Low temperature solvothermal synthesis of anatase TiO2 single crystals with wholly {100} and {001} faceted surfaces†
Abstract
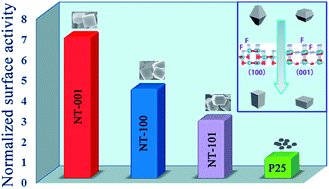
A facile hydrothermal approach for synthesizing anatase TiO2 single crystals with wholly {100} and {001} facets under a reaction temperature as low as 100 °C is reported. NaBF4 and NaF are used as the facet control agents instead of HF, which are safer and much easier to use. By adjusting the reaction conditions, the lowest energy {101} facets can be completely eliminated. The anatase TiO2 with 45% {100} and 55% {001} facets exhibits a considerably higher specific surface photoreactivity than P25. The formation mechanism of the high surface energy facets is explored by

 Please wait while we load your content...
Please wait while we load your content...