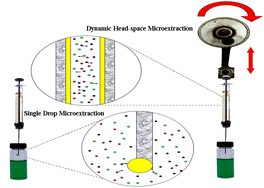
Headspace single-drop microextraction (HS-SDME) and dynamic headspace liquid-phase microextraction (DHS-LPME) combined with GC-FID were used to determine thirteen residual solvents in pharmaceuticals. Different extraction conditions such as microdrop volume, ionic strength of the sample, extraction time, stirring rate, extraction temperature, solvent volume, sampling volume and number of extraction cycles were studied. n-Octanol was used as the extracting solvent for both methods. Under the optimal conditions, the limits of detection for the analytes obtained by DHS-LPME and HS-SDME ranged from 1.9 to 44 μg L−1 and 0.3 to 21 μg L−1, respectively. Enrichment factors ranged 9–212 and 15–379 for DHS-LPME and HS-SDME, respectively. The relative standard deviations (RSDs, n = 5) for both methods are lower than 9.1%. The applicability of the selected method (HS-SDME) in real sample analysis was investigated.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...