DFT study of coverage-depended adsorption of NH3 on TiO2-B (100) surface
Abstract
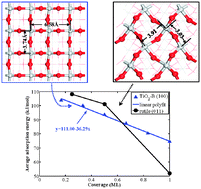
A previous study showed that TiO2-B (100) surface is very unique. It is characterised by high activity and a loose structure. In this study, we studied the adsorption of
- This article is part of the themed collection: Computational Catalysis and Materials for Energy Production, Storage and Utilization

 Please wait while we load your content...
Please wait while we load your content...