Novel organic solar thermal energy storage materials: efficient visible light-driven reversible solid–liquid phase transition†
Yunming
Wang
,
Bingtao
Tang
* and
Shufen
Zhang
State Key Laboratory of Fine Chemicals, Dalian University of Technology, P.O. Box 89, West Campus, 2# Linggong Rd, Dalian 116024, China. E-mail: tangbt@dlut.edu.cn; Fax: +86-411-84986264; Tel: +86-411-84986267
First published on 10th July 2012
Abstract
Solar-thermal energy conversion and storage are one promising solution to directly and efficiently harvest energy from solar radiation. We reported novel organic photothermal conversion-thermal storage materials (OPTCMs) displaying a rapid visible light-harvesting, light-thermal conversion and solid–liquid phase transition thermal energy storage characteristic for solar energy, which is promoted by a dye as an effective “photon capturer and molecular heater” for direct and efficient use of solar radiation.
Introduction
The conversion, storage and application of solar energy represent one of the main challenges in the development of a sustainable energy system.1–8 Although solar-thermal conversion is a direct and efficient application of solar radiation, it suffers from a number of intrinsic drawbacks. For example, it is difficult to overcome the intermittence of time and space solar irradiation, and the visible light which accounts for approximately 44% (ref. 9) of solar radiation almost cannot be directly or effectively applied due to low thermal efficiency.10 Both of these factors can decrease solar energy utilization efficiency greatly. Therefore, solar energy conversion materials with efficient visible light-harvesting, photothermal conversion-thermal storage are very promising for renewable and clean energy sources.Ideally, solar thermal energy production should be linked directly to the harvesting of solar light.11,12 Here we address these issues by obtaining novel organic photothermal conversion materials (OPTCM) via a visible light-driven reversible solid–liquid phase transition based on dyes and phase change materials (Fig. 1a and b). The dye in the OPTCM was used as a photon antenna that served as an effective “photon capturer and molecular heater” of light-to-heat conversion,13,14 and the phase change material stored heat energy by a phase transition with high energy storage density.15,16 Surprisingly, compared with common solar thermal water tanks with sensible heat storage (SHS),17,18 the new materials have some smart features, such as latent heat storage (LHS) with high energy storage density and adjustable phase transition temperature. Simultaneously, the novel materials are able to harvest visible light and convert visible light to thermal energy effectively, as compared to the traditional organic phase change materials for latent heat thermal energy storage.19–21 So, a rapid visible sunlight-harvesting, light-thermal conversion and thermal storage material with solid–liquid phase transition was achieved. Using photothermal calculation methods, the visible light-to-heat conversion efficiency of OPTCMs was determined (η > 0.94) under irradiation conditions. The novel materials show a reversible (more than 200 cycles) phase transition (crystalline state change) via ON/OFF switching of visible light irradiation. The obtained OPTCMs should have potential applications in renewable and clean energy sources.
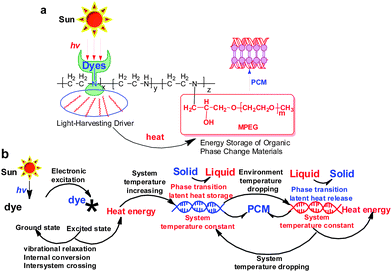 | ||
| Fig. 1 (a) Chemical structures of solar thermal conversion materials with phase change energy storage. (b) Schematic diagram of light-to-heat conversion and storage. | ||
Experimental
Synthesis
The synthesis procedure of OPTCMs is illustrated in Scheme 1. The key intermediate, compound 4 (ESI: structural characterizations†) following the procedure described in the literature,22 with an epoxy group on the MPEG for highly reactive functionalizations, was prepared from MPEG, Na and epichlorohydrin in good yield by a two-step route: replacement and Williamson reaction. Such a route has been well established for the synthesis of ethers by alkylation of alkoxides with alkyl halides. The MPEG and dye (compound 5, ESI: structural characterizations†) were then sequentially introduced to the polyethylene polyamine (M = 423) by epoxy groups and chlorosulfonyl or active chlorine (S-triazine) groups. Throughout the manuscript, compound 7 (ESI: structural characterizations†) is abbreviated as OPTCMs (Scheme 1 and Table 1).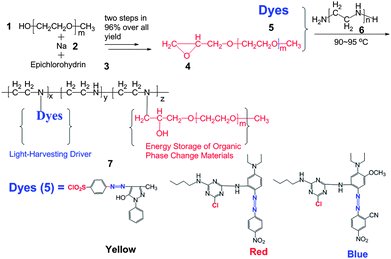 | ||
| Scheme 1 Chemical structures and synthetic scheme for OPTCMs. | ||
| Samples | Compositions |
|---|---|
| a The molar ratio of MPEG, polyethylene polyamine and dyes. | |
| PCM 1 | MPEG-750: polyamine (M = 423) = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| OPTCM 1 | MPEG-750: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1a 0.1a |
| OPTCM 2 | MPEG-750: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.3 0.3 |
| OPTCM 3 | MPEG-750: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
| OPTCM 4 | MPEG-750: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.7 0.7 |
| OPTCM 5 | MPEG-750: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| OPTCM 6 | MPEG-750: polyamine (M = 423): Dye Y = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : : ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| OPTCM 7 | MPEG-1000: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
| OPTCM 8 | MPEG-2000: polyamine (M = 423): Dye Y = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
| OPTCM 9 | MPEG-750: polyamine (M = 423): Dye R = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
| OPTCM 10 | MPEG-750: polyamine (M = 423): Dye B = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
To efficiently use the visible sunlight region in different bands, three-primary colours of OPTCMs (yellow: OPTCM 3, red: OPTCM 9, and blue: OPTCM 10) have been designed and synthesized, for which the UV-Vis absorption spectra were shown in Fig. 2.
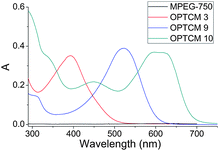 | ||
| Fig. 2 UV-visible absorption spectra of MPEG-750 and OPTCMs in tetrahydrofuran (THF) solution. | ||
Sunlight irradiation experimental section
The schematic of the light-to-heat conversion process under solar irradiation was shown in Fig. 3. The experimental apparatus can be divided into the following three main components: the light-to-heat conversion system, and the data collection and processing devices (Fig. 3). The light-to-heat conversion system consists of a foam insulation system, the OPTCM in a weighing bottle (R = 2.5 cm) and a light source. A power meter (VLP-2000, Changchun Femtosecond Technology Co., Ltd. Jilin, China) was used to measure and verify the power of the light irradiation from the solar and simulated light source (CHF-XM-500W, Beijing Chang Tuo Technology Co., Ltd. Beijing, China). The data collecting and processing devices consist of two subsystems, the temperature versus time data collection system and the processing system by a computer. The temperature versus time data collection system consists of a Pt thermocouple, a thermocouple-to-analog connector (RS-232-RS-485, Instrument Co., Ltd. Jiangsu Suke, China), and a data logger (SK-130RD106062560021A1, Instrument Co., Ltd. Jiangsu Suke, China).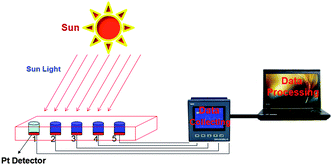 | ||
| Fig. 3 Light-driven transition diagram for the light-to-heat tandem device. | ||
The transduction and storage efficiency of OPTCMs
The visible light-to-heat transduction and energy storage efficiency (η) of OPTCMs was calculated by eqn (1) under light irradiation from the simulated light source (with a measured irradiation area of 4.90 cm2). The temperature change of OPTCMs was measured with foam insulation after simulating light source irradiation at a maximum absorption wavelength (half-wave width = 80 nm) by an optical filter (Fig. 4). The crystal state temperature (ΔT) was measured by DSC technology. The mΔH represented the stored thermal energy of the OPTCM harvesting visible light and conversion of light to thermal energy. The P(Tt−Tf) represents the total irradiation energy of the surface of the OPTCM. The rate value of mΔH to P(Tt−Tf) was the transduction and storage efficiency (η) of the light-to-heat. | ||
| Fig. 4 Efficiency diagram for the light-to-heat tandem device. | ||
Using photothermal calculation methods, the developed solar-thermal materials achieved a peak efficiency for OPTCMs, which turns out to be remarkably close to 1 according to the eqn (1). The parameter η is obtained from the following equation (eqn (1)):
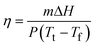 | (1) |
Characterization
Physical and chemistry characterizations of the OPTCMs, their intermediates and MPEG-750 were carried out as described below.A differential scanning calorimeter (DSC) was used in a nitrogen atmosphere by means of an American TA Instruments 910 S DSC thermal analyzer from −20 to 80 °C at a heating rate of 5 °C min−1, the flow rate was 20 ml min−1, the precision on the calorimeter and temperature measurements are ±2.0% and ±2.0 °C, respectively. Samples were measured in a sealed aluminum pan with a mass of about 5.0 mg. The latent heat was calculated as the total area under the peaks of the solid–liquid transitions of OPTCMs by thermal analysis software.
The thermal stability properties were characterized by thermogravimetric analysis (TG) using a Switzerland Mettler-Toledo TGA/SDTA851 thermal analyzer. In each case, the 10 mg specimens were heated from 40 to 700 °C using a linear heating rate of 10 °C min−1 under N2 atmosphere.
X-ray diffraction (XRD) experiments were performed directly on the samples at room temperature using a Japan Rigaku D/Max2400 in the range of 5°–60°.
The UV-visible absorption spectra were measured on a HP-8453 spectrophotometer (USA) in THF solution.
Results and discussion
Sunlight irradiation experiments for the OPTCMs and MPEG-750 (control sample) were carried out. As shown in Fig. 5, upon irradiation with sunlight, the OPTCMs rapidly increased in temperature (colored lines in Fig. 5a and b), while MPEG 750 as a phase change material slowly increased in temperature (black line in Fig. 5a and b). This phenomenon was ascribed to the OPTCMs being able to absorb visible sunlight well (colored lines in Fig. 2) and effectively convert light to thermal energy based on dyes, while the MPEG-750 without dye has no absorption in the visible region (black line in Fig. 2). The dyes in OPTCMs can be used as molecular heaters through photothermal conversion that occurs as a consequence of a nonradiative decay process (vibrational relaxation, internal conversion and intersystem crossing) of excited dyes by visible light irradiation. Under sunlight irradiation, the control sample (MPEG-750) temperature also gradually increased because of the absorption of the near-infrared (NIR) sunlight of PEG.23 As the irradiation time increased, the growth platform of the temperature appeared between 21.0 and 33.0 °C. This was the reason that the OPTCM had a phase transition via absorbing large amounts of heat energy. Therefore, under sunlight irradiation, the dye acted as an effective “photon capturer and molecular heater” to convert light into heat energy and stored heat in the OPTCM by phase transition.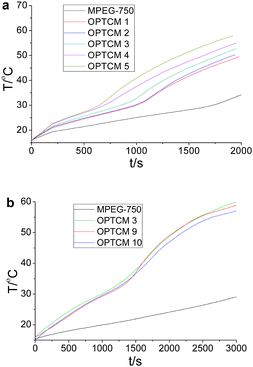 | ||
| Fig. 5 (a) The light-driven efficiencies of MPEG-750 (control sample) and OPTCM 3 (P = 0.26 W, ambient temperature = 16.0 °C, sample = 5.0 g, 10:46–11:23, 27-05-2011, Dalian, China.). (b) The light-driven efficiencies of MPEG-750 (control sample) and OPTCMs (P = 0.30 W, ambient temperature = 15.4 °C, 11:45–12:35, 17-04-2012, Dalian, China). | ||
To address the issue of sunlight-harvesting efficiency, we changed the quantity and type of the dye as a molecular heater for the thermal phase transition of phase change materials. As shown in Fig. 5a, when the used amount of yellow dye (Scheme 1) was increased, the temperature of OPTCM 2–5 was increased faster and the platform of the temperature was significantly shorter than the OPTCM 1 upon irradiation with the sunlight, however a change occurred to a certain extent in the phase transition enthalpy (Fig. 6a and Table 2). Evidently, a highly light-harvesting efficiency occurred when the quantity of the dye was increased. Based on the energy density distribution of visible sunlight irradiation, the type of dye with different absorption wavelength (yellow, red and blue) in the OPTCMs (Fig. 2) was changed to efficiently use the visible sunlight region in different bands. The UV-visible spectra of the synthesized OPTCMs showed absorption over a visible range of spectra (colored lines in Fig. 2). The corresponding results were virtually identical in Fig. 5b, and little effect of the OPTCMs was observed in the phase transition temperature, but the enthalpy of the OPTCMs changed, which is attributed to the steric effect of red and blue dyes (Fig. 6b and Table 2). So, the different bands of the visible sunlight region had been efficient utilized by OPTCMs under solar irradiation. Here, the dyes act as an excellent molecular heater to drive the phase transition of OPTCMs in solar irradiation.
 | ||
| Fig. 6 DSC curves of the OPTCMs, and MPEG-750 and its intermediate (the heating and cooling rate for DSC is 5 °C min−1). | ||
| Samples | ΔH (J g−1) | T p (°C) | ||
|---|---|---|---|---|
| Heating cycle | Cooling cycle | Heating cycle | Cooling cycle | |
| a T p = Peak transition temperature of samples; ΔH = phase transition enthalpy of samples. | ||||
| MPEG-750 | 132.1 | 136.8 | 32.5 | 15.0 |
| PCM 1 | 113.2 | 115.8 | 30.8 | 11.7 |
| OPTCM 1 | 108.0 | 108.2 | 34.3 | 2.2 |
| OPTCM 2 | 107.9 | 107.1 | 33.5 | 1.0 |
| OPTCM 3 | 103.7 | 107.0 | 32.7 | 5.5 |
| OPTCM 5 | 95.8 | 78.3 | 33.6 | −7.8 |
| OPTCM 6 | 84.9 | 74.1 | 33.9 | −0.9 |
| OPTCM 7 | 102.5 | 105.8 | 42.3 | 15.6 |
| OPTCM 8 | 147.5 | 151.3 | 53.0 | 29.0 |
| OPTCM 9 | 98.9 | 91.0 | 32.7 | 5.3 |
| OPTCM 10 | 92.8 | 89.3 | 29.4 | 12.0 |
To further demonstrate the controlled phase transition temperature and enthalpy, we changed the average molecular weight of MPEG 750, 1000 and 2000 (Fig. 6c) as well as the grafting amount of MPEG (Fig. 6d). The differential scanning calorimetric analysis was conducted to investigate the influence of the transition temperature and enthalpy of the OPTCMs. The DSC curves of OPTCMs were displayed in Fig. 6, respectively. As can be seen from Fig. 6c and Table 2, there was a significant increase in the phase transition temperature and enthalpy of OPTCM 3, 7 and 8, when the average molecular weight of MPEG gradually decreased. Additionally, with the amount of MPEG decreasing, the enthalpy of OPTCMs was gradually increased, and no change occurred in the phase transition temperature (OPTCM 5, 6 in Fig. 6d and Table 2). Therefore, the OPTCM exhibited promise as mentioned above.
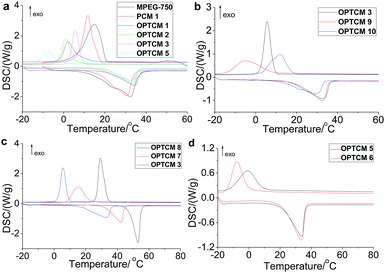
Of crucial importance is the sunlight-to-heat and energy storage efficiency (η) of OPTCMs. Using photothermal calculation methods, the obtained transduction and storage efficiency (η) of OPTCM 3, 9 and 10 was 0.973, 0.941 and 0.956 using the device of Fig. 4, respectively. The calculated η is according to eqn (1) and Fig. 7 (black line in a, c and e), as expected for solar energy materials.
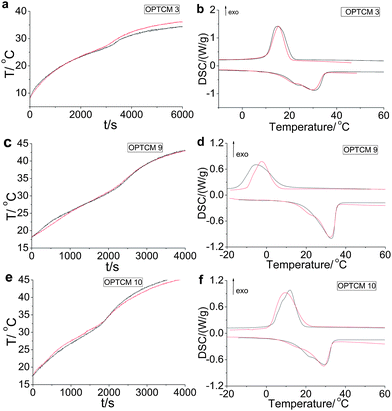 | ||
| Fig. 7 (a) The light-driven spectrum of OPTCM 3 before (black line) and after (red line) 200 cycle sunlight irradiations (P = 0.11 W, λmax = 400 nm, ambient temperature = 9.0 °C). (b) DSC curves of OPTCM 3 before (black line) and after (red line) 200 cycle irradiations. (c) The light-driven spectrum of OPTCM 9 before (black line) and after (red line) 200 cycle sunlight irradiations (P = 0.24 W, λmax = 500 nm, ambient temperature = 18.1 °C). (d) DSC curves of OPTCM 9 before (black line) and after (red line) 200 cycle irradiations. (e) The light-driven spectrum of OPTCM 10 before (black line) and after (red line) 200 cycle sunlight irradiations (P = 0.24 W, λmax = 600 nm, ambient temperature = 17.6 °C). (f) DSC curves of OPTCM 10 before (black line) and after (red line) 200 cycle irradiations. | ||
To prove the sunlight durability of OPTCMs, sunlight irradiation cycling tests were carried out, and the result of a 200 cycle test was shown in Fig. 7 (using the device of Fig. 4). The tests were conducted in a sunlight operation placed for 20 min, and followed by covering the irradiation for 20 min in a temperature-controlled water bath (5 °C) to ensure the solid–liquid cycles. Then, it was found that the irradiation spectral features obtained before (black line in Fig. 7a, c and e) and after (red line in Fig. 7a, c and e) the irradiation were virtually identical. Additionally, OPTCMs were then checked with DSC for their latent heat storage capacity and melting temperature change (Fig. 7b, d and f). As a representative example, the following thermal properties of OPTCMs were described by using OPTCM 3. Compared with 103.2 J g−1 and 106.7 J g−1 after 200 solid–liquid cycles, the latent heats of melting and freezing were 103.7 J g−1 and 107.0 J g−1 before the cycles, respectively. Amazingly, no notable deterioration of the OPTCM was observed even after 200 cycle operations.
Visible sunlight-energy conversion usually takes one of two forms: the ‘quantum’ approach and the ‘thermal’ approach (Fig. 8).5 Often the energy input from light produces an electronic excited state. The dπ–dπ* (or dπ*–dσ*) excited states of dyes are short-lived and dissociative. Rapid transition to the ground state by the ‘thermal’ approach is shown by a, b and c (Fig. 8). To prove the mechanism of solar thermal conversion, we did fluorescence experiments on the OPTCMs in different solvents, such as toluene, chloroform and water. It was found that no fluorescence was issued, and we determined the efficiency (η) of light-to-heat conversion and thermal energy storage which turned out to be close to 1 (η > 0.94). Therefore the sunlight irradiated OPTCMs, by way of non-radiative decay from excited states to the ground state, generated heat and stored the energy by phase transitions of the organic photothermal conversion materials.
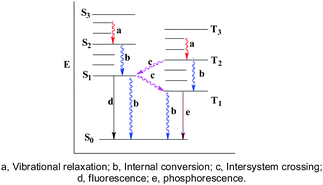 | ||
| Fig. 8 The proposed mechanism of solar thermal conversion. | ||
For pure organic compounds, thermal stability was of crucial importance. In order to demonstrate the thermal stability of the OPTCMs, the thermogravimetric analysis (TG) and derivative thermogravimetric analysis (DTG) were investigated. The corresponding results are presented in Fig. 9 and Table 3, where the degradation temperatures of the OPTCMs and their intermediate were all increased and the thermal stability of OPTCMs was confirmed. Corresponding to the degradation process, the sharp weight loss, when the temperature was around 386.5 °C, was ascribed to the decomposition of organic ingredients, namely the breaking of the PEG chains and dye destruction. As shown in Table 3, the charred residue of the OPTCMs can be deduced, which was identical to the percentages of the charred residues of the PEG at 647 °C and was very close to the theoretical result of organic compounds.
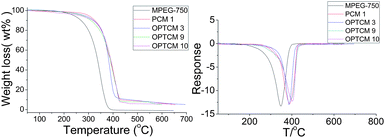 | ||
| Fig. 9 TG and DTG curves of OPTCMs, and MPEG-750 and its intermediate. | ||
| Sample | T −5 wt% (°C) | T max (°C) | Charred residue at 647 °C (wt%) |
|---|---|---|---|
| MPEG-750 | 238.4 | 347.8 | 0.04 |
| PCM 1 | 299.7 | 390.1 | 4.89 |
| OPTCM 3 | 282.9 | 386.5 | 5.11 |
| OPTCM 9 | 273.1 | 396.9 | 5.64 |
| OPTCM 10 | 263.7 | 403.6 | 7.89 |
To further prove the crystalline properties of OPTCMs, X-ray diffraction was used. Fig. 10 presents powder X-ray diffraction (XRD) patterns of the prepared samples from synthesized OPTCMs and MPEG-750 (control sample). Sharp and intense diffraction peaks at 19.14° and 23.26° were observed for the OPTCMs and MPEG-750, indicating that the MPEG was present in the crystalline state.24 The OPTCMs' diffraction peak heights are lower than that of pure MPEG, which means their crystallization decreases. Because they have the same phase change component (polyether), the crystallization peak position of OPTCMs was the same as MPEG, and their peak does not shift. According to the analysis results of the above DSC and XRD, the OPTCMs still have good crystalline properties.
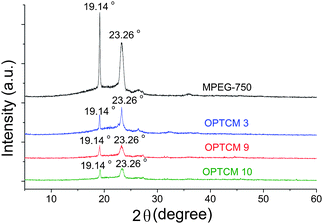 | ||
| Fig. 10 X-ray diffraction of MPEG-750 and OPTCMs. | ||
Conclusions
Novel organic solar thermal energy storage materials with an efficient light-harvesting driven reversible solid–liquid phase transition were designed and synthesized as solar energy storage materials for meeting the demands of mankind. There were some notable advantages such as, the OPTCMs had high efficiency (η > 0.94) of light-to-heat conversion and thermal energy storage by the solid–liquid phase transition with large heat storage density. Additionally, the photothermal energy conversion material had some intelligent characteristics in that the phase transition temperature and enthalpy can be well adjusted and controlled, and the visible light absorption wavelengths can be controlled by changing the structures of the dyes. In further work, the novel materials may be used as smart clothing by fabric blending. An excellent thermal stability and reversible solid–liquid phase transition of OPTCMs were achieved. The present result opens new vistas for the research of energy production schemes, using light-to-heat conversion and heat energy storage macromolecular organic structures as functional materials that are cheap and commonly available.Our concept of solar energy conversion materials will stimulate further fundamental work on the structures of the dyes as “photon capturers and molecular heaters” for OPTCMs. Further study is necessary to better understand and design more materials that enable and promote light-to-heat conversion and energy storage.
Acknowledgements
This work was supported by the National Natural Science Foundation for Young Scholar of China (20804007), the State Key Program of National Natural Science Foundation of China (20836001), Doctoral Fund of Ministry of Education of China (200801411032), the National Science and Technology Pillar Program (2011BAE07B01).Notes and references
- A. Sarı and A. Biçer, Sol. Energy Mater. Sol. Cells, 2012, 101, 114–122 CrossRef.
- A. Sarı, C. Alkan, A. Biçer and A. Karaipekli, Sol. Energy Mater. Sol. Cells, 2011, 95, 3195–3201 CrossRef.
- S. Shaikh and K. Lafdi, Energy Convers. Manage., 2006, 47, 2103–2117 CrossRef.
- F. Huth, M. Schnell, J. Wittborn, N. Ocelic and R. Hillenbrand, Nat. Mater., 2011, 10, 352–356 CrossRef CAS.
- J. W. Schwede, I. Bargatin, D. C. Riley, B. E. Hardin, S. J. Rosenthal, Y. Sun, F. Schmitt, P. Pianetta, R. T. Howe, Z. X. Shen and N. A. Melosh, Nat. Mater., 2010, 9, 762–767 CrossRef CAS.
- A. Tada, Y. Geng, Q. Wei, K. Hashimoto and K. Tajima, Nat. Mater., 2011, 10, 450–455 CrossRef CAS.
- H. H. Richardson, M. T. Carlson, P. J. Tandler, P. Hernandez and A. O. Govorov, Nano Lett., 2009, 9, 1139–1146 CrossRef CAS.
- L. Li, X. Yang, J. Zhao, J. Gao, A. Hagfeldt and L. Sun, J. Mater. Chem., 2011, 21, 5573–5575 RSC.
- X.-Z. Guo, Y.-D. Zhang, D. Qin, Y.-H. Luo, D.-M. Li, Y.-T. Pang and Q.-B. Meng, J. Power Sources, 2010, 195, 7684–7690 CrossRef CAS.
- N. Armaroli and V. Balzani, Angew. Chem., Int. Ed., 2007, 46, 52–66 CrossRef CAS.
- H. Chen, L. Shao, T. Ming, Z. Sun, C. Zhao, B. Yang and J. Wang, Small, 2010, 6, 2272–2280 CrossRef CAS.
- T. Fujigaya, T. Morimoto, Y. Niidome and N. Nakashima, Adv. Mater., 2008, 20, 3610–3614 CrossRef CAS.
- J. Y. Liu, W. H. Fan, K. L. Han, W. Q. Deng, D. L. Xu and N. Q. Lou, J. Phys. Chem. A, 2003, 107, 10857–10861 CrossRef CAS.
- K. Vanherck, S. Hermans, T. Verbiest and I. Vankelecom, J. Mater. Chem., 2011, 21, 6079–6087 RSC.
- Y. N. Xia, J. T. McCann and M. Marquez, Nano Lett., 2006, 6, 2868–2872 CrossRef.
- C. Liu, F. Li, L.-P. Ma and H.-M. Cheng, Adv. Mater., 2010, 22, E28–E62 CrossRef CAS.
- Y. Shen, A. G. Skirtach, T. Seki, S. Yagai, H. Li, H. Möhwald and T. Nakanishi, J. Am. Chem. Soc., 2010, 132, 8566–8568 CrossRef CAS.
- C. G. Granqvist, Adv. Mater., 2003, 15, 1789–1803 CrossRef CAS.
- K. A. R. Ismail and F. A. M. Lino, Exp. Therm. Fluid Sci., 2011, 35, 1010–1018 CrossRef CAS.
- F. Agyenim, N. Hewitt, P. Eames and M. Smyth, Renewable Sustainable Energy Rev., 2010, 14, 615–628 CrossRef CAS.
- A. Koca, H. F. Oztop, T. Koyun and Y. Varol, Renewable Energy, 2008, 33, 567–574 CrossRef CAS.
- A. W. Williamson, XXII.-On etherification, Q. J. Chem. Soc. London, 1852, 4, 229–239 RSC.
- N. Sarier and E. Onder, Thermochim. Acta, 2007, 454, 90–98 CrossRef CAS.
- J. Zhang, L.-Q. Wang, H. Wang and K. Tu, Biomacromolecules, 2006, 7, 2492–2500 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c2jm33289b |
| This journal is © The Royal Society of Chemistry 2012 |
