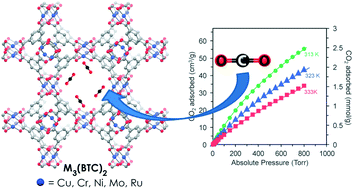
The synthesis, activation, and heats of CO2 adsorption for the known members of the M3(BTC)2 (HKUST-1) isostructural series (M = Cr, Fe, Ni, Zn, Ni, Cu, Mo) were investigated to gain insight into the impact of CO2–metal interactions for CO2 storage/separation applications. With the use of modified syntheses and activation procedures, improved BET surface areas were obtained for M = Ni, Mo, and Ru. The zero-coverage isosteric heats of CO2 adsorption were measured for the Cu, Cr, Ni, Mo, and Ru analogues and gave values consistent with those reported for MOFs containing coordinatively unsaturated metal sites, but lower than for amine functionalized materials. Notably, the Ni and Ru congeners exhibited the highest CO2 affinities in the studied series. These behaviors were attributed to the presence of residual guest molecules in the case of Ni3(BTC)2(Me2NH)2(H2O) and the increased charge of the dimetal secondary building unit in [Ru3(BTC)2][BTC]0.5.

 Please wait while we load your content...
Please wait while we load your content...