Effect of ancillary ligand on electronic structure as probed by 51V solid-state NMR spectroscopy for vanadium–o-dioxolene complexes
Abstract
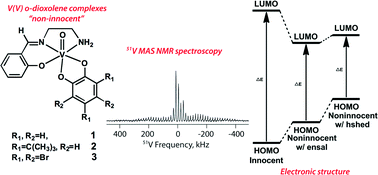
A series of vanadium(V) complexes with o-dioxolene (catecholato) ligands and an ancillary ligand, (N-(salicylideneaminato)ethylenediamine) (hensal), were investigated using 51V solid-state magic angle spinning NMR spectroscopy (51V MAS NMR) to assess the local environment of vanadium(V). The solid-state 51V NMR parameters of vanadium(V) complexes with a related potentially tetradentate ancillary ligand (N-salicylidene-N′-(2-hydroxyethyl)ethylenediamine) (h2shed) were previously shown to be associated with the size of the HOMO–LUMO gap in the complex and, as such, provide insights on the interaction between metal ion and ligand (P. B. Chatterjee et al., Inorg. Chem., 2011, 50, 9794). Our results show that the modification of the ancillary ligand does not affect the observed trend between complexes ranging from catechols with electron-rich to electron-poor substituents. However, the ancillary ligand does affect the size of the HOMO–LUMO separation in the parent complex and thus the solid-state vanadium NMR chemical shift of the unsubstituted vanadium complex. For these complexes, significant changes observed in the isotropic shifts and more modest changes detected in the CQ reflect the electronic changes in the complex as the catechol is varied. However, no obvious trend was observed in the chemical shift anisotropies (δσ and ησ) with the variation in the catechol. The electronic changes in the coordination environment of the vanadium can be determined using solid-state 51V NMR spectroscopy.
- This article is part of the themed collection: NMR crystallography

 Please wait while we load your content...
Please wait while we load your content...