Low-temperature hydrothermal synthesis of WO3 nanorods and their sensing properties for NO2†
Abstract
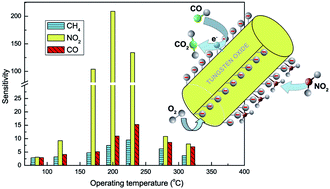
Tungsten trioxide (WO3) nanorods with an aspect ratio of ∼50 have been successfully synthesized by hydrothermal reaction at a low temperature of 100 °C. The crystal structure, morphology evolution and thermal stability of the products are characterized in detail by XRD, FESEM, FTIR, and TG/DTA techniques. The diameter evolution and distribution of WO3 nanorods strongly depend on hydrothermal temperature and time. Hydrothermal conditions of 100 °C and 24 h ensure the formation of well-defined WO3 nanorods. The transition of the crystal structure from monoclinic WO3 to hexagonal WO3 occurs after calcination at 400 °C. The appropriate calcination conditions of the WO3 nanorods are defined to be 600 °C and 4 h for gas-sensing applications. Response measurements reveal that the WO3 sensor operating at 200 °C exhibits high sensitivity to ppm-level NO2 and small cross-sensing to CO and CH4, which makes this kind of sensor a competitive candidate for NO2-sensing applications. Moreover, impedance measurements indicate that a conductivity mechanism of the sensor is mainly dependent on the grain boundaries of WO3 nanorods. A possible adsorption and reaction model is proposed to illustrate the gas-sensing mechanism.

 Please wait while we load your content...
Please wait while we load your content...