425 Jülich, Germany
iSchool of Chemistry, Bangor University, Deiniol Road Bangor, Gwynedd LL57 2UW, UK
jOtto Diels Institute of Organic Chemistry, Christiana Albertina University of Kiel, Otto-Hahn-Platz 3-4, D-24098 Kiel, Germany
kLaboratory of GlycoNanotechnology, CIC biomaGUNE and CIBER-BBN, P° de Miramón 182, 20009 San Sebastián, Spain
lDepartment of NanoBiotechnology, NanoGlycobiology Unit, University of Natural Resources and Life Sciences, Muthgasse 11, A-1190 Vienna, Austria
mSchool of Chemistry, National University of Ireland, Galway, University Road, Galway, Ireland
nDipartimento di Chimica, Universitá degli Studi di Firenze, Via della Lastruccia, 13, I-50019 Sesto Fiorentino – Firenze, Italy
oCentre for Synthesis and Chemical Biology, UCD School of Chemistry and Chemical Biology, University College Dublin, Belfield, Dublin 4, Ireland
pOrganic and Medicinal Chemistry, University of Milano-Bicocca, Piazza della Scienza, 2, 20126 Milano, Italy
qDepartment of Medicinal Chemistry and Chemical Biology, Utrecht Institute for Pharmaceutical Sciences, Utrecht University, P.O. Box 80082, 3508 TB Utrecht, The Netherlands
rDépartement de Chimie Moléculaire, UMR-CNRS 5250 & ICMG FR 2607, Université Joseph Fourier, BP53, 38041 Grenoble Cedex 9, France
sGlycosystems Laboratory, Instituto de Investigaciones Químicas, CSIC – Universidad de Sevilla, Av. Américo Vespucio, 49, Seville 41092, Spain
tSchool of Chemistry and Astbury Centre for Structural Molecular Biology, University of Leeds, Leeds, LS2 9JT, UK
uDepartment of Chemistry, National University of Ireland, Maynooth, Maynooth Co. Kildare, Ireland
vInstitut de Chimie et Biochimie Moléculaires et Supramoléculaires UMR 5246, CNRS, Université Claude Bernard Lyon 1, 43 Boulevard du 11 Novembre 1918, F-69622 Villeurbanne, France
wUniversity of Namur (FUNDP), Département de Chimie, Laboratoire de Chimie Bio-Organique, rue de Bruxelles 61, B-5000 Namur, Belgium
xLaboratory of Organic Chemistry, Wageningen University, Dreijenplein 8, 6703 HB Wageningen, The Netherlands
yDepartment of Chemical and Materials Engineering, King Abdulaziz University, Jeddah, Saudi Arabia
zCentre de Recherche sur les Macromolécules Végétales (CERMAV – CNRS), affiliated with Grenoble-Université and ICMG, F-38041 Grenoble, France. E-mail: Imberty@cermav.cnrs.fr
First published on 19th December 2012
Abstract
Multivalency plays a major role in biological processes and particularly in the relationship between pathogenic microorganisms and their host that involves protein–glycan recognition. These interactions occur during the first steps of infection, for specific recognition between host and bacteria, but also at different stages of the immune response. The search for high-affinity ligands for studying such interactions involves the combination of carbohydrate head groups with different scaffolds and linkers generating multivalent glycocompounds with controlled spatial and topology parameters. By interfering with pathogen adhesion, such glycocompounds including glycopolymers, glycoclusters, glycodendrimers and glyconanoparticles have the potential to improve or replace antibiotic treatments that are now subverted by resistance. Multivalent glycoconjugates have also been used for stimulating the innate and adaptive immune systems, for example with carbohydrate-based vaccines. Bacteria present on their surfaces natural multivalent glycoconjugates such as lipopolysaccharides and S-layers that can also be exploited or targeted in anti-infectious strategies.
 Anne Imberty | Dr. Imberty is Research Director at the Centre de Recherches sur les Macromolécules Végétales (CERMAV), affiliated to the Centre National de la Recherche Scientifique (CNRS) based at Grenoble, France. She graduated in biology from Ecole Normale Supérieure in Paris. In 1984, she joined the CNRS in Grenoble and did her PhD on starch structure. She started studying protein-carbohydrate interaction during her post-doc in Toronto. Since 1999 she has a senior research position in CNRS-Grenoble and is the head of the Molecular Glycobiology group. Her research interests are in the field of structural glycobiology, with main interest on biologically active oligosaccharides and their interaction with lectins and glycosyltransferases. She now focuses on characterization of the molecular basis of recognition between lectins from pathogens and human glycoconjugates and design of glycocompounds with anti-infectious properties. |
1. Introduction
Carbohydrates constitute the most abundant class of biomolecules on Earth. They play roles in the natural world as diverse as energy storage, molecular recognition for intracellular trafficking or interactions between pathogenic microorganisms and viruses and the surfaces of mammalian cells.1 The carbohydrates present inside and at the surface of cells mediate many biological processes that are fundamentally important for both the healthy and diseased states of living organisms.2,3 Usually carbohydrates are covalently linked to other biomolecules such as proteins or lipids. The molecular and supramolecular scaffolds of such glycoconjugates provide them with a very important property: multivalency.4–7 Individual interactions between sugars and proteins are very weak, however, multiple simultaneous interactions between carbohydrate ligands and their receptors reinforce one-another like molecular velcro to achieve functionally useful avidities. Recent advances in carbohydrate microarray technologies8–14 combined with the utilization of freely accessible resources of the Consortium for Functional Glycomics (http://www.functionalglycomics.org/) have greatly expanded our knowledge of carbohydrate-binding proteins, and reinforced the importance of multivalent interactions.15–21 It has become apparent that the avidities and selectivities of specific interactions are very dependent on the density of the sugar groups and also the chemical structure of the linker group to the underlying multivalent scaffold. Translating this knowledge for the design and preparation of multivalent glycoconjugates remains a significant challenge to diagnose and treat diseases.22–25Protein–carbohydrate interactions frequently mediate the first step of the infection process for many pathogens including viruses, fungi, bacteria, and bacterial toxins.26–29 Therefore, a vast array of unnatural glycoconjugates (neoglycoconjugates) with various valencies and spatial arrangement of the ligands have been constructed to prevent or treat diseases caused by pathogens. Scaffolds based on proteins,30–32 polymers,33–36 calixarenes,37–40 dendrimers,41–45 cyclodextrins,46,47 cyclopeptides,48–50 fullerenes,51,52 gold nanoparticles,53,54 and quantum dots55–57 provide nanoscale materials with anti-adhesive and cell targeting properties. Such structures that can competitively interfere with the recognition processes between host cells and pathogens have the potential to prevent colonisation or even reverse the formation of biofilms. Another alternative to fight pathogens relies on the utilization of glycoconjugates that can act as vaccines and immunomodulators. Vaccines have long relied on attenuated strains of microorganisms as a means of delivering the extracellular carbohydrate antigens. As the cell surface of pathogenic bacteria and viruses is often covered with unusual carbohydrates, structurally defined glycoconjugates displaying these structures are starting to emerge as the vaccines for the future.58,59 Following a lectin-mediated cellular uptake mechanism, such carbohydrate-based vaccines can prepare the immune defense mechanisms in advance of an infection, or to stimulate the body to protect itself against an existing chronic infection.
This review will give an overview of synthetic or natural multivalent glycoconjugates that can be used to inhibit the adhesion of viruses, bacterial toxins, and bacteria to host cells or to stimulate the innate and adaptive immune systems against these pathogens. In addition to the pioneering work of several groups mainly from North America,7,17,25,60,61 this field is currently flourishing in Europe, and this review is aimed at giving an overview of this very active domain.
2. Multivalent glycoconjugates against viral infection
2.1 Targeting DC-SIGN uptake by dendritic cells
Among pathogens, human immunodeficiency virus (HIV) represents one of the big challenges of current basic and medical research. Attempts to generate effective microbicides have failed, so that integrated systems and new strategies are needed in order to prevent or block the viral infection. Multivalent glycoconjugates can be utilized in novel vaccine approaches (see Section 6.2.1) or as inhibitors of first step of infection. One of the main pathways of HIV infection is mediated by antigen-presenting cells (APC) at the mucosal endothelium. The high-mannose glycans of envelope glycoprotein gp120 promote HIV infection by interaction with the C-type lectin DC-SIGN (dendritic cell-specific ICAM 3-grabbing non-integrin) expressed on dendritic cells.62 Dendritic cells, a main group of APC, migrate to the lymph nodes where they efficiently transfer the virus by so-called trans-infection to T lymphocytes where viral replication occurs. Mimicking the surface of the virus is a strategy for designing carbohydrate-based antiviral agents against HIV and Ebola infections.Glycodendritic compounds have been used as tools to study and to interfere with infectious processes in which DC-SIGN is involved with the aim to develop new antiviral drugs and immune modulators.63 The conjugation of glycomimetics to dendritic compounds has provided multivalent compounds with interesting antiviral activity. IC50s in the low nanomolar range have been obtained in biological assays using pseudotyped Ebola viral particles.64 Also, these compounds present very good activity as inhibitors of HIV trans-infection of T-cells, a more relevant infection model where DC-SIGN is implicated.65,66
Glycodendritic structures have also been prepared recently using a convergent approach with extensive use of the Cu(I) catalyzed azide–alkyne cycloaddition (CuAAC) reaction also known as “click” chemistry (Fig. 1). In this new strategy, first and second generation glycodendrons have been prepared conveniently functionalized for further conjugation on different scaffolds including a fullerene molecule or a virus-like particle protein.52,67
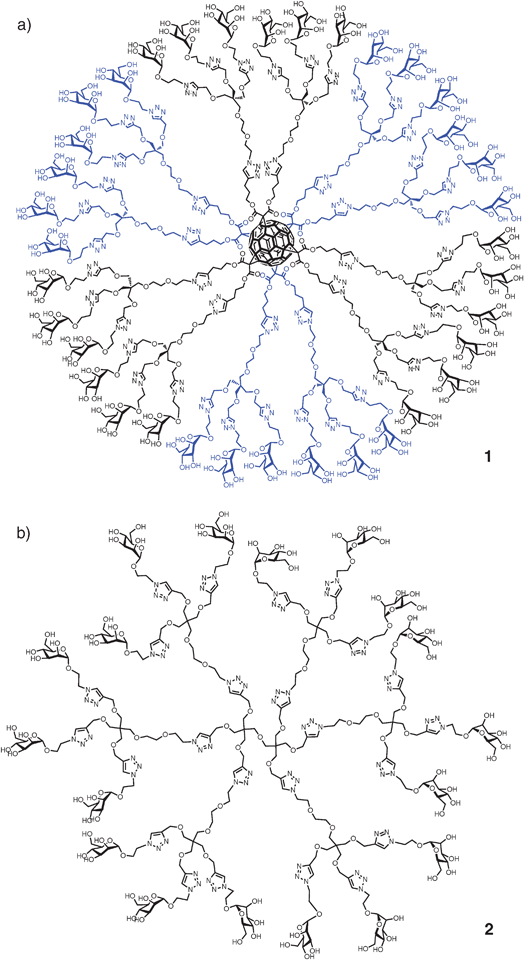 | ||
| Fig. 1 (a) Glycodendrofullerene 1 with 36 mannoses; (b) glycodendrimer 2 with 18 mannoses prepared using a CuAAC click reaction. | ||
The new glycodendritic compounds display a wide variety of valencies and spatial presentation of carbohydrate ligands. The glycodendrofullerenes (e.g., 1) prepared using this strategy are soluble under physiological conditions and present a very low cellular toxicity. The globular disposition of carbohydrates on this spherical scaffold provides an interesting multivalent system which allows the carbohydrates to be recognized by lectins in a multivalent manner. Antiviral activity of these compounds using pseudotyped Ebola viral particles is in the micromolar range.52,67
In another attempt to mimic the cluster presentation of high-mannose-type glycans on the HIV envelope, gold nanoparticles biofunctionalized with oligomannosides (manno-GNPs, 3a–3d, Fig. 2) of gp120 high-mannose type glycans have been prepared and tested as anti-HIV agents. These manno-GNPs inhibited the DC-SIGN/gp120 binding in the micro- to nanomolar range, while the corresponding monovalent oligomannosides required millimolar concentrations, as measured by surface plasmon resonance (SPR) experiments.68 Furthermore, manno-GNPs were able to inhibit the DC-SIGN-mediated HIV trans-infection of human activated peripheral blood mononuclear cells at nanomolar concentrations in an experimental setting, which mimics the natural route of virus transmission from dendritic cells to T lymphocytes.69
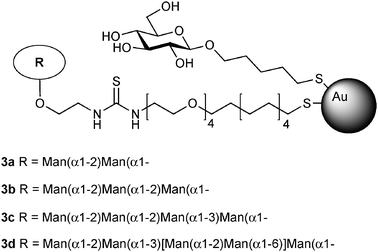 | ||
| Fig. 2 Gold nanoparticles 3a–3d bearing high-mannose type glycans (manno-GNPs) present on HIV envelope glycoprotein gp120 as anti-HIV synthetic glycoconjugates. | ||
2.2 Interfering with galectin attachment in lymph nodes
Galectins are lectins with affinity for β-galactosides which are involved in self/nonself recognition. Galectins from both invertebrates and vertebrates recognize a variety of viral and bacterial pathogens and protozoan parasites.70 Galectin-1 is abundant in thymus and lymph nodes and promotes HIV-1 infection by facilitating virus attachment to the host cell surface glycan.71 Given that this lectin is directly involved in pathogen recognition, it could provide a strategy for disrupting the galectins in virus–host invasion. Host galectins also recognize endogenous glycans on the host cell surface, which are important for certain necessary developmental and immunological processes.Structurally defined bivalent lactose-containing clusters have been designed for optimal binding to galectins.41,72–74 These compounds were evaluated for binding to the entire set of adhesion/growth-regulatory galectins from chicken. Differential sensitivities were detected between distinct galectin forms within the chicken series. Two of the bivalent glycoclusters, 4 and 5 (Fig. 3), were identified as sensors for different galectin subtypes. Most pronounced were the selectivities of these two glycoclusters for the chimera-type galectin (galectin-3).
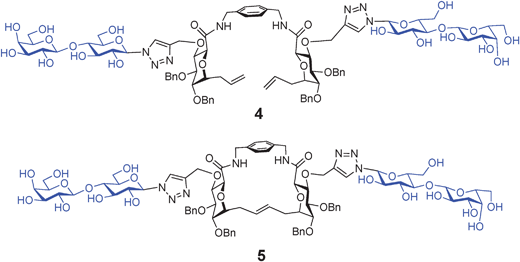 | ||
| Fig. 3 Bivalent glycoclusters 4 (acyclic) and 5 (macrocyclic), identified as sensors for different galectin subtypes. | ||
3. Multivalent glycoconjugates against bacterial toxins
Cholera, still a life-threatening disease in many parts of the world, is caused by the cholera toxin (CT) produced by Vibrio cholerae. This toxin consists of a single disease-causing A-subunit that is surrounded by five lectin-like B-subunits (CTB). The B-subunits are responsible for attachment of the toxin to the intestinal surface by binding to exposed GM1-oligosaccharide (GM1os) moieties.75 The five B-subunits represent a well-defined multivalent protein target with binding sites for the GM1os units spaced ca. 30 Å apart. Polyvalent CT inhibitors were developed starting from weak ligands, such as galactose. Huge potency increases (up to 106-fold with IC50 = 40 nM) compared to that of D-galactose were reported by Fan and co-workers, who prepared and screened a series of penta- and decavalent ligands with linkers of various lengths.76The advent of ‘click’ chemistry in combination with chemoenzymatic synthesis of the complex oligosaccharide enabled the assembly of multivalent versions of the GM1os77 into glycodendrimers (Fig. 4).78 Divalent compound 7a was almost 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times more potent as an inhibitor of CTB binding to GM1os than monovalent compound 6. This enhancement in inhibitory potency is related to the multivalent interactions between CTB and the divalent compound 7a, as the latter does not show an enhancement in binding to antibodies that do not allow multiple interactions.79
000 times more potent as an inhibitor of CTB binding to GM1os than monovalent compound 6. This enhancement in inhibitory potency is related to the multivalent interactions between CTB and the divalent compound 7a, as the latter does not show an enhancement in binding to antibodies that do not allow multiple interactions.79
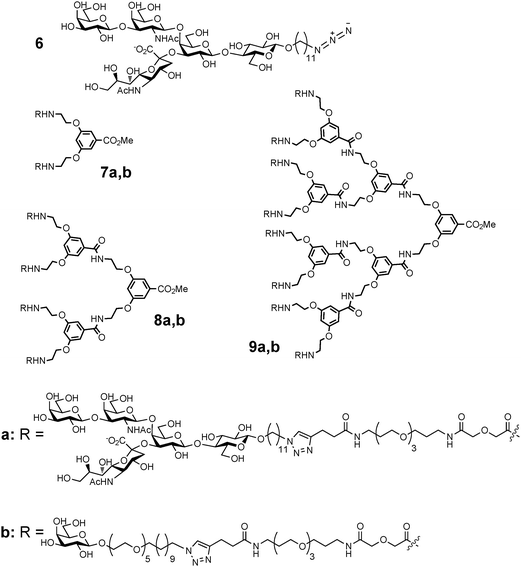 | ||
| Fig. 4 Structures of GM1os- (7a, 8a and 9a) and galactose (7b, 8b and 9b)-based inhibitors of cholera toxin binding. | ||
An additional increase was observed for tetravalent 8a (83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fold). The most complex glycodendrimer in this study, octavalent 9a, was 380
000 fold). The most complex glycodendrimer in this study, octavalent 9a, was 380![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fold more potent (IC50 = 50 pM and relative inhibitory potency of 47
000 fold more potent (IC50 = 50 pM and relative inhibitory potency of 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 per GM1os).78 A detailed study of the mode of action revealed that complex aggregates between the inhibitor and toxin are formed. These are possible because of the mismatch between the valencies of the toxin (five) versus those of the inhibitors (two, four, eight).80
500 per GM1os).78 A detailed study of the mode of action revealed that complex aggregates between the inhibitor and toxin are formed. These are possible because of the mismatch between the valencies of the toxin (five) versus those of the inhibitors (two, four, eight).80
The galactose dendrimers 7b, 8b and 9b are a simplified glycomimetic version of the multivalent GM1 derivatives. The inhibitory potency did suffer due to this modification since the relatively large binding site of the B-subunit remains partly unoccupied resulting in a lower binding affinity. Nevertheless, multivalency effects were able to counteract the lower binding affinity of galactose and the inhibitory potencies of compounds 8b and 9b were shown to be competitive with the natural GM1os ligand.81
A series of ganglioside mimics, in which the non-interacting oligosaccharide backbone of GM1os was replaced by an appropriate cyclohexanediol, was chosen to reproduce the topological features of the 3,4-disubstituted galactose residue (Gal-II) in GM1os.82,83 The divalent presentation of a structurally simplified second generation mimic 10 on a functionalized calix[4]arene scaffold (Fig. 5) led to a 3800-fold (1900-fold per sugar mimic) enhancement of CTB affinity, thus reaching the potency of GM1os itself.84 Although computational studies show that the divalent ligand 11 could easily span two binding sites on cholera toxin, NMR data indicate that the action of this divalent ligand is likely to involve additional interactions between the linker and the protein.
![Structurally simplified GM1os mimic 10 grafted onto a functionalized calix[4]arene scaffold to give divalent ligand 11.](/image/article/2013/CS/c2cs35408j/c2cs35408j-f5.gif) | ||
| Fig. 5 Structurally simplified GM1os mimic 10 grafted onto a functionalized calix[4]arene scaffold to give divalent ligand 11. | ||
4. Multivalent glycoconjugates against bacterial adhesion to human cells
4.1 Uropathogenic E. coli
Nevertheless, avidity effects have been frequently observed with a variety of multivalent mannose-containing glycomimetics, like 12–14 (Fig. 6a–c). Such avidity can originate from statistical effects arising from (i) a higher concentration of mannose in the proximity of the carbohydrate binding site, (ii) existence of additional carbohydrate binding sites on the lectin FimH, or (iii) occurrence of the natural multivalent process, since fimbriae occur on the bacterial surface in several hundreds of copies. Thus, mannose-terminated multivalent glycocompounds have become important to test mannose-specific bacterial adhesion in a supramolecular context.88,91 Meanwhile, testing of type 1 fimbriae-mediated bacterial adhesion has been greatly facilitated by employing GFP-transfected strains.92 Interestingly, adhesion on multivalent glycomaterials can be utilized for aggregating E. coli and removing them from solution with the use of appropriate filters. Glyconanodiamonds decorated with mannose (Fig. 6d) have been shown to be able to clean bacteria-polluted water.93
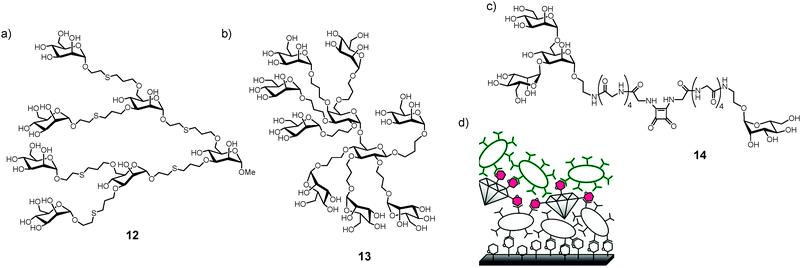 | ||
| Fig. 6 Examples of various multivalent glycoconjugates inhibiting type 1 fimbriae-mediated bacterial adhesion. (a) Octopus glycosides 12; (b) glycodendrimer 13; (c) bifunctional ligand 14 to test multiple binding sites on FimH; (d) glyconanodiamonds to remove pathogenic bacteria from polluted water sources, a sandwich assay is displayed, utilizing two different bacterial strains. | ||
Fullerene hexakis-adducts bearing 12 peripheral mannose moieties (15–20) have been prepared by grafting sugar derivatives onto the fullerene core52 and assayed as inhibitors of FimH (Fig. 7).88 Dissociation constants in the range of 12 to 95 nM were measured using isothermal titration calorimetry (ITC), surface plasmon resonance (SPR) and hemagglutination assays. Most importantly, the number of possible interactions between the multimers and the lectin and the average binding strength per functional mannose unit could be measured. Thus, this study demonstrated for the first time that a globular C60 structure can accommodate up to seven FimH molecules.
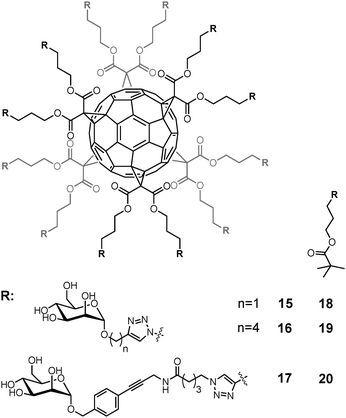 | ||
| Fig. 7 Dodecavalent mannofullerenes 15–20 as FimH inhibitors. | ||
Synthetic mono- and multivalent galabiose derivatives 21a–d (Fig. 8) inhibited bacterial adhesion to the coated chip surfaces in a dose-dependent manner (Table 1). An octavalent galabiose compound 21d was superior to the tetravalent derivative 21c, which in turn was a better inhibitor than the monovalent galabiose derivative 21a. However, the multivalency effect was much more pronounced in the case of the Streptococcus suis adhesion when compared to E. coli PapG. On the other hand, a more significant multivalency effect was observed in the inhibition of the mannose-specific type-1-fimbriated E. coli with similar multivalent mannose molecules.97 It would appear that multivalent inhibitors do not reach multiple E. coli adhesin molecules as effectively as in the case of other bacteria such as S. suis (Table 1), and therefore the spacing of the binding sites in the adhesins may differ.
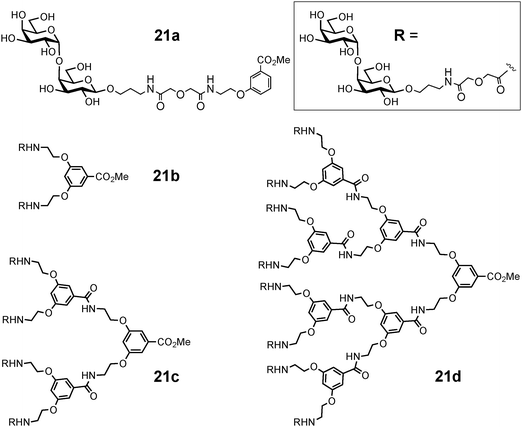 | ||
| Fig. 8 Oligovalent galabiose derivatives. | ||
4.2 Lung pathogens
LecA (also called PA-IL) is a tetrameric cytotoxic lectin consisting of four subunits of 121 amino acids (12.75 kDa)98,102 with specificity for α-D-galactose and binding preferentially to Galα(1-4)Gal containing globotriaosylceramide Gb3 sphingolipid. The LecA crystal structure demonstrated the structural basis of the affinity for galactose monosaccharides with the participation of a calcium ion in the binding site.103 In addition to its cytotoxicity, it has been suggested that this lectin contributes to the formation of bacterial microcolonies and the formation of biofilms.104
Many of multivalent glycoconjugates have been synthesized for inhibiting the binding of LecA to galactosylated surfaces (Table 2 and Fig. 9). High-valency compounds such as galactosylated helical poly(phenylacetylene) polymer 29,34 fullerenes 23,51 glyconanoparticles 31,105 or glycodendrimers 32106 are efficient ligands for inhibition, but their aggregative properties and the strong resulting precipitation create difficulties for measuring affinity constants. Excellent results were obtained with calix[4]arenes 28,38,107 calix[6]arenes 26, β-peptoids 25, porphyrins 27108 and resorcin[4]arenes 22.109 Among these molecules, the 1,3-alternate conformer of calix[4]arene demonstrated the most efficient and dramatic increase in affinity. A chelate-binding mode with two galactose residues interacting with two neighbouring binding sites in a single LecA tetramer could be confirmed by the observation of well-defined nanometric fibers of lectin–glycocluster complexes through atomic force microscopy (AFM) study.110
| Comp. | Valency | HIA (MIC) | ELLA (IC50) | SPR (Kd) | ITC (Kd) | Ref. |
|---|---|---|---|---|---|---|
| a β value calculated with galactose as reference. | ||||||
| αMeGal | 1 | 150 μM | 50 μM | 106 | ||
| βMeGal | 1 | 190 μM | 94 μM | 106 | ||
| 22 | 4 | Haemolysis | 0.7 μM, β = 315 | Not soluble | —/— | 109 |
| 23 | 12 | 0.78 μM, β = 12820 | 0.040 μM, β = 458 | 0.367 μM, β = 173 | —/— | 51 |
| 24 | 4 | > 2000 μM | —/— | 3.5 μM, β = 18 | 1.8 μM, β = 83 | 108 |
| 25 | 4 | > 2000 μM | —/— | 2.5 μM, β = 25 | 0.3 μM, β = 500 | 108 |
| 26 | 6 | 63 μM, β = 159 | —/— | 0.8 μM, β = 80 | 0.14 μM, β = 1071 | 108 |
| 27 | 4 | 63 μM, β = 159 | —/— | 1.4 μM, β = 45 | 0.33 μM, β = 454 | 108 |
| 28 | 4 | 500 μM, β = 20 | —/— | 0.5 μM, β = 144 | 0.176 μM, β = 852 | 38 |
| 29 | Polymer | 9 μM | —/— | —/— | 4.12 μM | 34 |
| 30 | 2 | —/— | 0.22 μM, β = 545 | —/— | —/— | 111 |
| 31 | 67 | 0.45 μM, β = 100 | —/— | 33 μM | 0.05 μM, β = 2824 | 105 |
| 32 | 9 | —/— | —/— | —/— | 230 nM, β = 409 | 106 |
| 33 | 4 | 0.78 μM, β = 4000a | —/— | —/— | 0.1 μM, β = 875 | 112 |
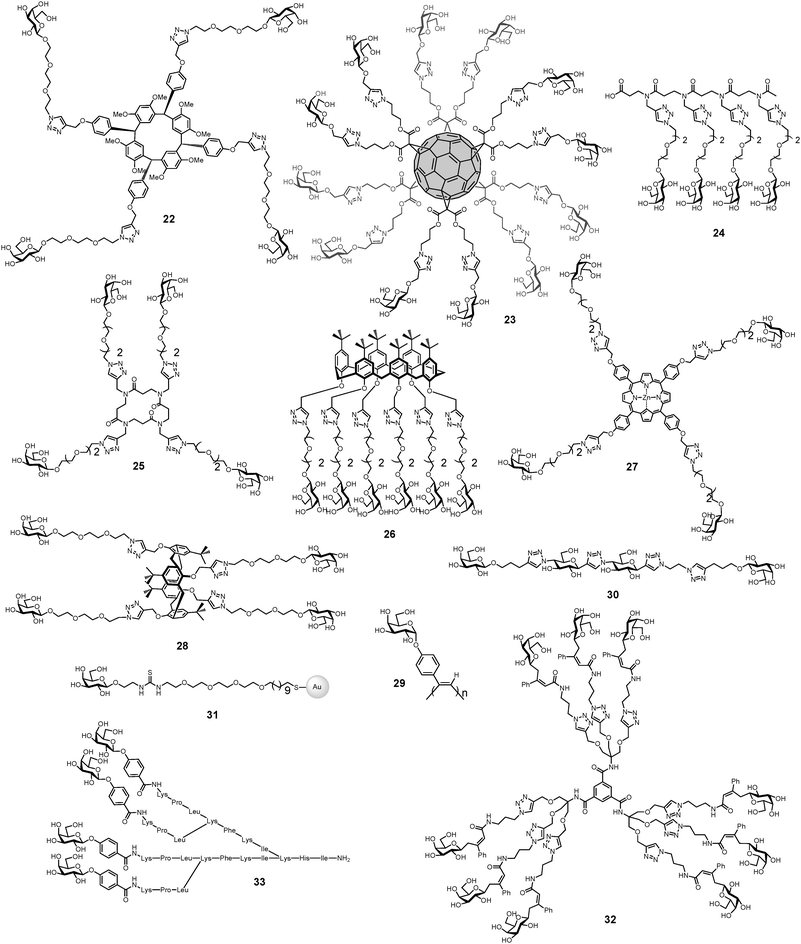 | ||
| Fig. 9 Multivalent glycoconjugates 22–33 as LecA high affinity ligands. | ||
A recent study demonstrated that a divalent ligand 30 with an appropriate linker was sufficient to induce a chelation effect with LecA.111 A rigid spacer was designed based on the alternation of glucose moieties linked at the 1 and 4 positions by a 1,2,3-triazole unit via “click” chemistry. The resulting spacer was relatively rigid and straight and was linked to galactose units at both ends. The number of building blocks was varied, as well as the linker between the spacer and galactose ligand. This linker was shown to be of great importance and only compound 30 (Fig. 9) has the appropriate linker-length for achieving an inhibitory potency increase of 545-fold over a relevant reference compound.
Potent ligands for lectin LecA have been also obtained by synthesis of glycopeptide dendrimers GalAG2 33 and GalBG2 34 (Fig. 10).112 Multivalency strongly influenced binding, with the monovalent and divalent analogs showing much weaker interactions with the lectin. The much stronger binding of the phenyl galactosides to LecA compared to the thiopropyl-galactosides was explained by crystallographic analysis of the lectin–glycopeptide complexes, which revealed a specific interaction between a histidine residue on the lectin and the phenyl group in the ligands, while the thiopropyl side-chain was more disordered.112
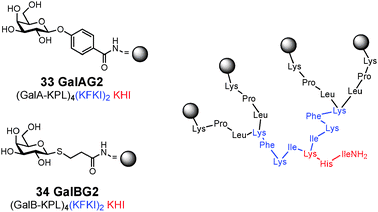 | ||
| Fig. 10 Structure of glycopeptide dendrimer inhibitors of P. aeruginosa biofilms. | ||
LecB (also called PA-IIL) is a tetramer consisting of four 11.73 kDa subunits with high specificity for L-fucose and a weaker one for D-mannose.98,102 The LecB crystal structure revealed the occurrence of two bridging calcium ions in the binding site. This unique mode of binding is not observed in other lectins,113 but explains the very high affinity for fucosides and Lewis a. Although most of the LecB is cytoplasmic, it could also be detected in the outer membrane, including on the surface of biofilm cells, from which it can be released by application of L-fucose.114 It has recently been hypothesized that LecB undergoes transient N-glycosylation that could play a role in the secretion mechanism.115 The search for a putative binding partner led to the proposal of outer membrane protein OprF which is a nonspecific, weakly cation-selective porin channel protein. LecB may mediate the adhesion of P. aeruginosa cells to receptors that are located on its surface and facilitate biofilm formation, thereby promoting colonization of host tissues.
The search for high affinity ligands for LecB initiated the synthesis of several classes of fucose-containing compounds (Table 3 and Fig. 11) based on calixarene 35,116 pentaerythritol 39117 or peptide dendrimer 36–38 scaffolds. Compounds have also been designed for bivalent presentation of αFuc(1 → 4)GlcNAc 40,118 and N-fucosyl amides 41.119
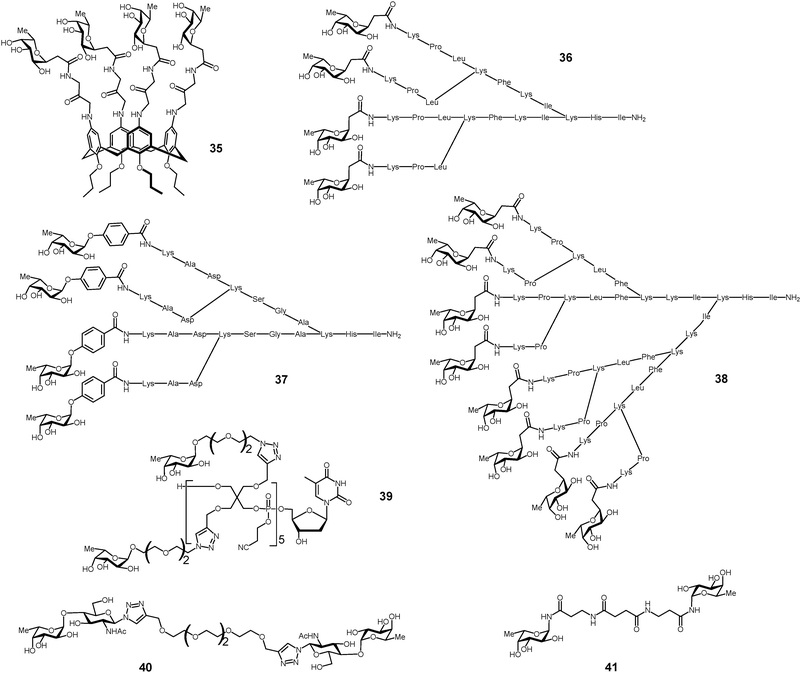 | ||
| Fig. 11 Multivalent glycoconjugates 35–41 as LecB high affinity ligands. | ||
Glycopeptide dendrimer ligands for LecB were identified by screening combinatorial libraries of peptide dendrimers120–122 functionalized with N-terminal C-fucoside residues at the end of the dendrimer branches. FD2 36 and PA8 37 (Fig. 11) turned out to be potent ligands for LecB.123,124
Structure–activity relationship studies showed that multivalency was important for activity, in particular divalent and linear peptide analogs of the dendrimers showed strongly reduced binding at the level of monosaccharides (Table 4). These studies led to the identification of dendrimer 2G3 38 with 8 fucosyl endgroups as the most potent glycopeptide dendrimer ligand to LecB. The diastereoisomer D-36 prepared from D-amino acids was also demonstrated to be a similarly potent ligand to LecB.125
| No | Structurea | Lectin | IC50, μM (ELLA)b | K D, μM (ITC)c | r.p./nd | Biofilm inhibitione |
|---|---|---|---|---|---|---|
| a Standard peptide notation with N-terminus at left and C-terminus at right. Amino acids are given in one-letter codes, italics indicate branching diaminoacids, B is L-2,3-diaminopropionic acid, the C-terminus (at right) is carboxamide (CONH2) in all cases. See also Fig. 10 for exemplification of the topology and the structure of the glycoside groups C-Fuc, GalA and GalB. b Enzyme-linked lectin assay. c Isothermal titration calorimetry. d r.p./n is the relative potency compared to the free sugar (L-fucose or D-galactose) per glycoside group. e Biofilm inhibition measured with the steel coupon assay at 50 μM. n.d. = not determined, — = no inhibition, + = less than 20% inhibition, ++ = 30 to 50% inhibition, +++ = up to 90% inhibition, ++++ = 100% inhibition. | ||||||
| α-NPF | α-(p-Nitrophenyl)-L-fucoside | LecB | 5.27 ± 0.55 | 2.1 | — | |
| T1 | (cFuc-RL)2BRIFV | LecB | 5 ± 0.45 | 1.7 | n.d. | |
| KT1 37 | (cFuc)4(KRL)2BRIFV | LecB | 0.59 ± 0.059 | 7.2 | n.d. | |
| 2G0 | cFuc-KPL | LecB | 5.94 ± 1.24 | 1.9 | n.d. | |
| 2G1 | (cFuc-KPL)2KFKI | LecB | 2.7 ± 0.56 | 2.0 | n.d. | |
| 37 | (cFuc-KPL)4(KFKI)2KHI | LecB | 0.14 ± 0.035 | 20 | ++++ | |
| Dd-36 | (cFuc-kpl)4(Kfkl)2Khl | LecB | 0.66 ± 0.12 | 4.2 | +++ | |
| 2G3 | (cFuc-KP)8(KLF)4(KKI)2KHI | LecB | 0.025 ± 0.005 | 55 | n.d. | |
| NPG | p-Nitrophenyl β-D-galactopyranoside | LecA | 14.1 ± 0.2 | 6.2 | — | |
| GalAG0 | GalA-KPL | LecA | 4.2 ± 0.9 | 21 | + | |
| GalAG1 | (GalA-KPL)2KFKI | LecA | 0.5 ± 0.2 | 91 | ++ | |
| GalAG2 33 | (GalA-KPL)4(KFKI)2KHI | LecA | 0.1 ± 0.01 | 220 | +++ | |
| GalBG0 | GalB-KPL | LecA | 51.5 ± 6.7 | 1.7 | + | |
| GalBG1 | (GalB-KPL)2KFKI | LecA | 2.1 ± 1.0 | 21 | ++ | |
| GalBG2 34 | (GalB-KPL)4(KFKI)2KHI | LecA | 0.4 ± 0.1 | 60 | +++ | |
BC2L-A has a strong affinity for α-D-mannosides (Kd of 2 μM for methyl α-D-mannopyranoside) and mannobioses. Bridging interaction with the branched trimannoside Manα1-3(Manα1-6)Man resulted in the formation of molecular strings as detected by protein crystallography and AFM. Oligomannose analogs presenting two mannosides separated by either rigid (42) or flexible (43) spacer arms were also tested (Fig. 12). Only the rigid linker yielded high affinity with a fast kinetics of clustering, while the flexible analog and the trimannoside displayed moderate affinities and no clustering.128
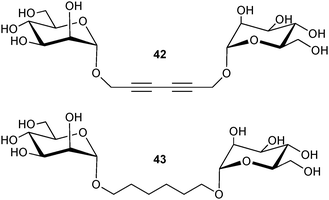 | ||
| Fig. 12 Divalent mannosylated compounds as ligands of BC2L. | ||
Micelles formed from mannosylated poly(ethylene oxide)-b-poly(ε-caprolactone) (PEO-b-PCL) diblock copolymer and nanoparticles of poly(D,L-lactic acid) functionalized with the same copolymer have also been demonstrated to bind efficiently to BC2L-A.36,129
4.3 Zoonotic bacteria
Streptococcus suis is an important emerging worldwide pig pathogen and zoonotic agent, which causes meningitis, pneumonia and sepsis in pigs and also meningitis in humans.130 An adhesion activity based on the recognition of glycoconjugates containing the disaccharide galabiose (i.e. Galα(1-4)Gal) has been characterized in S. suis. The hydroxyl groups identified to be involved in the interaction indicate that S. suis adhesin has a different binding mode to galabiose than PapG from uropathogenic E. coli. These two adhesins do not display sequence similarity neither, indicating that they have evolved through convergent evolution.131Synthetic derivatives of the receptor disaccharide galabiose on one hand, or polyvalent dendrimers of galabiose on the other hand (Fig. 8), have turned out to be exceptionally efficient inhibitors of S. suis adhesion, both at nanomolar concentrations.132,133 Inhibitors of the adhesion of S. suis to cells have also been found in natural sources, berries and juices, but their chemical nature has not yet been identified.134 In the characterization of the adhesion specificities and comparison of various mono- or oligovalent inhibitors, a live-bacteria application of surface plasmon resonance has turned out to be very useful.96
Due to phase variation, the expression of bacterial adhesins is not uniform even within a single bacterial strain. Therefore it may become important to be able to detect specifically bacteria that express a specific adhesin of potential clinical impact. Magnetic glyconanoparticles may represent ideal tools for this purpose. Magnetic beads containing derivatives of galabiose (Fig. 13) were capable of selecting bacteria in a mixture and determining their amounts in a luminescence assay.135 Optimisation of the carbohydrate ligand and its multivalent presentation in appropriate carriers are predicted to further improve the efficiency of the ligand in bacterial adhesion inhibition and detection.
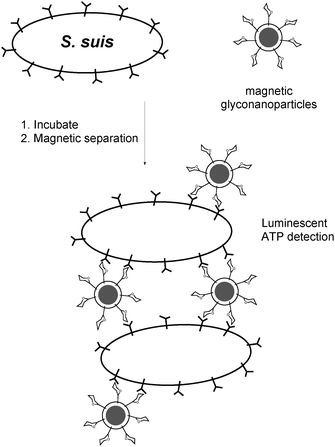 | ||
| Fig. 13 Use of galabiose-functionalized magnetic beads for identifying and isolating S. suis bacteria. | ||
5. Bacterial cell surface as a target for anti-infectious agents
5.1 Bacterial LPS: a natural multivalent glycoconjugate
In the cell wall of Gram-negative bacteria, carbohyrates are mostly conjugated to lipids in the outer membrane, where they are present in a very high concentration. The bacterial outer membrane is asymmetric.136 Its external leaflet is almost entirely constituted of lipopolysaccharides (LPSs) which cover approximately 75% of the surface giving rise to a multivalent glycoconjugate surface with very low fluidity and a highly ordered structure. LPSs are necessary for bacterial viability and this natural multivalent assembly plays many roles in host–bacterium interactions.136 LPSs are classified as MAMPs (Microbe-Associated Molecular Patterns) because they are recognized by the innate immune system. This recognition takes place through the involvement of specific PRRs (Pathogen Related Receptors) finely tuned on a MAMP motif, the lipid A domain.LPSs isolated or extracted from their natural environment aggregate in solution due to their amphiphilic nature, resulting in a supramolecular aggregate which still displays multivalent features that can be of direct use. Indeed, it has recently been shown that the HIV protective antibody 2G12 is able to recognize the LPS produced from Rhizobium radiobacter Rv3.137 This particular LPS possesses a carbohydrate structure (Fig. 14) which resembles the epitope on the surface of HIV for which the antibody is specific. It has been observed that 2G12 interacted with lipooligosaccharides (LOS) only when the lipid moiety was present, so as to allow formation of a supramolecular aggregate in water (C. De Castro, personal communication).
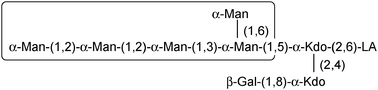 | ||
| Fig. 14 Structure of the LPS from Rhizobium radiobacter Rv3. The area in the box displays strong structural similarity with the epitope recognized from the mAb 2G12. LA stands for lipid A. | ||
Another interesting example of LPS multivalency relates to the stabilizing effect on the protein conformation embedded in the membrane. This concept guided the formulation of the new generation vaccine for Neisseria meningitidis group B (MenB),138 which is the causative agent of meningitis. The strain belonging to group B is the only member within its species for which the synthesis of a synthetic glycoprotein vaccine has not been successful so far as its capsular polysaccharide is not immunogenic. The current MenB vaccine is a vesicle prepared from fragments of the bacterial membrane; the antigenicity mainly arises from a selected pool of Neisseria proteins embedded in this artificial membrane, in which LPSs are thought to work as a multivalent glycoconjugate that stabilises the vesicle.
Given the high biological importance of LPSs as natural multivalent glycoconjugates in the elicitation/suppression of eukaryotic immunity, the structural and supramolecular study of such molecules plays an important role. In an attempt to create a “non-natural” LPS multivalent surface, these molecules have been extracted from different microbial sources and reconstituted in liposomes. The physico-chemical investigation of these systems has been performed by a combined experimental strategy, which has allowed characterisation at different observation scales, from the morphological to the micro-structural level.139,140 The next step will be the study of the elicitation of eukaryotic immunity of such an “artificial” bacterial surface.
5.2 Inhibition of LPS synthesis by multivalent glycoconjugates
L-Heptosides (L-glycero-D-manno-heptopyranoses) are components of the core region of LPS. Since LPS is essential for bacterial viability, its biosynthesis can be targeted for the development of novel antibacterial agents. A series of fullerene hexakis-adducts bearing twelve copies of peripheral sugars displaying the mannopyranose core structure of bacterial L-heptosides have been synthesized (Fig. 15). The multimers were assembled through an efficient CuAAC reaction as the final step. The final fullerene sugar balls were assayed as inhibitors of heptosyltransferase WaaC, the glycosyltransferase catalyzing the incorporation of the first L-heptose into LPS. Interestingly, the inhibition of the final molecules was found in the low micromolar range (IC50 value of 7 to 45 μM) while the corresponding monomeric glycosides displayed high micromolar to low millimolar inhibition levels (IC50 always above 400 μM). When evaluated on a “per-sugar” basis, these inhibition data showed that, in each case, the average affinity of a single glycoside of the fullerenes towards WaaC was significantly enhanced when displayed as a multimer, thus demonstrating an unexpected multivalent effect.141 To date, such a multivalent mode of inhibition had never been evidenced with glycosyltransferases. | ||
| Fig. 15 Multivalent glycosylated fullerenes for inhibition of LPS heptosyltransferase WaaC. | ||
5.3 Exploitation of bacterial S-layers as a natural multivalent glycan display system
Natural molecular self-assembly systems are prime candidates for use in nanobiotechnology. Crystalline-cell surface (S-) layers of prokaryotic organisms are very potent self-assembly systems, which can be used in bottom-up processes as a patterning element for the multivalent display of biofunctional epitopes such as glycans.142The S-layer system is being exploited for multivalent glycan display based on the groundbreaking demonstrations that proteins can be recombinantly equipped with tailor-made glycosylation in an easily tractable bacterial system such as Escherichia coli143 and that glycosylation modules from different bacterial sources, including glycoproteins, lipopolysaccharides or exopolysaccharides, can be combined to achieve functional glycosylation.144 Using a combination of protein- and glycosylation-engineering approaches to produce self-assembling S-layer neoglycoconjugates, the feasibility of this system could be proven in vitro as well as in vivo, with the latter approach presenting interesting possibilities for live glycoconjugate delivery in future antipathogenic therapy.145
In a proof-of-concept study, the S-layer protein SgsE from Geobacillus stearothermophilus NRS 2004/3a (AF328862) was used as a matrix for the display of a branched heptasaccharide from the Campylobacter jejuni protein AcrA as well as for the E. coli O7 antigen.146 SgsE is a 903-amino acid protein which aligns in a 2D lattice with oblique (p2) symmetry and which is naturally O-glycosylated at multiple sites. The SgsE protein was engineered by including the signal peptide of PelB (pectate lyase from Erwinia carotovora) for periplasmic targeting. Furthermore, one of the natural protein O-glycosylation sites was engineered into an N-glycosylation site to be recognized by the heterologous oligosaccharyltransferase PglB. In this way, S-layer neo-glycoproteins could be produced based on plasmid-encoded glycosylation information for either of the model glycan structures. The degree of glycosylation of the S-layer neoglycoproteins after purification from the periplasmic fraction of the E. coli cell factory reached up to 100%. Electron microscopy revealed that recombinant glycosylation is fully compatible with the S-layer protein self-assembly system (Fig. 16). Thus, the S-layer system is a promising strategy for multivalent glycan display approaches, where strict (“nanometer-scale”) control over position and orientation of the glycan epitopes is desired.
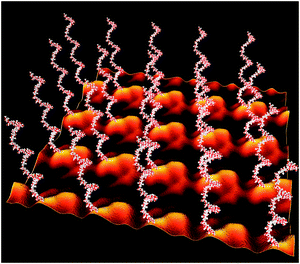 | ||
| Fig. 16 Model of a self-assembled SgsE-neoglycoprotein monolayer periodically displaying recombinant E. coli O7 antigens with nanometer-scale precision. Image reconstruction using Cinema 4 is based on a negatively stained preparation of the S-layer protein self-assembled in solution and on the pdb data of the glycans generated with Sweet at http://www.glycosciences.de/ (adapted from ref. 146, Wiley-VCH Weinheim). | ||
6. Glycoconjugate analogs and multivalent glycoconjugates for immune system stimulation and vaccine approach
6.1 Glycoconjugate analogs to trigger the innate immunity
Early recognition of invading bacteria by the innate immune system has a crucial function in antibacterial defense by triggering inflammatory responses that prevent the spread of infection and suppress bacterial growth. Toll-like receptor 4 (TLR4), the innate immunity receptor of bacterial endotoxins, plays a pivotal role in the induction of inflammatory responses. TLR4 activation by bacterial lipopolysaccharide (LPS) is achieved by the coordinate and sequential action of three other proteins, LBP, CD14 and MD-2 receptors, that bind lipopolysaccharide (LPS) and present it to TLR4 by forming the activated (TLR4-MD-2-LPS)2 complex (Fig. 17).147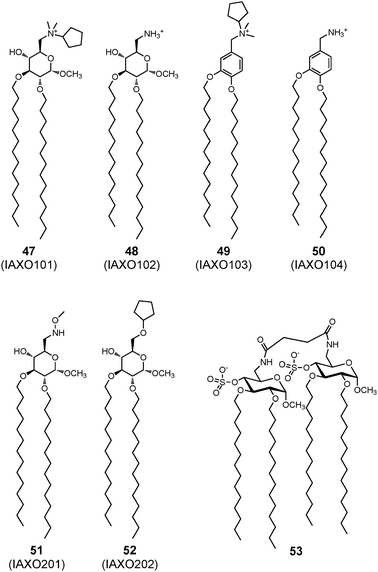 | ||
| Fig. 17 Synthetic lipids with TLR4-modulating activity. | ||
TLR4 trigger can be remarkably sensitive and robust, stimulating prompt and powerful host defence responses to different species of invading bacteria. However, an excessively potent host response generates life-threatening syndromes such as acute sepsis and septic shock. Non-toxic LPSs or lipid A obtained from non-pathogenic bacteria such as Rhodobacter capsulatus and Rhodobacter sphaeroides are potent LPS antagonists in vitro148 although no molecules usable for pharmacological treatment for sepsis are not available yet.
Several synthetic molecules capable of modulating TLR4 activity have been developed.149,150 IAXO compounds 47–52, which include lipidated monosaccharides with an amino group on the C-6 position of the pyranose ring of D-glucose, were active in blocking the TLR4 signal in cells and in vivo models (Fig. 17).151 Their antagonist effect was due to the capacity of these molecules to displace from CD14 and TLR4-MD-2 complexes.152 These molecules are now commercially available with the proprietary name of IAXO (Innaxon) as selective small-molecule inhibitors of the TLR4 signal pathway, and lead compound IAXO-102 48 is in a preclinical phase as an antisepsis agent. More recently, lipid A analogues such as 53 were developed with a structure composed of two glucoside units connected by a linker both units bearing on C-4 an anionic sulfate group (Fig. 17).153 These compounds have an antagonist effect if administered together with endotoxin, and a mild agonist effect if administered alone. In the context of vaccine adjuvants acting on innate immunity receptors, natural LPS immobilized on nanoparticles was also demonstrated to have interesting TLR4 activating properties.154
6.2 Multivalent glycoconjugates in vaccine development
Together with new studies on the molecular basis of the interactions between glycans and anti-HIV antibodies, gold nanoparticles functionalized with oligomannosides (manno-GNPs, Fig. 2) can offer an alternative in this direction. In order to gain deeper insights into the interactions between 2G12 and selected oligomannosides at the molecular level, the structural and affinity details of the 2G12/oligomannosides interactions have been studied by saturation transfer difference NMR spectroscopy (STD-NMR) and transferred NOE in isotropic solution.163 It was found that linear oligomannosides show a single binding mode to 2G12, with the non-reducing terminal disaccharide Man(α1-2)Man(α1-, making the closest antibody/oligosaccharide contacts in the bound state. In contrast, a branched pentamannoside showed two alternate binding modes involving both ligand arms, contrary to previous X-ray studies.164 Among the analysed series of ligands, the strongest 2G12 binders were the linear tri- and tetramannosides. This information is of key importance for the design of synthetic multivalent gp120 high-mannose mimics for HIV vaccine development. Indeed manno-GNPs were able to bind with high affinity and to interfere with the 2G12/gp120 binding as determined by SPR-based biosensors and STD-NMR.165 Cellular neutralization assays with manno-GNPs also demonstrated that GNPs coated with a linear tetramannoside could block the 2G12-mediated neutralization of a replication-competent HIV-1 under conditions that resemble those in which normal serum prevents infection of the target cell. All these results prove that selected manno-GNPs could function as an anti-adhesive barrier at an early stage of HIV infection, but also as synthetic mimics of the 2G12 epitope in the route of a carbohydrate-based vaccine against HIV.
![Gold nanoparticles bearing Galβ(1-4)Glcβ(1-6)[Galβ(1-4)]GlcNAcβ(1- (repeating units of S. pneumoniae type 14 capsular polysaccharide), T-helper ovalbumin (OVA) peptide OVA323–239 and d-glucose as carriers for synthetic carbohydrate-based vaccines.](/image/article/2013/CS/c2cs35408j/c2cs35408j-f18.gif) | ||
| Fig. 18 Gold nanoparticles bearing Galβ(1-4)Glcβ(1-6)[Galβ(1-4)]GlcNAcβ(1- (repeating units of S. pneumoniae type 14 capsular polysaccharide), T-helper ovalbumin (OVA) peptide OVA323–239 and D-glucose as carriers for synthetic carbohydrate-based vaccines. | ||
7. Conclusions
The recent advances in the synthesis of complex glycomolecules with controlled topology and valency coupled with the progress in understanding the importance of protein–carbohydrate interactions have driven forward the domain of multivalent glycomolecules. Indeed, these molecules have proven to be very potent frameworks for binding to a range of glycan receptors, and therefore have the ability to stimulate, mediate or inhibit a variety of biological or pathological processes. The different applications of glycocompounds in anti-infectious strategies should be further developed in the future, with knowledge-based approaches that take into account the structure of the protein receptors. Designing multivalent glycoconjugates that perfectly fit the topology of binding sites will increase their affinity toward the target, and help in fine-tuning their specificity. Based on the reports summarized in this review, it appears that multivalent glycoconjugates are close to be utilised in anti-adhesive therapies against viral and bacterial infection, but can are be used to mimic bacterial cell surfaces in order to stimulate the immune systems.Acknowledgements
The authors acknowledge the support from EU COST Actions CM1102 and COST BM1003. In addition, the work reviewed has been supported by the sources listed in the original publications cited and by institutions mentioned in the affiliation list.References
- A. Varki, R. D. Cummings, J. D. Esko, H. H. Freeze, P. Stanley, C. R. Bertozzi, G. W. Hart and M. E. Etzler, Essentials of Glycobiology, Cold Spring Harbour, New York, 2nd edn, 2009 Search PubMed.
- H. J. Gabius, S. Andre, J. Jimenez-Barbero, A. Romero and D. Solis, Trends Biochem. Sci., 2012, 36, 298–313 CrossRef.
- H. Lis and N. Sharon, Chem. Rev., 1998, 98, 637–674 CrossRef CAS.
- L. L. Kiessling, J. E. Gestwicki and L. E. Strong, Curr. Opin. Chem. Biol., 2000, 4, 696–703 CrossRef CAS.
- Y. C. Lee and R. T. Lee, Acc. Chem. Res., 1995, 28, 321–327 CrossRef CAS.
- J. J. Lundquist and E. J. Toone, Chem. Rev., 2002, 102, 555–578 CrossRef CAS.
- M. Mammen, S.-K. Choi and G. M. Whitesides, Angew. Chem., Int. Ed., 1998, 37, 2754–2794 CrossRef.
- V. I. Dyukova, N. V. Shilova, O. E. Galanina, A. Y. Rubina and N. V. Bovin, Biochim. Biophys. Acta, 2006, 1760, 603–609 CrossRef CAS.
- T. Feizi, F. Fazio, W. Chai and C. H. Wong, Curr. Opin. Struct. Biol., 2003, 13, 637–645 CrossRef CAS.
- T. Horlacher and P. H. Seeberger, Chem. Soc. Rev., 2008, 37, 1414–1422 RSC.
- O. Oyelaran and J. C. Gildersleeve, Curr. Opin. Chem. Biol., 2009, 13, 406–413 CrossRef CAS.
- J. C. Paulson, O. Blixt and B. E. Collins, Nat. Chem. Biol., 2006, 2, 238–248 CrossRef CAS.
- E. H. Song and N. L. Pohl, Curr. Opin. Chem. Biol., 2009, 13, 626–632 CrossRef CAS.
- C. D. Rillahan and J. C. Paulson, Annu. Rev. Biochem., 2011, 80, 797–823 CrossRef CAS.
- T. K. Dam and C. F. Brewer, Adv. Carbohydr. Chem. Biochem., 2010, 63, 139–164 CrossRef CAS.
- T. K. Dam, T. A. Gerken and C. F. Brewer, Biochemistry, 2009, 48, 3822–3827 CrossRef CAS.
- J. E. Gestwicki, C. W. Cairo, L. E. Strong, K. A. Oetjen and L. L. Kiessling, J. Am. Chem. Soc., 2002, 124, 14922–14933 CrossRef CAS.
- M. Reynolds and S. Perez, C. R. Chim., 2011, 14, 74–95 CrossRef CAS.
- Y. M. Chabre and R. Roy, Adv. Carbohydr. Chem. Biochem., 2010, 63, 165–393 CrossRef CAS.
- D. Deniaud, K. Julienne and S. G. Gouin, Org. Biomol. Chem., 2011, 9, 966–979 CAS.
- R. J. Pieters, Org. Biomol. Chem., 2009, 7, 2013–2025 CAS.
- T. Darbre and J. L. Reymond, Curr. Top. Med. Chem., 2008, 8, 1286–1293 CrossRef CAS.
- K. J. Doores, D. P. Gamblin and B. G. Davis, Chem.–Eur. J., 2006, 12, 656–665 CrossRef CAS.
- A. Imberty, Y. M. Chabre and R. Roy, Chem.–Eur. J., 2008, 14, 7490–7499 CrossRef CAS.
- P. I. Kitov, J. M. Sadowska, G. Mulvey, G. D. Armstrong, H. Ling, N. S. Pannu, R. J. Read and D. R. Bundle, Nature, 2000, 403, 669–672 CrossRef CAS.
- A. Imberty and A. Varrot, Curr. Opin. Struct. Biol., 2008, 18, 567–576 CrossRef CAS.
- K. A. Karlsson, Adv. Exp. Med. Biol., 2001, 491, 431–443 CrossRef CAS.
- R. J. Pieters, Adv. Exp. Med. Biol., 2011, 715, 227–240 CrossRef CAS.
- N. Sharon, Adv. Exp. Med. Biol., 1996, 408, 1–8 CrossRef CAS.
- B. G. Davis, Chem. Rev., 2002, 102, 579–602 CrossRef CAS.
- R. J. Payne and C. H. Wong, Chem. Commun., 2010, 46, 21–43 RSC.
- P. M. Rendle, A. Seger, J. Rodrigues, N. J. Oldham, R. R. Bott, J. B. Jones, M. M. Cowan and B. G. Davis, J. Am. Chem. Soc., 2004, 126, 4750–4751 CrossRef CAS.
- L. Bes, S. Angot, A. Limer and D. M. Haddleton, Macromolecules, 2003, 36, 2493–2499 CrossRef CAS.
- I. Otsuka, B. Blanchard, R. Borsali, A. Imberty and T. Kakuchi, ChemBioChem, 2010, 11, 2399–2408 CrossRef CAS.
- D. Ponader, F. Wojcik, F. Beceren-Braun, J. Dernedde and L. Hartmann, Biomacromolecules, 2012, 13, 1845–1852 CrossRef CAS.
- J. Rieger, F. Stoffelbach, D. Cui, A. Imberty, E. Lameignere, J. L. Putaux, R. Jérôme, C. Jérôme and R. Auzély-Velty, Biomacromolecules, 2007, 8, 2717–2725 CrossRef CAS.
- L. Baldini, A. Casnati, F. Sansone and R. Ungaro, Chem. Soc. Rev., 2007, 36, 254–266 RSC.
- S. Cecioni, R. Lalor, B. Blanchard, J.-P. Praly, A. Imberty, S. E. Matthews and S. Vidal, Chem.–Eur. J., 2009, 15, 13232–13240 CrossRef CAS.
- A. Dondoni and A. Marra, Chem. Rev., 2010, 110, 4949–4977 CrossRef CAS.
- S. Andre, C. Grandjean, F. M. Gautier, S. Bernardi, F. Sansone, H. J. Gabius and R. Ungaro, Chem. Commun., 2011, 47, 6126–6128 RSC.
- S. Andre, R. J. Pieters, I. Vrasidas, H. Kaltner, I. Kuwabara, F. T. Liu, R. M. Liskamp and H. J. Gabius, ChemBioChem, 2001, 2, 822–830 CrossRef CAS.
- C. Heidecke and T. K. Lindhorst, Chem.–Eur. J., 2007, 13, 9056–9067 CrossRef CAS.
- M. A. Mintzer, E. L. Dane, G. A. O'Toole and M. W. Grinstaff, Mol. Pharm., 2012, 9, 342–354 CrossRef CAS.
- M. Touaibia and R. Roy, Mini–Rev. Med. Chem., 2007, 7, 1270–1283 CrossRef CAS.
- W. B. Turnbull and J. F. Stoddart, J. Biotechnol., 2002, 90, 231–255 CAS.
- D. A. Fulton and J. F. Stoddart, Bioconjugate Chem., 2001, 12, 655–672 CrossRef CAS.
- M. Gomez-Garcia, J. M. Benito, A. P. Butera, C. Ortiz Mellet, J. M. Garcia Fernandez and J. L. Jimenez Blanco, J. Org. Chem., 2012, 77, 1273–1288 CrossRef CAS.
- I. J. Krauss, J. G. Joyce, A. C. Finnefrock, H. C. Song, V. Y. Dudkin, X. Geng, J. D. Warren, M. Chastain, J. W. Shiver and S. J. Danishefsky, J. Am. Chem. Soc., 2007, 129, 11042–11044 CrossRef CAS.
- T. Ohta, N. Miura, N. Fujitani, F. Nakajima, K. Niikura, R. Sadamoto, C. T. Guo, T. Suzuki, Y. Suzuki, K. Monde and S. Nishimura, Angew. Chem., Int. Ed., 2003, 42, 5186–5189 CrossRef CAS.
- A. M. Pujol, M. Cuillel, O. Renaudet, C. Lebrun, P. Charbonnier, D. Cassio, C. Gateau, P. Dumy, E. Mintz and P. Delangle, J. Am. Chem. Soc., 2011, 133, 286–296 CrossRef CAS.
- S. Cecioni, V. Oerthel, J. Iehl, M. Holler, D. Goyard, J.-P. Praly, A. Imberty, J.-F. Nierengarten and S. Vidal, Chem.–Eur. J., 2011, 17, 3252–3261 CrossRef CAS.
- J.-F. Nierengarten, J. Iehl, V. Oerthel, M. Holler, B. M. Illescas, A. Munoz, N. Martin, J. Rojo, M. Sanchez-Navarro, S. Cecioni, S. Vidal, K. Buffet, M. Durka and S. P. Vincent, Chem. Commun., 2010, 46, 3860–3862 RSC.
- M. Marradi, M. Martin-Lomas and S. Penades, Adv. Carbohydr. Chem. Biochem., 2010, 64, 211–290 CrossRef CAS.
- X. Wang, E. Matei, L. Deng, O. Ramstrom, A. M. Gronenborn and M. Yan, Chem. Commun., 2011, 47, 8620–8622 RSC.
- R. Kikkeri, P. Laurino, A. Odedra and P. H. Seeberger, Angew. Chem., Int. Ed., 2010, 49, 2054–2057 CrossRef CAS.
- A. Robinson, J. M. Fang, P. T. Chou, K. W. Liao, R. M. Chu and S. J. Lee, ChemBioChem, 2005, 6, 1899–1905 CrossRef CAS.
- Y. Yang, M. Yu, T. T. Yan, Z. H. Zhao, Y. L. Sha and Z. J. Li, Bioorg. Med. Chem., 2010, 18, 5234–5240 CrossRef CAS.
- R. D. Astronomo and D. R. Burton, Nat. Rev. Drug Discovery, 2010, 9, 308–324 CrossRef CAS.
- V. Verez-Bencomo, V. Fernandez-Santana, E. Hardy, M. E. Toledo, M. C. Rodriguez, L. Heynngnezz, A. Rodriguez, A. Baly, L. Herrera, M. Izquierdo, A. Villar, Y. Valdes, K. Cosme, M. L. Deler, M. Montane, E. Garcia, A. Ramos, A. Aguilar, E. Medina, G. Torano, I. Sosa, I. Hernandez, R. Martinez, A. Muzachio, A. Carmenates, L. Costa, F. Cardoso, C. Campa, M. Diaz and R. Roy, Science, 2004, 305, 522–525 CrossRef CAS.
- N. Nagahori, R. T. Lee, S. Nishimura, D. Page, R. Roy and Y. C. Lee, ChemBioChem, 2002, 3, 836–844 CrossRef CAS.
- A. Nelson, J. M. Belitsky, S. Vidal, C. S. Joiner, L. G. Baum and J. F. Stoddart, J. Am. Chem. Soc., 2004, 126, 11914–11922 CrossRef CAS.
- Y. van Kooyk and T. B. Geijtenbeek, Nat. Rev. Immunol., 2003, 3, 697–709 CrossRef CAS.
- M. Sánchez-Navarro and J. Rojo, Drug News Perspect., 2010, 23, 557–572 CrossRef.
- J. Luczkowiak, S. Sattin, I. Sitkeviciute, J. Reina, M. Sánchez-Navarro, M. Thépaut, L. Martínez-Prats, A. Daghetti, F. Fieschi, R. Delgado, A. Bernardi and J. Rojo, Bioconjugate Chem., 2011, 22, 1354–1365 CrossRef CAS.
- A. Berzi, J. Reina, R. Ottria, I. Sutkeviciute, P. Antonazzo, M. Sánchez-Navarro, E. M. Chabrol, M. Biasin, D. Trabattoni, I. Cetin, J. Rojo, F. Fieschi, A. Bernardi and M. Clerici, AIDS, 2012, 26, 127–137 CrossRef CAS.
- S. Sattin, A. Daghetti, M. Thépaut, A. Berzi, M. Sánchez-Navarro, G. Tabarani, J. Rojo, F. Fieschi, M. Clerici and A. Bernardi, ACS Chem. Biol., 2010, 301–312 CrossRef CAS.
- M. Sánchez-Navarro, A. Muñoz, B. Illescas, J. Rojo and N. Martín, Chem.–Eur. J., 2011, 17, 766–769 CrossRef.
- O. Martinez-Avila, K. Hijazi, M. Marradi, C. Clavel, C. Campion, C. Kelly and S. Penades, Chem.–Eur. J., 2009, 15, 9874–9888 CrossRef CAS.
- O. Martinez-Avila, L. M. Bedoya, M. Marradi, C. Clavel, J. Alcami and S. Penades, ChemBioChem, 2009, 10, 1806–1809 CrossRef CAS.
- G. R. Vasta, Nat. Rev. Microbiol., 2009, 7, 424–438 CrossRef CAS.
- M. Ouellet, S. Mercier, I. Pelletier, S. Bounou, J. Roy, J. Hirabayashi, S. Sato and M. J. Tremblay, J. Immunol., 2005, 174, 4120–4126 CAS.
- S. Andre, D. V. Jarikote, D. Yan, L. Vincenz, G. N. Wang, H. Kaltner, P. V. Murphy and H. J. Gabius, Bioorg. Med. Chem. Lett., 2012, 22, 313–318 CrossRef CAS.
- R. Leyden, T. Velasco-Torrijos, S. Andre, S. Gouin, H. J. Gabius and P. V. Murphy, J. Org. Chem., 2009, 74, 9010–9026 CrossRef CAS.
- G. N. Wang, S. Andre, H. J. Gabius and P. V. Murphy, Org. Biomol. Chem., 2012, 10, 6893–6907 CAS.
- E. Fan, E. A. Merritt, C. L. Verlinde and W. G. Hol, Curr. Opin. Struct. Biol., 2000, 10, 680–686 CrossRef CAS.
- Z. Zhang, E. A. Merritt, M. Ahn, C. Roach, Z. Hou, C. L. Verlinde, W. G. Hol and E. Fan, J. Am. Chem. Soc., 2002, 124, 12991–12998 CrossRef CAS.
- A. V. Pukin, C. A. Weijers, B. van Lagen, R. Wechselberger, B. Sun, M. Gilbert, M. F. Karwaski, D. E. Florack, B. C. Jacobs, A. P. Tio-Gillen, A. van Belkum, H. P. Endtz, G. M. Visser and H. Zuilhof, Carbohydr. Res., 2008, 343, 636–650 CrossRef CAS.
- A. V. Pukin, H. M. Branderhorst, C. Sisu, C. A. Weijers, M. Gilbert, R. M. Liskamp, G. M. Visser, H. Zuilhof and R. J. Pieters, ChemBioChem, 2007, 8, 1500–1503 CrossRef CAS.
- A. V. Pukin, B. C. Jacobs, A. P. Tio-Gillen, M. Gilbert, H. P. Endtz, A. van Belkum, G. M. Visser and H. Zuilhof, Glycobiology, 2011, 21, 1642–1650 CrossRef CAS.
- C. Sisu, A. J. Baron, H. M. Branderhorst, S. D. Connell, C. A. Weijers, R. de Vries, E. D. Hayes, A. V. Pukin, M. Gilbert, R. J. Pieters, H. Zuilhof, G. M. Visser and W. B. Turnbull, ChemBioChem, 2009, 10, 329–337 CrossRef CAS.
- H. M. Branderhorst, R. M. Liskamp, G. M. Visser and R. J. Pieters, Chem. Commun., 2007, 5043–5045 RSC.
- A. Bernardi, A. Checchia, P. Brocca, S. Sonnino and F. Zuccotto, J. Am. Chem. Soc., 1999, 121, 2032–2036 CrossRef CAS.
- A. Bernardi and P. Cheshev, Chem.–Eur. J., 2008, 14, 7434–7441 CrossRef CAS.
- D. Arosio, M. Fontanella, L. Baldini, L. Mauri, A. Bernardi, A. Casnati, F. Sansone and R. Ungaro, J. Am. Chem. Soc., 2005, 127, 3660–3661 CrossRef CAS.
- S. D. Knight and J. Bouckaert, Top. Curr. Chem., 2009, 288, 67–107 CrossRef CAS.
- M. Almant, V. Moreau, J. Kovensky, J. Bouckaert and S. G. Gouin, Chem.–Eur. J., 2011, 17, 10029–10038 CrossRef CAS.
- M. Dubber, O. Sperling and T. K. Lindhorst, Org. Biomol. Chem., 2006, 4, 3901–3912 CAS.
- M. Durka, K. Buffet, J. Iehl, M. Holler, J. F. Nierengarten, J. Taganna, J. Bouckaert and S. P. Vincent, Chem. Commun., 2011, 47, 1321–1323 RSC.
- M. Hartmann and T. K. Lindhorst, Eur. J. Org. Chem., 2011, 3583–3609 CrossRef CAS.
- M. Touaibia, A. Wellens, T. C. Shiao, Q. Wang, S. Sirois, J. Bouckaert and R. Roy, ChemMedChem, 2007, 2, 1190–1201 CrossRef CAS.
- M. J. Weissenborn, R. Castangia, J. W. Wehner, R. Sardzik, T. K. Lindhorst and S. L. Flitsch, Chem. Commun., 2012, 48, 4444–4446 RSC.
- M. Hartmann, A. K. Horst, P. Klemm and T. K. Lindhorst, Chem. Commun., 2010, 46, 330–332 RSC.
- M. Hartmann, P. Betz, Y. Sun, S. N. Gorb, T. K. Lindhorst and A. Krueger, Chem.–Eur. J., 2012, 18, 6485–6492 CrossRef CAS.
- J. R. Johnson, Clin. Microbiol. Rev., 1991, 4, 80–128 CAS.
- M. C. Lane and H. L. Mobley, Kidney Int., 2007, 72, 19–25 CrossRef CAS.
- A. Salminen, V. Loimaranta, J. A. Joosten, A. S. Khan, J. Hacker, R. J. Pieters and J. Finne, J. Antimicrob. Chemother., 2007, 60, 495–501 CrossRef CAS.
- C. Appeldoorn, J. Joosten, F. el Maate, U. Dobrindt, J. Hacker, R. Liskamp, A. Khan and R. Pieters, Tetrahedron: Asymmetry, 2005, 16, 361–372 CrossRef CAS.
- N. Gilboa-Garber, Methods Enzymol., 1982, 83, 378–385 CrossRef CAS.
- C. Chemani, A. Imberty, S. de Bentzmann, M. Pierre, M. Wimmerova, B. P. Guery and K. Faure, Infect. Immun., 2009, 77, 2065–2075 CrossRef CAS.
- H. P. Hauber, M. Schulz, A. Pforte, D. Mack, P. Zabel and U. Schumacher, Int. J. Med. Sci., 2008, 5, 371–376 CrossRef CAS.
- H. Gustke, R. Kleene, G. Loers, N. Nehmann, M. Jaehne, K. M. Bartels, K. E. Jaeger, M. Schachner and U. Schumacher, Eur. J. Clin. Microbiol. Infect. Dis., 2012, 31, 207–215 CrossRef CAS.
- A. Imberty, M. Wimmerova, E. P. Mitchell and N. Gilboa-Garber, Microbes Infect., 2004, 6, 222–229 CrossRef.
- G. Cioci, E. P. Mitchell, C. Gautier, M. Wimmerova, D. Sudakevitz, S. Pérez, N. Gilboa-Garber and A. Imberty, FEBS Lett., 2003, 555, 297–301 CrossRef CAS.
- S. P. Diggle, R. E. Stacey, C. Dodd, M. Camara, P. Williams and K. Winzer, Environ. Microbiol., 2006, 8, 1095–1104 CrossRef CAS.
- M. Reynolds, M. Marradi, A. Imberty, S. Penadés and S. Pérez, Chem.–Eur. J., 2012, 18, 4264–4273 CrossRef CAS.
- Y. M. Chabre, D. Giguère, B. Blanchard, J. Rodrigue, S. Rocheleau, M. Neault, S. Rauthu, A. Papadopoulos, A. A. Arnold, A. Imberty and R. Roy, Chem.–Eur. J., 2011, 17, 6545–6562 CrossRef CAS.
- S. Cecioni, J. P. Praly, S. E. Matthews, M. Wimmerová, A. Imberty and S. Vidal, Chem.–Eur. J., 2012, 18, 6250–6263 CrossRef CAS.
- S. Cecioni, S. Faure, U. Darbost, I. Bonnamour, H. Parrot-Lopez, O. Roy, C. Taillefumier, M. Wimmerová, J.-P. Praly, A. Imberty and S. Vidal, Chem.–Eur. J., 2011, 17, 2146–2159 CrossRef CAS.
- Z. H. Soomro, S. Cecioni, H. Blanchard, J.-P. Praly, A. Imberty, S. Vidal and S. E. Matthews, Org. Biomol. Chem., 2011, 9, 6587–6597 CAS.
- D. Sicard, S. Cecioni, M. Iazykov, Y. Chevolot, S. E. Matthews, J.-P. Praly, E. Souteyrand, A. Imberty, S. Vidal and M. Phaner-Goutorbe, Chem. Commun., 2011, 47, 9483–9485 RSC.
- F. Pertici and R. J. Pieters, Chem. Commun., 2012, 48, 4008–4010 RSC.
- R. U. Kadam, M. Bergmann, M. Hurley, D. Garg, M. Cacciarini, M. A. Swiderska, C. Nativi, M. Sattler, A. R. Smyth, P. Williams, M. Cámara, A. Stocker, T. Darbre and J.-L. Reymond, Angew. Chem., Int. Ed., 2011, 50, 10631–10635 CrossRef CAS.
- E. Mitchell, C. Houles, D. Sudakevitz, M. Wimmerova, C. Gautier, S. Perez, A. M. Wu, N. Gilboa-Garber and A. Imberty, Nat. Struct. Biol., 2002, 9, 918–921 CrossRef CAS.
- D. Tielker, S. Hacker, R. Loris, M. Strathmann, J. Wingender, S. Wilhelm, F. Rosenau and K.-E. Jaeger, Microbiology, 2005, 151, 1313–1323 CrossRef CAS.
- K. M. Bartels, H. Funken, A. Knapp, M. Brocker, M. Bott, S. Wilhelm, K. E. Jaeger and F. Rosenau, J. Bacteriol., 2011, 193, 1107–1113 CrossRef CAS.
- G. M. L. Consoli, G. Granata, V. Cafiso, S. Stefani and C. Geraci, Tetrahedron Lett., 2011, 52, 5831–5834 CrossRef CAS.
- F. Morvan, A. Meyer, A. Jochum, C. Sabin, Y. Chevolot, A. Imberty, J.-P. Praly, J.-J. Vasseur, E. Souteyrand and S. Vidal, Bioconjugate Chem., 2007, 18, 1637–1643 CrossRef CAS.
- K. Marotte, C. Preville, C. Sabin, M. Moume-Pymbock, A. Imberty and R. Roy, Org. Biomol. Chem., 2007, 5, 2953–2961 CAS.
- M. Andreini, M. Anderluh, A. Audfray, A. Bernardi and A. Imberty, Carbohydr. Res., 2010, 345, 1400–1407 CrossRef CAS.
- A. Clouet, T. Darbre and J. L. Reymond, Angew. Chem., Int. Ed., 2004, 43, 4612–4615 CrossRef CAS.
- N. Maillard, A. Clouet, T. Darbre and J. L. Reymond, Nat. Protocols, 2009, 4, 132–142 CAS.
- J.-L. Reymond and T. Darbre, Org. Biomol. Chem., 2012, 10, 1483–1492 CAS.
- E. M. V. Johansson, E. Kolomiets, F. Rosenau, K.-E. Jaeger, T. Darbre and J.-L. Reymond, New J. Chem., 2007, 31, 1291–1299 RSC.
- E. Kolomiets, E. M. Johansson, O. Renaudet, T. Darbre and J. L. Reymond, Org. Lett., 2007, 9, 1465–1468 CrossRef CAS.
- E. M. V. Johansson, R. U. Kadam, G. Rispoli, S. A. Crusz, K.-M. Bartels, S. P. Diggle, M. Camara, P. Williams, K.-E. Jaeger, T. Darbre and J.-L. Reymond, MedChemComm, 2011, 2, 418–420 RSC.
- E. M. V. Johansson, S. A. Crusz, E. Kolomiets, L. Buts, R. U. Kadam, M. Cacciarini, K.-M. Bartels, S. P. Diggle, M. Cámara, P. Williams, R. Loris, C. Nativi, F. Rosenau, K.-E. Jaeger, T. Darbre and J.-L. Reymond, Chem. Biol., 2008, 15, 1249–1257 CrossRef CAS.
- E. Lameignere, L. Malinovská, M. Sláviková, E. Duchaud, E. P. Mitchell, A. Varrot, O. Šedo, A. Imberty and M. Wimmerová, Biochem. J., 2008, 411, 307–318 CrossRef CAS.
- E. Lameignere, T. C. Shiao, R. Roy, M. Wimmerová, F. Dubreuil, A. Varrot and A. Imberty, Glycobiology, 2010, 20, 87–98 CrossRef CAS.
- J. Rieger, H. Freichels, A. Imberty, J. L. Putaux, T. Delair, C. Jérôme and R. Auzély-Velty, Biomacromolecules, 2009, 10, 651–657 CrossRef CAS.
- Z.-H. Lun, Q.-P. Wang, X.-G. Chen, A.-X. Li and X.-Q. Zhu, Lancet Infect. Dis., 2007, 7, 201–209 CrossRef.
- A. Kouki, S. Haataja, V. Loimaranta, A. T. Pulliainen, U. J. Nilsson and J. Finne, J. Biol. Chem., 2011, 286, 38854–38864 CrossRef CAS.
- H. M. Branderhorst, R. Kooij, A. Salminen, L. H. Jongeneel, C. J. Arnusch, R. M. Liskamp, J. Finne and R. J. Pieters, Org. Biomol. Chem., 2008, 6, 1425–1434 CAS.
- J. Ohlsson, A. Larsson, S. Haataja, J. Alajaaski, P. Stenlund, J. S. Pinkner, S. J. Hultgren, J. Finne, J. Kihlberg and U. J. Nilsson, Org. Biomol. Chem., 2005, 3, 886–900 CAS.
- M. Toivanen, S. Huttunen, J. Duricova, P. Soininen, R. Laatikainen, V. Loimaranta, S. Haataja, J. Finne, S. Lapinjoki and C. Tikkanen-Kaukanen, Phytother. Res., 2010, 24, S95–S101 CrossRef.
- N. Parera Pera, A. Kouki, S. Haataja, H. M. Branderhorst, R. M. Liskamp, G. M. Visser, J. Finne and R. J. Pieters, Org. Biomol. Chem., 2010, 8, 2425–2429 Search PubMed.
- C. Raetz and C. Whitfield, Annu. Rev. Biochem., 2002, 71, 635–700 CrossRef CAS.
- B. E. Clark, K. Auyeung, E. Fregolino, M. Parrilli, R. Lanzetta, C. De Castro and R. Pantophlet, Chem. Biol., 2012, 19, 254–263 CrossRef CAS.
- X. Bai, J. Findlow and R. Borrow, Expert Opin. Biol. Ther., 2011, 11, 969–985 CrossRef CAS.
- G. D'Errico, A. Silip, G. Mangiapia, A. Molinaro, L. Paduano and R. Lanzetta, Phys. Chem. Chem. Phys., 2009, 11, 2314–2322 RSC.
- G. D'Errico, A. Silipo, G. Mangiapia, G. Vitiello, A. Radulescu, A. Molinaro, R. Lanzetta and L. Paduano, Phys. Chem. Chem. Phys., 2010, 12, 13574–13585 RSC.
- M. Durka, K. Buffet, J. Iehl, M. Holler, J. F. Nierengarten and S. P. Vincent, Chem.–Eur. J., 2012, 18, 641–651 CrossRef CAS.
- R. Ristl, K. Steiner, K. Zarschler, S. Zayni, P. Messner and C. Schaffer, Int. J. Microbiol., 2011, 2011, 127870 Search PubMed.
- M. F. Feldman, M. Wacker, M. Hernandez, P. G. Hitchen, C. L. Marolda, M. Kowarik, H. R. Morris, A. Dell, M. A. Valvano and M. Aebi, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 3016–3021 CrossRef CAS.
- I. Hug and M. F. Feldman, Glycobiology, 2011, 21, 138–151 CrossRef CAS.
- K. Zarschler, B. Janesch, B. Kainz, R. Ristl, P. Messner and C. Schaffer, Carbohydr. Res., 2010, 345, 1422–1431 CrossRef CAS.
- K. Steiner, A. Hanreich, B. Kainz, P. G. Hitchen, A. Dell, P. Messner and C. Schaffer, Small, 2008, 4, 1728–1740 CrossRef CAS.
- R. Jerala, Int. J. Med. Microbiol., 2007, 297, 353–363 CrossRef CAS.
- H. Loppnow, P. Libby, M. Freudenberg, J. H. Krauss, J. Weckesser and H. Mayer, Infect. Immun., 1990, 58, 3743–3750 CAS.
- F. Peri and M. Piazza, Biotechnol. Adv., 2012, 30, 251–260 CrossRef CAS.
- F. Peri, M. Piazza, V. Calabrese, G. Damore and R. Cighetti, Biochem. Soc. Trans., 2010, 38, 1390–1395 CrossRef CAS.
- M. Piazza, C. Rossini, S. Della Fiorentina, C. Pozzi, F. Comelli, I. Bettoni, P. Fusi, B. Costa and F. Peri, J. Med. Chem., 2009, 52, 1209–1213 CrossRef CAS.
- M. Piazza, L. Yu, A. Teghanemt, T. Gioannini, J. Weiss and F. Peri, Biochemistry, 2009, 48, 12337–12344 CrossRef CAS.
- M. Piazza, V. Calabrese, G. Damore, R. Cighetti, T. Gioannini, J. Weiss and F. Peri, ChemMedChem, 2012, 7, 213–217 CrossRef CAS.
- M. Piazza, M. Colombo, I. Zanoni, F. Granucci, P. Tortora, J. Weiss, T. Gioannini, D. Prosperi and F. Peri, Angew. Chem., Int. Ed., 2011, 50, 622–626 CrossRef CAS.
- C. N. Scanlan, R. Pantophlet, M. R. Wormald, E. Ollmann Saphire, R. Stanfield, I. A. Wilson, H. Katinger, R. A. Dwek, P. M. Rudd and D. R. Burton, J. Virol., 2002, 76, 7306–7321 CrossRef CAS.
- R. Pejchal, K. J. Doores, L. M. Walker, R. Khayat, P. S. Huang, S. K. Wang, R. L. Stanfield, J. P. Julien, A. Ramos, M. Crispin, R. Depetris, U. Katpally, A. Marozsan, A. Cupo, S. Maloveste, Y. Liu, R. McBride, Y. Ito, R. W. Sanders, C. Ogohara, J. C. Paulson, T. Feizi, C. N. Scanlan, C. H. Wong, J. P. Moore, W. C. Olson, A. B. Ward, P. Poignard, W. R. Schief, D. R. Burton and I. A. Wilson, Science, 2011, 334, 1097–1103 CrossRef CAS.
- J. Ni, H. Song, Y. Wang, N. M. Stamatos and L. X. Wang, Bioconjugate Chem., 2006, 17, 493–500 CrossRef CAS.
- J. Wang, H. Li, G. Zou and L.-X. Wang, Org. Biomol. Chem., 2007, 5, 1529–1540 CAS.
- R. D. Astronomo, E. Kaltgrad, A. K. Udit, S. K. Wang, K. J. Doores, C. Y. Huang, R. Pantophlet, J. C. Paulson, C. H. Wong, M. G. Finn and D. R. Burton, Chem. Biol., 2010, 17, 357–370 CrossRef CAS.
- R. D. Astronomo, H. K. Lee, C. N. Scanlan, R. Pantophlet, C. Y. Huang, I. A. Wilson, O. Blixt, R. A. Dwek, C. H. Wong and D. R. Burton, J. Virol., 2008, 82, 6359–6368 CrossRef CAS.
- J. G. Joyce, I. J. Krauss, H. C. Song, D. W. Opalka, K. M. Grimm, D. D. Nahas, M. T. Esser, R. Hrin, M. Feng, V. Y. Dudkin, M. Chastain, J. W. Shiver and S. J. Danishefsky, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 15684–15689 CrossRef CAS.
- A. Kabanova, R. Adamo, D. Proietti, F. Berti, M. Tontini, R. Rappuoli and P. Costantino, Glycoconjugate J., 2010, 27, 501–513 CrossRef CAS.
- P. M. Enriquez-Navas, M. Marradi, D. Padro, J. Angulo and S. Penades, Chem.–Eur. J., 2011, 17, 1547–1560 CrossRef CAS.
- D. A. Calarese, C. N. Scanlan, M. B. Zwick, S. Deechongkit, Y. Mimura, R. Kunert, P. Zhu, M. R. Wormald, R. L. Stanfield, K. H. Roux, J. W. Kelly, P. M. Rudd, R. A. Dwek, H. Katinger, D. R. Burton and I. A. Wilson, Science, 2003, 300, 2065–2071 CrossRef CAS.
- M. Marradi, P. Di Gianvincenzo, P. M. Enriquez-Navas, O. M. Martinez-Avila, F. Chiodo, E. Yuste, J. Angulo and S. Penades, J. Mol. Biol., 2011, 410, 798–810 CrossRef CAS.
- D. Safari, M. Marradi, F. Chiodo, H. A. T. Dekker, Y. Shan, R. Adamo, S. Oscarson, G. T. Rijkers, M. Lahmann, J. P. Kamerling, S. Penadés and H. Snippe, Nanomedicine, 2012, 7, 651–662 CrossRef CAS.
- C. Sabin, E. P. Mitchell, M. Pokorná, C. Gautier, J.-P. Utille, M. Wimmerová and A. Imberty, FEBS Lett., 2006, 580, 982–987 CrossRef CAS.
- E. Kolomiets, M. A. Swiderska, R. U. Kadam, E. M. V. Johansson, K.-E. Jaeger, T. Darbre and J.-L. Reymond, ChemMedChem, 2009, 4, 562–569 CrossRef CAS.
Footnote |
| † Part of the Multivalent Scaffolds in Glycosciences themed issue. |
| This journal is © The Royal Society of Chemistry 2013 |
