Comparative cradle-to-gate life cycle assessments of cellulose dissolution with 1-butyl-3-methylimidazolium chloride and N-methyl-morpholine-N-oxide
Serena
Righi
*a,
Andrea
Morfino
a,
Paola
Galletti
a,
Chiara
Samorì
a,
Alessandro
Tugnoli
b and
Carlo
Stramigioli
b
aCentro Interdipartimentale di Ricerca per le Scienze Ambientali (CIRSA), Alma Mater Studiorum - University of Bologna, via S.Alberto 163, 48123, Ravenna, Italy
bDipartimento di Ingegneria Chimica, Mineraria e delle Tecnologie Ambientali (DICMA), Alma Mater Studiorum - University of Bologna, via Terracini 28, 40131, Bologna, Italy
First published on 24th December 2010
Abstract
In this paper the expected environmental impacts of the industrial cellulose dissolution with the ionic liquid 1-butyl-3-methylimidazolium chloride (Bmim Cl) were analyzed through a “cradle to gate” Life Cycle Assessment (LCA). In order to weigh up the “greenness” of the process, the analysis was performed through a comparison with the well established environmental friendly N-methyl-morpholine-N-oxide (NMMO)/H2O process. Although dissolution of cellulose in Bmim Cl has not been used for industrial production of cellulose fibers to date, results from LCA suggest that it could be interesting from an environmental point of view since its impacts are similar to those of the NMMO/H2O process. Specifically, the process with Bmim Cl generates a higher environmental load on abiotic resource depletion, emissions of volatile organic compounds and ecotoxicity than the NMMO/H2O process. Conversely it has some environmental advantages with regards to human toxicity. In both cellulose dissolution processes, the major contributions to the environmental impacts come from precursor syntheses. In addition to the comparative analysis of the two cellulose dissolution processes, the paper reports the complete life cycle inventory (LCI) of the two solvents, Bmim Cl and NMMO, and their life cycle impact assessment (LCIA).
Introduction
Cellulose is the most abundant renewable resource in the world, and its use as an unmodified material, e.g. cotton, or as a derivative, through chemical, enzymatic or microbiological modification processes, is largely widespread. Cellulose has very strong intermolecular bonds, which prevent the melting of the material and the dissolution in the major part of traditional available solvents. For this reason cellulose and lignocellulosic biomasses in general have been isolated and treated with technologies which cannot be considered as environmental friendly (e.g. harsh alkaline or acidic conditions are involved, the few suitable organic solvents lack greenness since they are often highly volatile, toxic, instable, costly, not recyclable, and give difficulty in the modification of the properties of the polymer itself).1This means that traditional cellulose dissolution processes, including the cuprammonium and xanthate processes (Rayon process), are often cumbersome, expensive and non-sustainable. For example, the viscose Rayon process is a worldwide technology based on the conversion of cellulose into sodium cellulose xanthate, soluble in a caustic solution, making it possible to wet-spin the polymer into a fibre or film, but, at the same time, this process is accompanied by environmentally hazardous by-products, namely H2S, CS2 and heavy metals.
In recent years new cellulose dissolution methods have been researched quite extensively, looking for new environmentally friendly solvents suitable for the process. Among the new non-viscose processes currently in use in the industry for manufacturing regenerated cellulose fibres and films, the N-methyl-morpholine-N-oxide (NMMO)/H2O process (called the Lyocell process) appears to be the most promising replacement for conventional viscose Rayon process.1
The feature of the NMMO/H2O process is a closed water circuit with an extremely high recovery rate of the solvent (99%), a short production cycle (the dissolution and spinning process is usually done within 5 h) and the possibility to operate with highly concentrated cellulose solutions (25–35%). Additionally, it is more environmentally-friendly than the viscose Rayon processes,2 because it does not produce toxic gases or by-products and the solvent itself is biodegradable and nontoxic.3,4
However it is important to recognize that Lyocell process also involves some drawbacks, such as high operation temperatures, partial cellulose degradation and temporary or permanent discoloration of the resulting fibres, side reactions, pronounced decomposition of NMMO, increased consumption of stabilizers and high costs.5 For this reason the investigation on new solvents able to overcome these disadvantages and, at the same time, to efficiently dissolve cellulose is still very important.
Ionic liquids (ILs) are a class of organic salts with relatively low melting points (below 100 °C) and unique combinations of properties dependent on the combination of cations and anions which can be independently modified.6–8 So far, they have been used to replace the organic solvents in a large number of applications, taking advantage of unique properties such as the capability to dissolve hardly soluble materials, the lack of vapour pressure, the chemical and thermal stability and the ease of recovery and recycle.9–12
Some recent studies on a laboratory scale have demonstrated the use of ionic liquids as alternative “clean” technology in biomass applications, in particular in the dissolution and derivatisation processes of cellulose and wood;13–20 the most important results have been obtained with hydrophilic ionic liquids based on the cation 1-alkyl-3-methylimidazolium, ion paired with coordinating anions such as chlorides, phosphates or acetates.17,211-Butyl-3-methylimidazolium chloride (Bmim Cl) is the most studied ionic liquid in biomass dissolution processes because of the ability of chloride anions to break the hydrogen bonding network present in cellulose; moreover, by exploiting the immiscibility properties of cellulose and the high hydrophilicity of Bmim Cl, the polymer can be precipitated by simply adding water (or organic precipitation solutions) to the mixture. The regenerated cellulose obtained in this way has a similar degree of polymerization and polydispersion of the cellulose in the original material. The dissolution of cellulose in Bmim Cl ranges from 3% to 25% (wt%) according to the degree of polymerization of the biopolymer and to the source of energy applied (ultrasounds, microwaves irradiation and heating);13,15 the best results have been obtained under microwaves impulses (25% wt) probably because the adsorption of microwaves by polar solvents, such as ionic liquids, is very strong and warming occurs quickly.13
This article aims to compare the expected environmental impacts of the two cellulose dissolution methods, if applied at industrial production level: the well established environmentally friendly non-viscose processes with NMMO/H2O and the candidate process with Bmim Cl. The energy and environmental loads expected from each process were estimated by using a chemical process simulation software. The results constituted the input for the application of the Life Cycle Assessment (LCA) methodology. In addition, the complete life cycle inventory (LCI) of the two solvents, Bmim Cl and NMMO, and their life cycle impact assessment (LCIA) were quantified, and the most significant environmental input and output flows were identified (i.e. the hot spots, to provide a foundation for future improvement and optimisation of the processes).
Life cycle assessment
LCA is the most extensively developed and standardized methodology for identifying and assessing the potential environmental impacts associated with a product, process or service over its entire life cycle. A complete LCA study addresses the environmental aspects and potential environmental impacts throughout a product life cycle, from raw material acquisition through production, use, end-of-life treatment, recycling and final disposal (i.e. cradle-to-grave).22 However, LCA studies can also include only selected life cycle stages, e.g. from raw materials extraction to product manufacture; in this case it is referred to as cradle-to-gate analysis, which is the approach utilized in the current study.According to ISO 14040 guidelines,23 the LCA methodology comprises four steps: goal and scope definition, inventory analysis, impact assessment, interpretation. Goal and scope definition is the step in which the study objectives, the function of the product, the functional unit and the boundaries of the system are defined. Life cycle inventory analysis (LCI) is the step of an LCA in which the material and energy flows are compiled and quantified. The third phase, Life cycle impact assessment (LCIA), is based on the aggregation of the environmental impacts quantified in the inventory analysis into a limited set of recognizable impact categories (e.g. global warming potential) and corresponding indicators (e.g. kg CO2 equivalents). The last step, Interpretation, is a systematic procedure to evaluate the information from the inventory analysis and the impact assessment of the product system and to draw conclusions from all of the foregoing results of the study.
In the present study the LCA software Gabi 4 by PE International24 was employed to support the life cycle inventory and analysis of the two cellulose dissolution processes.
The analyzed systems were: i) the cellulose dissolution process with NMMO/H2O, ii) the dissolution process with Bmim Cl and iii) all the upstream processes in production chain. Since the main purpose of the system was to dissolve cellulose pulp to obtain textile fibres, the chosen LCA functional unit (i.e. reference unit of production) was 1 kg of the dissolved cellulose.
The system boundaries (Fig. 1) included all upstream processes, as cellulose pulp production, energy and chemicals production, and transportations, necessary to operate the pulp dissolution process. These boundaries have been chosen since downstream contributions are expected to be independent of the dissolution process used.
 | ||
| Fig. 1 System boundaries. | ||
For both the assessed systems, the following assumptions have been made:
• For database consistency issues, all the industrial processes analysed in this paper were located in Germany. For import of resources (e.g. natural gas), the boundaries were extended: the available data was partly based on global, European or Swiss average values. Cellulose pulp production process was from Eucalyptus ssp. and has been located in Thailand.25
• According to Frischknecht et al.26 and to Hischier et al.,27 standard distances were assumed for transport of goods. With regard to basic chemicals we estimated these standard distances to be 100 km lorry and 600 km train transport within Europe.
• According to Hischier et al.,27 emissions to air were estimated on the assumption that 0.2% of the input materials were emitted to air. The water emissions were calculated as the difference between the unreacted input materials and the air emissions.
• As the importance of the infrastructure of a chemical production plant is commonly assumed to be low27 or insignificant,28 in this study the impacts from construction and maintenance of production plant and equipment were neglected.
In addition to Gabi 4 database and the CPS software, also technical literature and reference books were used as information sources.
The typical inputs of a CPS involve the definition of the chemical components, the selection of thermodynamic and property models, the definition of a process flow diagram (scheme of operations and units) and the specification of input flows and operating conditions (flow rate, composition, temperature, pressure, etc.). The properties of most of the common chemicals were available in the simulator database and were utilized. In cases in which the information was not available, the required physical properties were obtained from relevant technical literature. The process flow diagrams (PFD) were designed by selecting and connecting the unit operations relevant for the industrial process of interest (e.g. reactors, separators, distillation columns, heat exchangers, etc.). The solution by the CPS of the energy and material balances as well as the preliminary sizing of major units allowed for the estimation of the overall process conversions, the energy consumption (both thermal and electric) and the waste stream production. These data were the essential input for the definition of the process LCI by Gabi 4.
In the calculation of impact indicators, the LCA methodology based on CML 2001 developed by the University of Leiden have been used.34 The environmental impact categories here considered have been: abiotic resources depletion potential (ADP), global warming potential (GWP), ozone depletion potential (ODP), human toxicity potential (HTP), acidification potential (AP), eutrophication potential (EP), photochemical ozone creation potential (POCP), freshwater aquatic ecotoxicity potential (FAETP) and terrestrial ecotoxicity potential (TETP). Moreover, emission of volatile organic compounds (VOCs) has been considered, since a low emission of these compounds is usually considered as an important issue in favour of the “greenness” of ILs.
Chemical process description
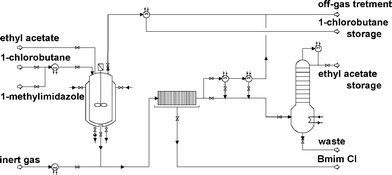 | ||
| Fig. 2 Process flow diagram considered for Bmim Cl manufacturing process. | ||
Cellulose dissolution process
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 instead of 1
6 instead of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) and the spinning mass temperature (116 °C instead of 94 °C) as used by Kosan and co-authors on laboratory-scale.45 These changes have been done in order to obtain similar mechanic characteristics of the fibre found by Kosan and co-authors in their approach.
8) and the spinning mass temperature (116 °C instead of 94 °C) as used by Kosan and co-authors on laboratory-scale.45 These changes have been done in order to obtain similar mechanic characteristics of the fibre found by Kosan and co-authors in their approach.
Since water solubility of the two solvent is very high and both have a boiling point higher than water, the same recovery of the solvent, i.e. 99%, was assumed.
It is important to note that, all common phases (which would not affect the comparison) were neglected.
The block diagram for the alternative processes with Bmim Cl and with NMMO/H2O is shown in Fig. 4.
 | ||
| Fig. 4 Alternative block diagram of cellulose dissolution (left branch with Bmim Cl and right branch with NMMO/H2O). MR: molar ratio. | ||
Results and discussion
Inventory and impact analysis of Bmim Cl synthesis
The Bmim Cl synthesis process presents a long supply chain from natural resources to the end product, therefore it requires large quantities of materials, energy and solvents and it involves organic compounds emissions in air and water. The total score of each environmental impact category (LCIA) resulting from classification and characterization of Bmim Cl production is shown in Table 1. Global warming, freshwater aquatic ecotoxicity and human toxicity potentials were the most significant environmental impacts associated with “cradle to gate” Bmim Cl LCA. This conclusion agreed with findings by Kralisch and co-authors46 which highlighted high cumulative energy demand (related to GWP) and medium human toxicity of 1-methylimidazole and medium ecotoxicity of 1-chlorobutane for aquatic organisms. Our characterization results were also in good agreement with those reported by Zhang47 for the ionic liquid Bmim BF4 obtainable directly from Bmim Cl and NaBF4, confirming the high environmental impact of the production chain leading to the synthesis of these compounds.| Impact category | Total score | Unit | |
|---|---|---|---|
| Bmim Cl | NMMO | ||
| Abiotic resources depletion (ADP) | 6.1E−02 | 2.9E−02 | kg Sb-eq |
| Global warming potential (GWP) | 6.4E+00 | 3.3E+00 | kg CO2-eq |
| Ozone depletion potential (ODP) | 1.3E−07 | 1.0E−07 | kg CFC11-eq |
| Human toxicity potential (HTP) | 1.3E−01 | 2.2E−01 | kg DCB-eq |
| Acidification potential (AP) | 1.6E−02 | 5.6E−03 | kg SO2-eq |
| Eutrophication potential (EP) | 3.8E−03 | 3.2E−03 | kg PO43−-eq |
| Photochemical ozone creation potential (POCP) | 7.0E−03 | 8.1E−04 | kg C2H-eq |
| Freshwater aquatic ecotoxicity potential (FAETP) | 1.0E+00 | 3.3E−02 | kg DCB-eq |
| Terrestrial ecotoxicity potential (TETP) | 8.2E−03 | 4.6E−03 | kg DCB-eq |
| Volatile organic compounds (VOCs) | 4.0E−02 | 1.2E−02 | kg |
Fig. 5 shows the percentage contributions to impact categories of main starting materials for the syntheses of Bmim Cl and NMMO. As regards Bmim Cl, it appears that the major impacts in all categories come from 1-methylimidazole synthesis. About 60% of abiotic resource depletion came from 1-methylimidazole and was associated with fossil fuels consumption for energy and chemicals production. Acidification potential was mainly contributed by glyoxal, direct precursor in the synthesis of 1-methylimidazole, (30%) and butanol (40%) production principally due to sulfur and nitrogen oxides air emissions. Eutrophication potential was almost entirely generated by the synthesis and precursors of 1-methylimidazolium (about 80%), largely due to emission to air of nitrogen oxide, and emission to water of ammonia and nitrogen ions (from the wastewater treatment). Freshwater aquatic ecotoxicity potential derived almost exclusively from 1-methylimidazolium synthesis (about 99%) mainly due to discharge of formaldehyde into the wastewater. Also terrestrial ecotoxicity potential derived for a large part (about 80%) from 1-methylimidazolium synthesis and in particular from formaldehyde emissions. In GWP glyoxal production was the first responsible (about 30%) of the impact. Regarding the emissions, CO2 was the main impact followed by VOCs, methane and hydrocarbons (HC). Human toxicity was mostly caused by airborne emissions of non-methane VOCs (NMVOC), mainly from the ethylene glycol (about 56%) and butanol (about 20%) syntheses. Also ozone depletion potential derived almost exclusively from 1-methylimidazolium (over 90%). Photochemical ozone creation potential was mainly due to the synthesis and precursors of 1-methylimidazolium (about 65%), in particular to formaldehyde and hydrocarbon emissions. VOCs emissions mainly derived from 1-methylimidazolium (about 60%) and 1-chlorobutane (about 30%).
 | ||
| Fig. 5 Solvent production of Bmim Cl (upper panel) and NMMO (lower panel): contribution to LCIA of the solvent manufacturing process (S.M.P.) and the direct precursor syntheses on each of the 10 environmental categories: abiotic resources depletion (ADP), acidification potential (AP), eutrophication potential (EP), freshwater aquatic ecotoxicity potential (FAETP), global warming potential (GWP), human toxicity potential (HTP), ozone depletion potential (ODP), photochemical ozone creation potential (POCP), terrestrial ecotoxicity potential (TETP) and emission of volatile organic compounds (VOC). | ||
Inventory and impact analysis of NMMO synthesis
The total score of each environmental impact category (LCIA) resulting from classification and characterization of NMMO production is shown in Table 1. Like for Bmim Cl, global warming, human toxicity and freshwater aquatic ecotoxicity potentials resulted in the most significant environmental impacts. For the NMMO synthesis process, the main impacts came from the NMM production (Fig. 5), in particular from the methanol and diethylene glycol synthesis. The only exception was the ODP category, where the contribution of H2O2 was about 80%. Like Bmim Cl, the abiotic depletion was due to fossil fuels consumption for energy and chemicals production. Acidification potential was shared between methylamine (about 30%) and diethylene glycol (about 30%) syntheses due to sulfur and nitrogen oxides air emissions. Eutrophication potential was generated by the synthesis of diethylene glycol (about 70%), due to the chemical oxygen demand and nitrogen oxides emissions. FAETP was completely generated (over 95%) by industrial effluents from diethylene glycol production. The global warming impact, GWP, was primarily caused by the production of hydrogen peroxide (about 40%) and of NMM (about 50%) and it was mainly due to CO2 emission to air. The human toxicity impact, HTP, was almost entirely associated to the sulfur dioxide emission to air in the production of diethylene glycol (about 90%). For photochemical ozone creation, terrestrial ecotoxicity and VOCs emission potentials, the contribution of NMM was always higher than 65%.Comparative LCA of the cellulose dissolution methods
Both impact profiles resulting from 1 kg cellulose dissolution with Bmim Cl and with NMMO/H2O are presented in Table 2. Results showed the large impact in global warming for both the solvents (3.7 kg and 3.5 kg of CO2, respectively). Human toxicity (0.22 kg and 0.23 kg of 1,4 dichlorobenzene equivalent, DCB-eq, respectively) and freshwater ecotoxicity (about 0.12 kg and 0.06 kg of DCB-eq, respectively) also showed significant impacts. It must be underlined that the scores reported in Table 2 do not take into account the contribution from the common phases of the two alternative dissolution processes, since they were not included in the LCI.| Impact category | Total score | Unit | |
|---|---|---|---|
| Bmim Cl-process | NMMO-process | ||
| Abiotic resources depletion (ADP) | 9.1E−03 | 7.7E−03 | kg Sb-eq |
| Global warming potential (GWP) | 3.7E+00 | 3.5E+00 | kg CO2-eq |
| Ozone depletion potential (ODP) | 7.7E−08 | 7.7E−08 | kg CFC11-eq |
| Human toxicity potential (HTP) | 2.2E−01 | 2.3E−01 | kg DCB-eq |
| Acidification potential (AP) | 9.4E−03 | 8.7E−03 | kg SO2-eq |
| Eutrophication potential (EP) | 1.8E−03 | 1.8E−03 | kg PO43−-eq |
| Photochemical ozone creation potential (POCP) | 1.1E−03 | 7.6E−04 | kg C2H-eq |
| Freshwater aquatic ecotoxicity potential (FAETP) | 1.2E−01 | 5.6E−02 | kg DCB-eq |
| Terrestrial ecotoxicity potential (TETP) | 5.6E−03 | 5.5E−03 | kg DCB-eq |
| Volatile organic compounds (VOCs) | 5.2E−03 | 3.7E−03 | kg |
Relative comparisons between Bmim Cl (white bar) and NMMO/H2O (grey bar) dissolution processes were made for each environmental impact, with the first one set as 100% and the other displayed as the level relative to the former (Fig. 6). The environmental impacts of Bmim Cl and NMMO/H2O dissolution processes were nearly of the same order of magnitude for all the impact categories. All categories, except human toxicity, had somehow higher impacts for dissolution with Bmim Cl than with NMMO/H2O. Generally, the differences in impacts were under 25%. Abiotic depletion, acidification, eutrophication, global warming, human toxicity, ozone depletion and terrestrial ecotoxicity impact scores of the two processes are essentially equal. On the contrary, freshwater ecotoxicity, photochemical ozone creation and VOCs emission were about 50% 30% and 30%, respectively, lower for dissolution with NMMO/H2O than the dissolution with the IL. Looking at these findings, it is interesting to note that VOCs emission, known as completely irrelevant during the IL use phase, results in significant extending the system boundaries. The similar environmental balances of the two cellulose solution processes gave evidence of the potential environmental suitability of the process with Bmim Cl. As a matter of fact, many environmental advantages of NMMO/H2O process are common to dissolution with Bmim Cl: high recovery of solvent and highly concentrated cellulose solutions,45 no generation of dangerous by-products like as H2S, CS2 and heavy metals, very simple and relatively short processes (less than 8 h, in comparison with the more than 40 h of the conventional viscose Rayon process) which involve a general reduction of electric energy consumptions. On the other hand, some advantages of NMMO/H2O are not common to Bmim Cl, i.e. low toxicity and relative biodegradability.4
The influence of solvent recovery is studied through a sensitivity analysis assuming recovery rates ranging from 99% to 85% (Fig. 7). Recoveries less than 85% are not supposed economically sustainable. As it is possible to observe from the figure, the results are very sensitive to changes in the assumptions about the recovery rate. The impact categories that appear particularly sensitive to this parameter are: abiotic resources depletion, freshwater aquatic ecotoxicity, photochemical ozone creation and emission of VOCs. These results show that the solvent recovery is a key parameter in the environmental assessment and an important task for chemical engineers to minimize burden upon the environment.
 | ||
| Fig. 6 Comparison of the environmental impacts due to the cellulose dissolution with NMMO/H2O (grey bar) and with Bmim Cl (white bar) on each of the 10 environmental categories: abiotic resources depletion (ADP), acidification potential (AP), eutrophication potential (EP), freshwater aquatic ecotoxicity potential (FAETP), global warming potential (GWP), human toxicity potential (HTP), ozone depletion potential (ODP), photochemical ozone creation potential (POCP), terrestrial ecotoxicity potential (TETP) and emission of volatile organic compounds (VOC). | ||
 | ||
| Fig. 7 Characterization results for 1 kg of the dissolved cellulose through Bmim Cl assuming four different scenarios of solvent recovery rate (99%, 95%, 90% and 85%). | ||
Some remarks on toxicity and biodegradability of Bmim Cl appear necessary although: 1) we have preformed a cradle to gate analysis and the human- and eco-toxicity of both solvents was not included in the balance; 2) a very high recovery of Bmim Cl from cellulose dissolution process can be reasonably hypothesized and 3) its potential impact on aquatic ecosystems could be significantly reduced by adequate management of the industrial wastewaters. At first, volatility of this ionic liquid would prevent atmospheric emissions, therefore the most significant source of exposure should be the liquid emission. Secondly, Bmim Cl is less toxic than other ionic liquids, but in any case its toxicity is higher than that of common organic solvents (such as acetone, methanol, ethanol, etc.) and comparable to toxicity of chlorinated organic compounds (such as dichloromethane and chloroform).48 Finally, 28 day tests of biodegradability in water have shown Bmim Cl as not biodegradable.49 All these evidences indicate that emissions of Bmim Cl in the freshwater could induce damages to the ecosystem and therefore their inclusion into boundary system could modify our results. Unfortunately, at present the lack of the characterisation factors of Bmim Cl for human toxicity and ecotoxicity impact categories prevent its inclusion in the LCIA. So, a complete LCIA of a chemical process involving Bmim Cl will be possible only when its characterisation factors will be available. In any case, in order to attempt a preliminary evaluation, we have carried out a LCIA simulation considering the emission of an equal amount of a “surrogate compound”, the Chlormequat-chloride,50 present in the database and with ecotoxicity similar to that of Bmim Cl.51 In this way, the total score of freshwater aquatic ecotoxicity potential shows a weak enhancement, increasing of about 4%. Obviously, in future, more consolidate data will permit less uncertain conclusions.
Besides the overall impact profiles, a more detailed study for each impact category was performed in order to explore the relative contribution of the four main groups of processes in the lifecycle chain: 1) cellulose pulp production, 2) solvent production, 3) transport and 4) cellulose dissolution process. Fig. 8 shows the relative contribution of the four sub-processes for dissolution with Bmim Cl and NMMO/H2O; the dominance of cellulose pulp production and the irrelevance of transport were clear in almost all the impact categories under study for both the dissolution methods. The low contribution of transportation to impact scores is quite common of “cradle-to-gate” LCA studies of chemical processes.52,53 The contributions of cellulose dissolution process appeared low (<10%) for both the methods. In any case, it is possible to observe some exceptions. The contribution of solvent production to the total score of abiotic resources depletion was about 40% and 30% for Bmim Cl and NMMO/H2O dissolution process, respectively, and the contribution of cellulose dissolution process was about 10% for both methods. The contribution to abiotic resource depletion was mainly due to the extraction of fossil fuels, in particular methane, used to electric and thermal energy production and due to crude oil and methane as raw materials for organic solvent syntheses. In the same way, the contribution of solvent production to the total score of VOCs emission was about 50% and 30% for Bmim Cl and NMMO/H2O dissolution process, respectively.
 | ||
| Fig. 8 Cellulose dissolution by Bmim Cl (upper panel) and by NMMO (lower panel): contribution to LCIA of the four sub-processes transport, cellulose pulp production, solvent production and cellulose dissolution process (C.D.P.) on each of the 10 environmental categories: abiotic resources depletion (ADP), acidification potential (AP), eutrophication potential (EP), freshwater aquatic ecotoxicity potential (FAETP), global warming potential (GWP), human toxicity potential (HTP), ozone depletion potential (ODP), photochemical ozone creation potential (POCP), terrestrial ecotoxicity potential (TETP) and emission of volatile organic compounds (VOC). | ||
The results provided evidence that solvents production involved in Bmim Cl dissolution system contributed significantly also to freshwater ecotoxicity (about 50%) and to photochemical ozone creation (about 40%). The main contribution to photochemical oxidants formation was the emission of formaldehyde and HC related mainly to 1-methylimidazolium (about 64%), in particular to glyoxal production (about 43%). In the same categories, the contributions of solvent production involved in NMMO/H2O dissolution method are less significant (about 5% and little less than 10%, respectively).
Finally, it is observable that in general the percentage contributions to each impact category score of the two cellulose dissolution processes are very similar.
Conclusions
Cradle to gate LCA analysis of cellulose dissolution with the ionic liquid Bmim Cl pointed out the environmental potentials and limits of the possible industrial application of this process, that showed very promising features on laboratory scale studies such as highly concentrated cellulose solutions, no generation of dangerous by-products like as H2S, CS2 and heavy metals, simple and relatively short processes involving electric energy saving.By comparison with the NMMO/H2O dissolution process, the most friendly dissolution process already used on industrial scale, the dissolution with ionic liquid appears promising from an environmental point of view. The most significant increase in environmental impacts results from photochemical ozone creation potential, VOCs emission and freshwater aquatic ecotoxicity. Conversely, it is possible to observe a small reduction of toxic compound emission with Bmim Cl use.
In both cellulose dissolution processes, the major contributions to the environmental impacts come from precursor syntheses, characterized by long supply chains from natural resources to the end product, requiring large quantities of organic materials, energy and solvents and involving harmful emissions in air and water. As a matter of fact, syntheses of both Bmim Cl and NMMO show significant environmental impacts for many of the categories of impact chosen. For Bmim Cl, major contributions come from the starting materials in the synthetic pathway to imidazolium cation, in particular from glyoxal and formaldehyde synthesis and use. Thus, in the scale-up process of a possible future industrial application, the recycling of the solvent appears a crucial point that could highly mitigate the environmental impacts coming from its synthesis and from the cellulose extraction-transformation phase.
These results encourage further investigation on the use of ionic liquids in the dissolution and derivatisation processes of cellulose and wood and confirm that to evaluate the “greenness” of a product or process it is necessary to consider the impacts over the entire life cycle so avoiding problem-shifting from one life cycle stage to another, from one geographic area to another, and from one environmental medium to another.
Acknowledgements
We acknowledge the ministry MiUR and the University of Bologna (Polo Scientifico Didattico di Ravenna and RFO) for funding. The authors wish to thank Dr E. Scimia (Febe Ecologic) for her kind assistance during GABI 4 software implementation and operation and Dr L. Galasso for data processing and collaboration.References
- R. Kotek, in Handbook of fiber chemistry, Taylor & Francis Group, LLC, 2006, pp. 678-771 Search PubMed.
- B. Łaszkiewicz, A. C. G. M. Lodart and S. A. Łódź, Poland, 1997 Search PubMed.
- H. Firgo, M. Eibl and D. Eichinger, Lenzinger Berichte, 1995, 75(96), 47–50 Search PubMed.
- G. Meister and M. Wechsler, Biodegradation, 1998, 9(2), 91–102 CrossRef CAS.
- T. Rosenau, A. Potthast, H. Sixta and P. Kosma, Prog. Polym. Sci., 2001, 26(9), 1763 CrossRef CAS.
- M. J. Earle and K. R. Seddon, ACS Symp. Ser., 2002, 819(2), 10–25 CAS.
- P. Wasserscheid and T. Welton, in Ionic liquids in synthesis, Wiley-VCH, Weinheim, 2nd edn, 2007 Search PubMed.
- T. Welton, Chem. Rev., 1999, 99, 2071–2083 CrossRef CAS.
- V. I. Parvulescu and C. Hardacre, Chem. Rev., 2007, 107, 2615–2665 CrossRef CAS.
- H. Zhao, Chem. Eng. Commun., 2006, 193, 1660–1677 CrossRef CAS.
- N. V. Plechkova and K. R. Seddon, Chem. Soc. Rev., 2008, 37, 123–150 RSC.
- E. F. Borra, O. Seddiki, R. Angel, D. Eisenstein, P. Hickson, K. R. Seddon and S. P. Worden, Nature, 2007, 447, 979–981 CrossRef CAS.
- R. Swatloski, S. Spear, J. Holbrey and R. Rogers, J. Am. Chem. Soc., 2002, 124, 4974–4975 CrossRef CAS.
- S. Zhu, Y. Wu, Q. Chen, Z. Yu, C. Wang, S. Jin, Y. Ding and G. Wu, Green Chem., 2006, 8, 325–327 RSC.
- J. Mikkola, A. Kirilin, J. Tuuf, A. Pranovich, B. Holmbom, L. Kustov, D. Murzin and T. Salmi, Green Chem., 2007, 9, 1229–1237 RSC.
- H. Zhang, J. Wu, J. Zhang and J. He, Macromolecules, 2005, 38, 8272–8277 CrossRef CAS.
- Y. Fukaya, K. Hayashi, M. Wada and H. Ohno, Green Chem., 2008, 10, 44–46 RSC.
- I. Kilpeläinen, H. Xie, A. King, M. Granstrom, S. Heikkinen and D. Argyropoulos, J. Agric. Food Chem., 2007, 55, 9142–9148 CrossRef.
- D. Fort, R. Remsing, R. Swatloski, P. Moyna, G. Moyna and R. Rogers, Green Chem., 2007, 9, 63–67 RSC.
- J. Anderson, J. Ding, T. Welton and D. Armstrong, J. Am. Chem. Soc., 2002, 124, 4974–4975 CrossRef CAS.
- J. Vitz, T. Erdmenger, C. Haensch and U. Schubert, Green Chem., 2009, 11, 417–424 RSC.
- ISO 14044. Environmental management – life cycle assessment – requirements and guidelines; 2006.
- ISO 14040. Environmental management – life cycle assessment – principals and framework; 2006.
- GaBi software-system and databases for life cycle engineering, ver. 4.3, PE International, Stuttgart, Germany, 2002 Search PubMed.
- R. Hischier, Life cycle inventories of packaging and graphical paper. Final report ecoinvent Data v2.0 No. 11, Swiss Centre for Life Cycle Inventories, Dübendorf, 2007 Search PubMed.
- R. Frischknecht, N. Jungbluth, H. J. Althaus, G. Doka, R. Dones, T. Heck, S. Hellweg, T. Heck, R. Hischier, T. Nemecek, G. Rebitzer and M. Spielmann, Int. J. Life Cycle Assess., 2004, 10(1), 3–9.
- G. Geisler, T. B. Hofstetter and C. K. Hungerbühler, Int. J. Life Cycle Assess., 2004, 9(2), 101–113 CrossRef CAS.
- R. Hischier, S. Hellweg, C. Capello and A. Primas, Int. J. Life Cycle Assess., 2005, 10(1), 59–67 CrossRef CAS.
- R. K. Sinnott, Coulson & Richardson's Chemical Engineering Volume 6, Third edition, Butterworth-Heinemann, 1999, Oxford, UK Search PubMed.
- W. D. Baasel, Preliminary chemical engineering plant design 2nd ed., Van Nostrand Reinhold, 1990, New York, NY Search PubMed.
- T. E. Casavant and R. P. Côte, J. Clean. Prod., 2004, 12, 901–908 CrossRef.
- D. A. Glasscock and J. C. Hale, Chem. Eng., 1994, 101(11), 82–89 Search PubMed.
- Aspen Plus®, 10.2-1, Aspen Technology, Burlington, Massachusetts (USA), 2000 Search PubMed.
- J. B. Guinée, M. Gorrée, R. Heijungs, G. Huppes, R. Kleijn, A. de Koning, L. van Oers, A. Wegener Sleeswijk, S. Suh, H. A. Udo de Haes, H. de Bruijn, R. van Duin, M. A. J. Huijbregts, E. Lindeijer, A. A. H Roorda, B. L. Van Der Ven and B. P. Weidema, Life cycle assessment: an operational guide to the ISO standards: Parts 1 and 2, Ministry of Housing, Spatial Planning and Environment (VROM) and Centre of Environmental Science (CML), Den Haag and Leiden, The Netherlands, 2001 Search PubMed.
- S. Park and R. J. Kazlauskas, J. Org. Chem., 2001, 66, 8395–8401 CrossRef CAS.
- Hydrocarbon Processing, The Leonard Process Company, 1979, vol. 58, issue 11, p. 194ff Search PubMed.
- M. W. Forkner, J. H. Robson and W. M. Snellings, in Kirk-Othmer Encyclopedia of Chemical Technology, Forth Edition, Wiley, New York, 1994, vol.12, p. 695 Search PubMed.
- G. Mattioda and A. Blanc, in Ullmann's Encyclopedia of Industrial Chemistry, Fifth Edition, Verlag Chemie, Weinheim, New York, 1989, vol. A12, p. 491 Search PubMed.
- GB Patent, 1 272 592, 1963.
- K. Ebel, H. Koehle, A. Gamer and R. Jäckh, in Ullmann's Encyclopedia of Industrial Chemistry, Fifth Edition, Verlag Chemie, Weinheim, New York, 1989, vol. A13, p. 661 Search PubMed.
- A. von Daniken and M. Chudacoff, Vergleichende okologische Bewertung von Anstrichstoffen in Baubereich – band 2: Daten. Buwal Schriftenreihe Unwelt Nr 232, Buwal, Bern, 1995 Search PubMed.
- US Patent, 4 748 241, 1988.
- US Patent, 5 917 039, 1999.
- US Patent, 4 246 221, 1981.
- B. Kosan, C. Michels and F. Meister, Cellulose, 2008, 15, 59–66 CrossRef CAS.
- D. Kralisch, A. Stark, S. Körsten, G. Kreisel and B. Ondruschka, Green Chem., 2005, 7, 301–309 RSC.
- Y. Zangh, B. R. Bakshi and E. S. Demessie, Environ. Sci. Technol., 2008, 42(5), 1724–1730 CrossRef CAS.
- T. P. Thuy Pham, C.-W. Cho and Y.-S. Yun, Water Res., 2010, 44, 352–372 CrossRef.
- D. Coleman and N. Gathergood, Chem. Soc. Rev., 2010, 39, 600–637 RSC.
- Cycocel® Extra BASF, Safety data Sheet, Version 1.0, revision date 2009/03/18.
- R. J. Bernot, M. A. Brueseke, M. A. Evans-White and G. A. Lamberti, Environ. Toxicol. Chem., 2005, 24, 87–92 CrossRef.
- S. Gonzales-Garcia, A. Hospido, M. T. Moreira, J. Romero and G. Feijoo, J. Clean. Prod., 2009, 17, 1010–1016 CrossRef.
- C. Jimenez-Gonzales, A. D. Curzons, D. J. C. Constable and V. L. Cunningham, Int. J. Life Cycle Assess., 2004, 9, 114–121 CrossRef.
| This journal is © The Royal Society of Chemistry 2011 |

