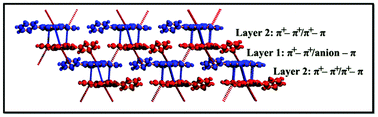
The solid-state complex [PTPH3](NO3)3·2(HNO3) (1) has been synthesized and characterized by X-ray studies, where PTPH3 is the triply protonated form of 4′-(4-pyridyl)-2,2′:6′,2′′-terpyridine (PTP). The solid-state structure of the complex reveals that the π+–π+ interactions are the major driving force in the crystal packing while π+–π, π–π and π–anion interactions assist the overall stabilization of self-assembly. Complex 1 exhibits two different π-stack layers, where layer 1 is generated through π+–π+ interactions and the mutual forces of π+–π+ and π+–π form layer 2. The interaction energies of the main driving forces (π+–π+, π+–π and π–anion interactions) observed in the crystal structure have been calculated using dispersion-corrected density functional theory (DFT-D). An analysis of the Hirshfeld surface of complex 1 shows the intermolecular interactions involved within the crystal structure and corresponding quantitative information are presented by fingerprint plots.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...