DOI:
10.1039/C0NR00585A
(Paper)
Nanoscale, 2010,
2, 2790-2797
Encapsulated nano-heat-sinks for thermal management of heterogeneous chemical
reactions
Received
11th August 2010
, Accepted 7th September 2010
First published on 22nd October 2010
Abstract
This paper describes a new way to control temperatures of heterogeneous exothermic
reactions such as heterogeneous catalytic reaction and
polymerization by using encapsulated nanoparticles of phase change materials as thermally functional
additives. Silica-encapsulated indium
nanoparticles and silica encapsulated paraffin nanoparticles
are used to absorb heat released in catalytic reaction and
to mitigate gel effect of polymerization, respectively. The
local hot spots that are induced by non-homogenous catalyst
packing, reactant concentration fluctuation, and abrupt change of polymerization rate lead to solid to liquid phase change of nanoparticle cores so as to avoid thermal runaway by converting
energies from exothermic reactions to latent heat of fusion. By quenching local hot spots
at initial stage, reaction rates do not rise significantly because the thermal energy
produced in reaction is isothermally removed. Nanoparticles
of phase change materials will open a new dimension for thermal management of exothermic
reactions to quench local hot spots, prevent thermal runaway of reaction, and change
product distribution.
Introduction
The thermal runaway of an exothermic chemical reaction refers to a situation, in which
increases in temperature will change reaction condition in such a way that temperature
increases further.1–3 The thermal runaway affects
the yield, selectivity and safety of many reactions including heterogeneous catalytic reaction, free radical
polymerization, electrochemical energy conversion, etc. The thermal
runaway of heterogeneous catalytic reactions can cause side
reactions, catalyst deactivation, and loss in productivity
or selectivity, and leads to explosion if released reaction heat cannot be removed
quickly.4–8 A similar effect in
polymerization is also called auto-acceleration or gel
effect, in which positive feedback will speed up polymerization, and results in heat accumulation due to increased
viscosity.9,10 In the case of electrochemical
reaction, thermal runaway is the major cause of battery explosion due to temperature rise.
A common feature in these exothermic reaction systems is the existence of hot spots
induced by non-uniformities of catalyst packing, reactant
concentration, as well as heating and cooling effects.11–13
The temperature of heterogeneous reactions is often controlled by
proportional–integral–derivative (PID) unit, where
temperature around or inside a reactor will be monitored continuously. Once the measured
temperature is higher than the designed value, cold air or liquid will be circulated to
remove extra heat, or reactant feeding rate will be reduced to limit reaction heat.
However, the performance of temperature control depends on the locations and sizes of
thermocouples, and heating/cooling units, which cannot be
distributed uniformly in the reactor or made sufficiently small to detect or quench
microsize hot spots. For heterogeneous catalytic reactions,
the reactions take place inside micropores, where the temperatures are higher than those
around the reactor. It takes some time for reaction heat transferring to thermocouple from hot spots to activate control units. Such
response delay causes temperature increase and leads to ineffectiveness in temperature
control.14–16 The thermal runaways of
polymerizations cannot be controlled readily using
PID units as well, because high viscosity products
prevent efficient heat transfer from hot spots. A small quantity of inhibitor is often added into the reactor at early stage of runaway, or
polymerization is carried out in water to absorb the reaction heat.
Single phase substances in either liquid or solid phases have small heat capacity and
cannot take much thermal energy. As a result, significant temperature rises occur even if
small amounts of thermal energy are absorbed by solids or liquids. An effective way to
enhance the heat capacity of a pure substance is to add
materials that undergo phase change at certain temperature where the latent heat of fusion
will contribute significantly to the heat capacity of mixture.17 The solid–liquid phase change material (PCM) involves small volume change,
and sufficient heat energy, and has been added into single phase fluid in order to enhance
its heat capacity.18,19 Dissipating heat into PCM
results in a nearly isothermal heat sink during melting and the operating temperature can
be adjusted by using different PCMs so that melting does not occur until needed. A variety
of PCM materials such as paraffin waxes, inorganic salt
hydrates, or metal eutectic alloys have been used to
maintain desired temperature and reduce the fluctuation of temperature.20–23 However, bulk PCMs are not very useful
for many applications such as temperature controls of heterogeneous chemical reactors for
several reasons. (1) In order to exploit latent heats of fusion for heat absorbing, PCMs
should be sufficiently small so that it melts rapidly. It has been shown that if the
radius of PCM is reduced by 10 times, the time required for complete melting reduces by
100 times.24 (2) To work in a heterogeneous reactor, the
PCM should be encapsulated inside a shell with good sealing and stability to prevent
leakage or agglomerate. (3) PCM should form homogeneous mixture with reactants or catalysts in order to quench local microsized hot spots.
Instead of using inert solid (sand) to support catalytic
species or liquid (water) to reduce gel effect of
polymerization reaction, the advent of nanostructured
materials provides a new approach for thermal managements of exothermic reactions. This
article describes the use of a new nanostructured material, encapsulated phase change
nanoparticles (nano-PCM), to control the thermal runaway
of catalytic reactions and polymerizations, where nano-PCMs can absorb reaction heat, and change phases
from solid to liquid. The core materials are metal nanoparticles or paraffin wax nanoparticles,
which change phases at suitable temperature. The non-melting shells are made of silica or polymer, and are
stable at operating conditions to prevent the leakage or oxidization of core
material.25 Due to their small sizes, inert surfaces,
and large heat-absorbing capabilities, nano-PCMs can be mixed homogeneously with solid
catalysts or monomers to quench local hot spots before
accumulation of heat energy at a large scale, thus breaking the positive feedback loop
between the reaction rate and temperature, and postponing onset of thermal runaway by
absorbing reaction heat (Fig. 1). Nano-PCMs do not increase
the size of catalytic reactor, because active species can
be supported at shells of nano-PCMs; in addition, polymers
can be dissolved in its good solvent, and separated from
nano-PCM after polymerization. Two highly exothermic
reactions, platinum catalyzed methanol oxidation and polymerization of
methyl methacrylate (MMA), have been used as
examples to prove the generality of this approach.26,27
Experimental procedures
Chemicals such as tetraethoxysilane, ethanol, methanol, tetrahydrofuran, and polyethylene-block-polyethylene
glycol at molecular weight of 1400 were obtained from Aldrich and used as received.
Poly-α-olefin with boiling point of 250 °C is from
Air Force Research Laboratory. Indium, methyl methacrylate (MMA), azoisobutyronitrile (AIBN), toluene and acetic acid
are from Alfa Aesar. Polyethylene wax (POLYWAX 1000,
melting temperature of 110 °C) is provided by Baker Hughes.
Metallic phase change nanoparticles are made by directly
boiling metal powders. 1.0 g of indium powders (−100
mesh) is boiling in poly-α-olefin (PAO) at ∼200 °C
at a stirring rate of 800 rpm for 12 h.17 After cooling
down to room temperature, nanoparticles are separated by
centrifugation, washed with ethanol for three times, and dried in nitrogen. Sol–gel method is used to encapsulate nanoparticles with silica after removing
nanoparticles by centrifuging and washing by ethanol. The nanoparticles are
encapsulated in silica by decomposing tetraethoxysilane (TEOS) at 70 °C as below. 0.1 g of nanoparticles is dispersed into ethanol and sonicated for 10 min, 1 ml TEOS is added into the solution and
sonicated for 1 h, which is followed by adding ammonia
hydroxide as catalyst for formation of
silica. The thickness of silica shell is controlled by changing TEOS concentration and
TEOS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nanoparticle ratio.28,29
Silica encapsulated polyethylene (paraffin wax)
nanoparticles are made as following. 1.0 g of
polyethylene, 1.0 g of TEOS and 0.25 g polyethylene-block-polyethylene glycol
are dissolved in 4 ml of toluene at 100 °C. 50 ml of
water are added in 250 ml of three-neck flask
equipped with mechanic stirrer, and heated to 95 °C, and toluene solution is poured into three-neck flask, stir vigorously for 60 min
to form white emulsion. 0.4 g of acetic acid
solution (10 wt%) are added into the above mixture, and cool down to 55 °C and stir at
300 rpm for 3 h to ensure complete hydrolysis and
condensation of TEOS. The product is separated by centrifuging at 8500 rpm for 10 min
and wash with water for three times, dry in oven at
60 °C for 24 h.
nanoparticle ratio.28,29
Silica encapsulated polyethylene (paraffin wax)
nanoparticles are made as following. 1.0 g of
polyethylene, 1.0 g of TEOS and 0.25 g polyethylene-block-polyethylene glycol
are dissolved in 4 ml of toluene at 100 °C. 50 ml of
water are added in 250 ml of three-neck flask
equipped with mechanic stirrer, and heated to 95 °C, and toluene solution is poured into three-neck flask, stir vigorously for 60 min
to form white emulsion. 0.4 g of acetic acid
solution (10 wt%) are added into the above mixture, and cool down to 55 °C and stir at
300 rpm for 3 h to ensure complete hydrolysis and
condensation of TEOS. The product is separated by centrifuging at 8500 rpm for 10 min
and wash with water for three times, dry in oven at
60 °C for 24 h.
Characterization
The structures, compositions, and thermal properties of these nanoparticles have been studied by using a variety of techniques. Transmission electron microscopy (TEM) images are acquired on
a JEOL 1011 TEM
operating at 100 kV, and high resolution image is collected on a TECNAI F30 TEM. X-Ray
diffraction (XRD) analysis is carried out on
Rigaku X-ray diffractometer with CuKα radiation (40 kV, 30 mA). Energy-dispersed X-ray spectroscopy is performed using a Zeiss Ultra 55 scanning electron microscopy (SEM). A PerkinElmer differential scanning
calorimetry (DSC 7) is used to measure the
fusion enthalpy and melting temperature of nanoparticles.
Supported Pt/SiO2
catalyst is made by impregnating SiO2 (200
m2 g−1) with an aqueous solution of tetraammineplatinum nitrate at 25 °C for 4 h. The impregnated support is
dried in ambient air at 125 °C and heated at 500 °C for 4 h. The obtained precursor is
reduced in H2/He (10% v/v) mixture at
500 °C for 2 h. 10 mg of the catalyst are then mixed with
1 g of silica encapsulated indium
nanoparticles, and the mixture is loaded into a quartz reactor to form a 1 cm column. A nickel–chromium
thermocouple of 1 mm diameter is inserted in the reactor
to measure the temperature of the catalyst. Before the
catalysis experiments, the reactor is heated at 120 °C in a flow of helium gas for 0.5 h. A mixture gas of He, O2, and methanol (87/9/4, p/p/p) at 100 ml min−1 is switched into the
reactor. Two on-line chromatograph systems are used to
analyze products: a Porapak Q packed column with thermal
conductivity detector for carbon dioxide and a methyl-silicone
capillary column with flame ionization detector for organic products.
Methyl methacrylate (MMA) monomer is polymerized
under isothermal batch condition in a 10 ml glass bottle at 80 °C. The reaction is
initiated by adding 0.1 g of azoisobutyronitrile
(AIBN) into 2 ml of MMA monomer. Different
amounts of silica encapsulated paraffin nanoparticles have been added to prevent thermal runaway. A
thermocouple is inserted into the solution, and the
reaction temperature is recorded by using an Agilent (34970A) data acquisition unit. The
molecular weight of polymer product is measured by
gel permeation chromatography (GPC) in tetrahydrofuran (THF).
Results and discussions
1. Structure and composition of silica
encapsulated indium
nanoparticles
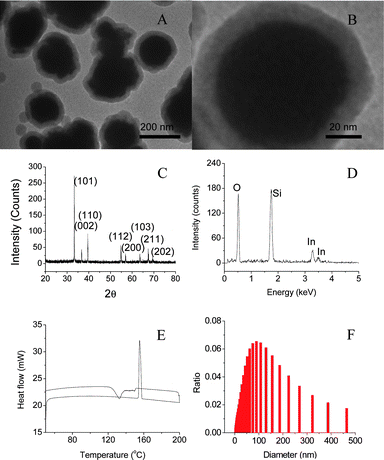
This direct boiling and sol–gel method can produce nano-PCMs of indium with core diameter of ∼150 nm and shell thickness of
10–20 nm as shown in Fig. 2A. High
resolution TEM image shows crystallized core and amorphous shell (Fig. 2B). XRD pattern shows
that body-centered tetragonal indium is formed in
amorphous silica shells (Fig.
2C). The composition of nanoparticles has been
confirmed by EDX analysis (Fig.
2D), where the signals of indium and
silicon can be identified clearly. Fig. 2E is the DSC curve of
nanoparticles collected at ramp rate of 10 °C
min−1, where the melting and solidifying of indium
nanoparticles occur at 156 and 130 °C, respectively.
Fig. 2F shows size distribution of the encapsulated
indium
nanoparticles, where the mean diameter of nanoparticles is determined as 150 nm by using dynamic light scattering. The core diameter can be controlled
in the range from 10 to 200 nm by changing reaction conditions including reaction time
and precursor concentration. The thickness of silica
shell has been controlled in a range of 10 and 30 nm by changing the molar ratio of
nanoparticle and TEOS.
2. Structure and composition of encapsulated paraffin nanoparticles
Interfacial polycondensation method has been used to
produce silica encapsulated polyethylene nanoparticles. Fig. 3A shows a
TEM image, where dark polyethylene core and light
silica shell can be seen clearly. The core
diameter and shell thickness are approximately 1000 and 500 nm, respectively. The mean
size of particles derived from DLS is about 2 µm (Fig.
3B). Fig. 3C shows Fourier transform infrared (FT-IR)
spectra of polyethylene and encapsulated polyethylene
nanoparticles, where the characteristic peaks of
polyethylene can be observed (Fig. 3C, red). The two
absorption peaks at 2916 and 2848 cm−1 are characteristic peaks of aliphatic
C–H stretching vibration; the peak at 1464 cm−1 is assigned to C–H bending
vibration; the peak at 721 cm−1 is associated with in-plane rocking vibration
of CH2
group. The absorption peaks at 941 and 3430
cm−1 are assigned to the stretching of hydroxyl
group in Si–OH and residual water and 1111 cm−1 is assigned to the
asymmetrical stretching of siloxane bond Si–O–Si.
The thermal property of nanoparticles has been studied by
DSC. Fig. 3D shows
DSC curves of polyethylene and silica encapsulated polyethylene nanoparticles. The melting and solidifying points of polyethylene are at 110
and 100 °C, respectively. The latent heats of fusion of paraffin and silica encapsulated paraffin nanoparticles are derived from DSC to be 200
and 160 J g−1, respectively, from which the ratio of PCM in core–shell
structure is determined to be approximately 80 wt%.
3. Preventing thermal runaway of catalytic reaction
using silica-encapsulated indium
nanoparticles
Encapsulated indium
nanoparticles are used to prevent thermal runaway of
methanol oxidation on supported platinum catalyst.
Fig. 4 shows the relation between measured temperature
and time after feeding methanol in vapor form, where
the temperature control unit is set at 120 °C. In the control experiment with 10 mg of
catalyst and 1 g silica as dilutor, the measured temperature quickly rises from 120 to 320
°C and remains constant at 320 °C due to strong heat liberation of the reaction. From
the distribution of products as listed in Table 1,
CO2 is the main product, suggesting burning
of methanol. Instead of using low thermal
conductivity silica (1.3 W m−1
K−1) as dilutor in catalyst bed, silicon carbide with high
thermal conductivity (∼360 W m−1
K−1) to conduct reaction heat has also
been used in the control experiment. However, for the strong exothermal reaction of
methanol oxidation catalyzed by Pt/SiO2, silicon
carbide cannot eliminate thermal runaway, and the highest and steady
temperatures reach 351 °C and 310 °C respectively. When 10 mg of catalyst and 1 g of nano-PCM are packed in the reactor, the reaction
temperature is constant at ∼120 °C, meaning effective control of thermal runaway.
Although the conversion is as low as 0.6% because of the low reaction temperature, the
trend of methanol
burning at initial stage of reaction is efficiently
controlled. The large latent heat of indium (28.52 J
g−1
K−1) takes heat from hot spots of
reaction. The ramp rate of temperature at the initial stage of runaway is dependent on
the relative mass ratio of nano-PCMs to Pt/SiO2
catalyst as shown in Fig.
4 inset. The highest ramp rate (29.5 °C min−1) is achieved in the
cases when there are no nano-PCMs (silica or
silicon carbide as dilutor). The ramp rate
decreases as the relative amounts of nano-PCMs increases. In the case of 30 mg catalyst and 1 g nano-PCMs, the temperature rises to 140 °C.
Increasing the catalyst to 50 mg and keeping PCMs at 1 g
leads to a further temperature increases to 165 °C, which is higher than the melting
temperature of indium (156 °C), and the temperature
of catalyst bed can still be controlled. It implies that
local hot spots can melt encapsulated indium
nanoparticles due to their high temperature, and the
phase change of indium
nanoparticles adsorbs reaction heat, quenches local hot
spots, and prevents thermal runaway of reaction.
 |
| | Fig. 4 Reaction time dependent temperature at different amount of nano-PCM and Pt/SiO2
catalyst: 0 g of nano-PCM and 10 mg of catalyst diluted in 1 g of SiO2 (circle) or 1 g
SiC (diamond);
1 g of nano-PCM and 50 mg of catalyst (down triangle),
1 g of nano-PCM and 30 mg of catalyst (up-triangle), 1
g of nano-PCM and 1 mg of catalyst (square). The ramp
rates at different amount of nano-PCM and catalyst
(inset). | |
| Samplesa |
Steady temp./°C |
Conv. (%) |
Selectivitya
(%) |
|
HCHO
|
MF
|
CO2 |
DMM
|
DMOH
|
|
In@SiO2 denotes silica encapsulated indium PCMs; MF, DMM and DMOH mean methyl
formate, dimethoxy methane
and methoxymethanol.
|
| 10 mg Pt/SiO2 1 g
SiO2 |
320 |
98.2 |
— |
12.1 |
83.2 |
4.7 |
— |
| 10 mg Pt/SiO2 1 g
SiC |
310 |
99.3 |
— |
11.2 |
87.7 |
1.1 |
— |
| 10 mg Pt/SiO2 1 g
In@SiO2 |
120 |
0.6 |
97.3 |
2.6 |
— |
— |
0.1 |
| 30 mg Pt/SiO2 1 g
In@SiO2 |
140 |
5.5 |
75.2 |
21.3 |
3.1 |
0.2 |
0.2 |
| 50 mg Pt/SiO2 1 g
In@SiO2 |
165 |
32.5 |
32.5 |
52.3 |
14.8 |
0.3 |
0.1 |
Controlling thermal runaway affects the distribution of the product as shown in
Table 1. The conversion of methanol on 10 mg catalyst and 1 g
silica is highest (98.2%), but the major product
is carbon dioxide. Adding nano-PCM reduces
conversion yield and amount of carbon dioxide. At
the same load of nano-PCMs (1 g), the conversion yield increases from 0.6 to 32.5% as
catalyst increases from 10 to 50 mg. The selectivity to
HCHO decreases from 97.3 to 32.5%, and
selectivity to methyl formate increases from 2.6 to
52.3%. Due to suppression of runaway, the selectivity to carbon dioxide in methanol oxidation is
reduced and the selectivity to partial oxidation
products increases. The selectivity to methyl
formate (MF) increases to 52.3% when
the amount of catalyst is 50 mg. In the mild condition,
the reaction rate is decreased, but the yield of methyl
formate is increased. Therefore, nano-PCMs can be used to control thermal
runaway and promote the yield of products of strong exothermic reaction.
4. Preventing thermal runaway of polymerization using
silica-encapsulated polyethylene nanoparticles
The polymerization of methyl
methacrylate (MMA) monomer is a strong exothermic reaction. DSC has been used as isothermal calorimeter to determine adiabatic thermal energy release from polymerization, which starts at 80 °C and reaches a maximum at
125 °C as shown in Fig. 5A (red curve). The
heat-releasing peak corresponds to reaction enthalpy of −1717.8 mJ for 3 mg of MMA. The
melting of polyethylene is endothermic with peak temperature of 110.3 °C and peak area
of 202.1 J g−1 (black curve). When 2.5 mg of silica encapsulated polyethylene nanoparticles containing 2 mg polyethylene is added into 2 mg MMA, the
exothermic heat decreases to 460.3 mJ (green curve). Doubling the amount of encapsulated
polyethylene nanoparticles to 5 mg while keeping amount
of MMA (2 mg) leads to reduction of heat to 94.2 mJ
(blue curve). Adding silica encapsulated
polyethylene nanoparticles in polymerization leads to reduction of
exothermic heat due to suppression of local hot spots. When the measured temperature is
between 80 and 105 °C, heat released from the polymerization is less than the fusion enthalpy of polyethylene, and a small
endothermic peak appears in green and blue curves. (The polyethylene melts due to hot
spots, even if the measured temperature is lower than its melting point.) When the
measured temperature is increased, the polymerization
rate is increased, and the released heat is absorbed by melting polyethylene. The
excessive heat gives a small exothermic peak.
 |
| | Fig. 5 DSC curves of polyethylene (black); adiabatic
polymerization of 2 mg MMA (red), 2 mg MMA with 2.5
mg (green) or 5.0 mg (blue) silica encapsulated
polyethylene nanoparticles; the temperature rises of 1
g MMA (black), 1 g MMA with 0.63 g (red) or 1.25 g (blue) silica encapsulated polyethylene nanoparticles when temperature is maintained at 80 °C (B); the molecular
weights of PMMA with (black) and without (blue) silica encapsulated polyethylene nanoparticles from gel permeation
chromatography (C); morphologies of products from three reactions, where
the images from left to right correspond to no nanoparticles (D), 0.63 (E) and 1.25 g (F) nanoparticles, respectively. | |
In order to further confirm the suppression of thermal runaway of polymerization, encapsulated polyethylene nanoparticles are added into monomer of methyl
methacrylate. The temperature is controlled at 80 °C in a water bath, and a thermocouple
is inserted into the monomer to record reaction temperature. Fig.
5B shows the magnitudes of measured temperature as functions of time. In the
absence of phase change nanoparticles, when polymerization is initiated, the measured temperature increases
to 100 °C and the temperature increases abruptly to 140 °C after 600 s, which is
presumably because of viscosity increase and thermal runaway. When 0.63 g of silica encapsulated polyethylene nanoparticles are added into monomer, the temperature increases to 120 °C
after 700 s. Adding 1.25 g of encapsulated polyethylene nanoparticles leads to smooth reaction, where the highest temperature
measured by thermocouple is 110 °C, which is close to the
melting point of polyethylene. The molecular weights of generated PMMAs are derived from
gel permeation chromatography. The permeation time of
PMMA made without silica encapsulated polyethylene
nanoparticles is 7.92 min, corresponding to molecular
weight of 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000; the permeation time of PMMA made with encapsulated polyethylene
particles is 8.25 min, corresponding to molecular weight 15
000; the permeation time of PMMA made with encapsulated polyethylene
particles is 8.25 min, corresponding to molecular weight 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 (Fig. 5C). It is understandable that polymer
made at high polymerization temperature normally has
higher molecular weight. Fig. 5D–F show the morphologies
of polymer produced in the three polymerization reactions. In the absence of polyethylene nanoparticles, polymerization is
more acute, which generates bubbles in polymethyl methacrylate (PMMA); adding
encapsulated polyethylene particles makes polymerization
stable.
800 (Fig. 5C). It is understandable that polymer
made at high polymerization temperature normally has
higher molecular weight. Fig. 5D–F show the morphologies
of polymer produced in the three polymerization reactions. In the absence of polyethylene nanoparticles, polymerization is
more acute, which generates bubbles in polymethyl methacrylate (PMMA); adding
encapsulated polyethylene particles makes polymerization
stable.
5. Thermophysical characteristics of encapsulated phase change nanoparticles
The effect of nano-PCM on preventing thermal runaway can be derived by comparing heat
capacities of silica particles and silica encapsulated indium
nanoparticles. The specific heat of silica is 0.7 J g−1
K−1, and the latent heat of indium is 28.52 J g−1. If the melting range of
indium
nanoparticles is taken as 10 °C (151 to 161 °C), the heat
that can be taken by 1 g of indium
nanoparticles (ignore contribution of shell) will be four
times the heat that can be taken by 1 g of silica in
the same temperature range. The heat capacity of 1 g of indium
nanoparticles will be ∼80 times higher than that of the
catalyst, meaning temperature rise of the catalyst due to hot spots can be effectively lowered to avoid
heat accumulation.
In order to exploit the latent heat of fusion, nano-PCMs have to melt rapidly during
reaction. The heat absorption of nanoparticles depends on
characteristics of heat conduction in nanoparticles,
which in turn depends upon particle size and material properties. The heat transfer from
environment into particles is determined by the difference between environment
temperature and surface temperature of particles.
| |  | (1) |
where
τ is the melting
time when the solid radius is
r,
ρl is the density of
nanoparticles, and
Ql is the latent heat of fusion of the
nanoparticles,
Ts and
Tm are the surface temperature and
melting point of
nanoparticles, respectively,
rp is the
nanoparticle radius
before melting, and
kl is thermal conductivity of
nanoparticles.
kIn and
kSiO2 are 81.8 and 1.3 W m
−1
K
−1, respectively,
rIn and
rSiO2 are 100 and 120 nm, respectively,
QIn is 28.52 J g
−1 and
ρIn is 7.3 g cm
−3. Replace all symbols
with numbers, and let
r equal to 0 nm, the
eqn
(1) is rewritten as
| | | τ(Ts − Tm) = 0.92 ×
10−7 s K | (2) |
Fig. 6A shows the melting time as the function of
temperature difference between surface temperature (hot spot) and melting temperature
for indium
nanoparticles. The melting time τ is 0.92 µs
when Ts − Tm = 0.1 K. In case of silica encapsulated polyethylene nanoparticles, kWax and kshell are
taken as 0.15 and 1.3 W m−1
K−1, rWax and
rshell are 500 nm and 1 µm, respectively, QWax
is 232 J g−1 and ρWax is 0.7 g cm−3. The
melting equation can be written as:
| | | τ(Ts − Tm) =
1.49 × 10−5 s K | (3) |
 |
| | Fig. 6 Calculated melting times of 200 nm indium
nanoparticles with 40 nm silica shell (A); melting times of 1 µm paraffin nanoparticles with 500 nm silica shell
(B); simulated temperature rises as functions of time when the mass ratio of silica and indium is
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 (square), 2 0 (square), 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 (circle), and 1 0 (circle), and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (triangle) (C); melting delay as a function
of rate constant (D). 1 (triangle) (C); melting delay as a function
of rate constant (D). | |
Fig. 6B shows the melting time as a function of
temperature difference for polyethylene nanoparticles.
The melting time of silica encapsulated paraffin is
148.7 µs when Ts − Tm = 0.1 K. Thus, the phase change nanoparticles can response rapidly to local hot spots to remove
thermal energy.
6. Kinetics of nano-PCM in prevention of thermal runaway
The contribution of phase change materials to prevent thermal runaway is studied for a
general reaction aA + bB ↔ cC, in which heat flow can be
expressed as30| |  | (4) |
where dH/dt is the instant heat flow of reaction at a
certain time,  is the corresponding
accumulated heat, ΔH is the total heat generation from the reaction, k is the rate
constant, C0,A and C0,B are
the initial concentrations of reactants, a, b, and c are
stoichiometric constants, m and n are orders of the reaction, and
Hpcm is the fusion enthalpy of phase change nanoparticles. For methanol oxidation,
m is 1 and n is 0, and eqn (4) is
thus simplified to:
is the corresponding
accumulated heat, ΔH is the total heat generation from the reaction, k is the rate
constant, C0,A and C0,B are
the initial concentrations of reactants, a, b, and c are
stoichiometric constants, m and n are orders of the reaction, and
Hpcm is the fusion enthalpy of phase change nanoparticles. For methanol oxidation,
m is 1 and n is 0, and eqn (4) is
thus simplified to:| |  | (5) |
Solving the differential eqn (5) yields:
| |  | (6) |
Integrating eqn (6) gives
| | | H =
ΔHC0[kt + exp
(−kt)] | (7) |
The released heat is absorbed by the catalyst, and phase
change nanoparticles, if the mass of the catalyst (10 mg) is negligible; the heat released from the
reaction is absorbed by silica and indium as a discrete function of temperature:
| |  | (8) |
The complete oxidation process of methanol generates water
and carbon dioxide:
| CH3OH + 3/2O2 = 2H2O +
CO2 |
The enthalpy change of the reaction is ΔH* = −676.5 J g−1.31 Since the amount of methanol that is oxidized is a function of time, the total heat generation from the reaction can be described as
ΔH = 0.064t J. Thus, the temperature of the system is written
as:
| |  | (9) |
where
C0 = 0.17,
CP,SiO2 = 0.7 J g
−1
K
−1,
CP,In = 0.23 J
g
−1
K
−1, Δ
HIn = 28.58 J
g
−1,
T0 = 120 °C,
Tm = 156.7 °C,
mSiO2
= 1 g, and
mIn = 1 g. Inputting
these values into
eqn (9) gives
| |  | (10) |
When the temperature reaches the melting point of indium, the temperature will be maintained until all indium melts. Then the temperature will continue to increase
as shown in eqn (11):
| |  | (11) |
where
k =
Aexp (−
Eact/
RT),
32 and
k is dependent on temperature and
activation energy.
32–34Fig. 6C shows the relation between temperature and time, where
the temperature of 1 g of silica increases faster
than that of 2 g of silica due to low thermal mass;
the temperature of 1 g of silica with 1 g of
indium
nanoparticles shows a 25 min delay. The length of delay
is dependent on the mass of
indium
nanoparticles and the rate constant of the reaction. If
the mass of
indium
nanoparticles is constant, the delay time will decrease
as the rate constant increases (
Fig. 6D). The delay time
does not change for log (
k) < −4 S
−1, but decreases rapidly from
43 to 3 min when log (
k) increases from −4 to −1 S
−1. For log
(
k) = 1 S
−1, the delay lasts 16 s.
The temperature rise delay demonstrated in these experiments efficiently restricts
thermal runaway of the catalytic reactions, and can
provide one additional application benefit—temperature stabilization of pulsed or
interrupted reactions. One broad technological application would be stabilization of
electrode temperatures inside rechargeable ionic
battery cells. All modern battery technologies
incorporate nanostructure surfaces of electrodes to increase ionic exchange areas and achieve a high
density of the electrical energy storage. With such
technologies, there is an increased risk of the temperature spikes during rapid charging
and discharging event, which may lead to the battery thermal runaway with significant
degradation of properties, as well as an occasional
catastrophic failure by explosion and fire. The discussed here encapsulated PCM nanoparticles can efficiently be applicable for battery
electrochemical reaction temperature runaway mitigation. Particle dielectric shell can
provide for a reduced interference with the electrical current flow, while the small
size will allow for a rapid response time and reliable response in multiple thermal
cycles expected during the battery life.
Conclusions
Adding encapsulated phase change nanoparticles into
exothermic reaction systems such as catalytic and polymerization reactions can effectively quench local hot spots,
prevent thermal runaway, and change product distribution. A rational design of
nano-heat-sinks with different core materials and shell materials can extend the use of
this method to a broad range of exothermic reactions. By using different phase change
materials, the operating range of nano-heat-sinks can be changed. In particular, silica encapsulated phase change nanoparticles can be used as new multifunctional catalyst support with thermal management capability, which is critical for
multiple chemical processes ranging from high quality polymer manufacturing to battery applications.
Acknowledgements
This work is supported by National Science Foundations (CTET 0828466) and Air Force
Research Laboratory (AFRL). Some of characterizations are done at Materials
Characterization Facilities (MCF) at the University of Central Florida (UCF).
References
- P. K. Shukla and S. Pushpavanam, Ind. Eng. Chem. Res., 1994, 33, 3202 CrossRef CAS.
- M. Surianarayanan, R. Vijayaraghavan, G. Swaminathan and P. G. Rao, Curr. Sci., 2001, 80, 738 CAS.
- T. Ohgushi, S. Komarneni and A. S. Bhalla, J. Porous Mater., 2001, 8, 23 CrossRef CAS.
- J. Verhoeff and P. J. Vandenberg, J. Therm. Anal., 1984, 29, 533 CrossRef CAS.
- A. N. R. Bos, L. Vandebeld, J. B. Overkamp and K. R. Westerterp, Chem. Eng. Commun., 1993, 121, 27 CrossRef CAS.
- V. Balakotaiah, D. Kodra and D. Nguyen, Chem. Eng. Sci., 1995, 50, 1149 CrossRef CAS.
- B. K. Mandal, A. K. Padhi, Z. Shi, S. Chakraborty and R. Filler, J. Power Sources, 2006, 161, 1341 CrossRef CAS.
- R. Vijayaraghavan, M. Surianarayanan, V. Armel, D. R. MacFarlane and V. P. Sridhar, Chem. Commun., 2009, 6297 RSC.
- J. Albert and G. Luft, Chem. Eng. Process., 1998, 37, 55 CrossRef CAS.
- A. Maider, R. L. Jose and M. A. Jose, Macromol. Mater. Eng., 2005, 290, 242 CrossRef.
- D. Vanhove, Appl. Catal., A, 1996, 138, 215 CrossRef CAS.
- R. Henda, A. Machac and B. Nilsson, Chem. Eng. J., 2008, 143, 195 CrossRef CAS.
- N. Frikha, E. Schaer and J. L. Houzelot, Thermochim. Acta, 2006, 449, 47 CrossRef CAS.
- E. Velo, C. M. Bosch and F. Recasens, Ind. Eng. Chem. Res., 1996, 35, 1288 CrossRef CAS.
- S. Bashir, T. Chovan, B. J. Masri, A. Mukherjee, A. Pant, S. Sen, P. Vijayaraghavan and J. M. Berty, Ind. Eng. Chem. Res., 1992, 31, 2164 CrossRef CAS.
- M. M. J. Quina and R. M. Q. Ferreira, Ind. Eng. Chem. Res., 1999, 38, 4615 CrossRef CAS.
- Y. Hong, S. Ding, W. Wu, J. Hu, A. A. Voevodin, L. Gschwender, E. Snyder, L. Chow and M. Su, ACS Appl. Mater. Interfaces, 2010, 2, 1685 Search PubMed.
- Z.-X. Gong and A. S. Mujumdar, Appl. Therm. Eng., 1996, 16, 807 CrossRef CAS.
- Z. H. Han, F. Y. Cao and B. Yang, Appl. Phys. Lett., 2008, 92, 243104 CrossRef.
- H. Marc, D. W. Randy, J. P. Stephen, M. P. Jason, M. Lou and C. Calvin, J. Electron. Packag., 2002, 124, 419 CrossRef.
- M. J. Vesligaj and C. H. Amon, IEEE Trans. Compon. Packag. Technol., 1999, 22, 541 CrossRef.
- S. Ahmet, K. Ali and K. Kamil, Int. J. Energy Res., 2008, 32, 154 CrossRef.
- H. W. Ryu, S. W. Woo, B. C. Shin and S. D. Kim, Sol. Energy Mater. Sol. Cells, 1992, 27, 161 CrossRef CAS.
- T. Yokota, M. Murayama and J. M. Howe, Phys. Rev. Lett., 2003, 91, 265504 CrossRef.
- W. Welnic, A. Pamungkas, R. Detemple, C. Steimer, S. Blugel and M. Wuttig, Nat. Mater., 2006, 5, 56 CrossRef CAS.
- A. Wan and C.-t. Yeh, Catal. Today, 2007, 129, 293 CrossRef CAS.
- R. Tesser, M. Di Serio and E. Santacesaria, Catal. Today, 2003, 77, 325 CrossRef CAS.
- C. Graf, D. L. J. Vossen, A. Imhof and A. V. Blaaderen, Langmuir, 2003, 19, 6693 CrossRef CAS.
- L. Ma, Y. Hong, Z. Ma, C. Kaittanis and J. M. Perez, Appl. Phys. Lett., 2009, 95, 043701 CrossRef.
- X.-R. Li and H. Koseki, J. Loss Prev. Process Ind., 2005, 18, 460 CrossRef.
- B. E. Traxel and K. L. Hohn, Appl. Catal., A, 2003, 244, 129 CrossRef CAS.
- J. W. Tester, P. A. Webley and H. R. Holgate, Ind. Eng. Chem. Res., 1993, 32, 236 CrossRef CAS.
- R. W. McCabe and D. F. McCready, J. Phys. Chem., 1986, 90, 1428 CrossRef CAS.
- K. W. Frese and C. Chen, J. Phys. Chem., 1995, 99, 6074 CrossRef CAS.
Footnote |
| † M. Zhang, Y. Hong and S. Ding have contributed equally to this
work |
|
| This journal is © The Royal Society of Chemistry 2010 |
Click here to see how this site uses Cookies. View our privacy policy here. 
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nanoparticle ratio.28,29
nanoparticle ratio.28,29



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000; the permeation time of PMMA made with encapsulated polyethylene
particles is 8.25 min, corresponding to molecular weight 15
000; the permeation time of PMMA made with encapsulated polyethylene
particles is 8.25 min, corresponding to molecular weight 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 (Fig. 5C). It is understandable that polymer
made at high polymerization temperature normally has
higher molecular weight. Fig. 5D–F show the morphologies
of polymer produced in the three polymerization reactions. In the absence of polyethylene nanoparticles, polymerization is
more acute, which generates bubbles in polymethyl methacrylate (PMMA); adding
encapsulated polyethylene particles makes polymerization
stable.
800 (Fig. 5C). It is understandable that polymer
made at high polymerization temperature normally has
higher molecular weight. Fig. 5D–F show the morphologies
of polymer produced in the three polymerization reactions. In the absence of polyethylene nanoparticles, polymerization is
more acute, which generates bubbles in polymethyl methacrylate (PMMA); adding
encapsulated polyethylene particles makes polymerization
stable.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 (square), 2
0 (square), 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 (circle), and 1
0 (circle), and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (triangle) (C); melting delay as a function
of rate constant (D).
1 (triangle) (C); melting delay as a function
of rate constant (D).
 is the corresponding
accumulated heat, ΔH is the total heat generation from the reaction, k is the rate
constant, C0,A and C0,B are
the initial concentrations of reactants, a, b, and c are
stoichiometric constants, m and n are orders of the reaction, and
Hpcm is the fusion enthalpy of phase change nanoparticles. For methanol oxidation,
m is 1 and n is 0, and eqn (4) is
thus simplified to:
is the corresponding
accumulated heat, ΔH is the total heat generation from the reaction, k is the rate
constant, C0,A and C0,B are
the initial concentrations of reactants, a, b, and c are
stoichiometric constants, m and n are orders of the reaction, and
Hpcm is the fusion enthalpy of phase change nanoparticles. For methanol oxidation,
m is 1 and n is 0, and eqn (4) is
thus simplified to:







