DOI:
10.1039/B822015H
(Paper)
J. Mater. Chem., 2009,
19, 2912-2920
Green nanotechnology from tea: phytochemicals in tea as building blocks for production of biocompatible gold nanoparticles†
Received
8th December 2008
, Accepted 5th February 2009
First published on 11th March 2009
Abstract
Phytochemicals occluded in tea have been extensively used as dietary supplements and as natural pharmaceuticals in the treatment of various diseases including human cancer. Results on the reduction capabilities of phytochemicals present in tea to reduce gold salts to the corresponding gold nanoparticles are presented in this paper. The phytochemicals present in tea serve a dual role as effective reducing agents to reduce gold and also as stabilizers to provide a robust coating on the gold nanoparticles in a single step. The tea-generated gold nanoparticles (T-AuNPs), have demonstrated remarkable in vitro stability in various buffers including saline, histidine, HSA, and cysteine solutions. T-AuNPs with phytochemical coatings have shown significant affinity toward prostate (PC-3) and breast (MCF-7) cancer cells. Results on the cellular internalization of T-AuNPs through endocytosis into the PC-3 and MCF-7 cells are presented. The generation of T-AuNPs follows all principles of green chemistry and T-AuNPs have been found to be non toxic as assessed through MTT assays. No ‘man made’ chemicals, other than gold salts, are used in this truly biogenic, green nanotechnological process thus paving the way for excellent opportunities for their application in molecular imaging and therapy.
Introduction
Beginning in the bygone era of 2700 BC, when the second emperor of China, Shen Nung, discovered tea, this beverage has become the most popular, soothing and delicious drink in human history.1 Throughout history and transitioning into the 21st century, tea drinking has been directly attributed to a plethora of health benefits.2,3 Several studies suggest that consumption of tea results in lowering the risk of stroke,4 reducing the risk of cancer,5–9 lowering blood pressure,9 enhancing immune function,10 and preventing dental cavities11 and gingivitis.3,12–24 The growing evidence towards the health benefits of tea has resulted in extensive studies to unravel the scientific basis of the healing and curing power of tea.3,4 A well-accepted scientific consensus emanating from several scientific investigations is that tea contains high levels of antioxidant polyphenols, including flavonoids, and catechins, all of which scavenge the dangerous free radicals in the body and thus, prevent the progress of various diseases.25–34 Polyphenolic flavonoids in tea (Fig. 1), of which epigallocatechin gallate (EGCG) is the second major constituent, have anticarcinogenic activity in vitro, which may support the results of epidemiological research on the correlation between drinking tea and the risk of morbidity from cancer.25–34EGCG scavenges superoxide anion radicals (O2˙), hydrogen peroxide (H2O2), hydroxyl radicals (HO˙), peroxyl radicals, singlet oxygen, and peroxynitrite.25–34 The one-electron reduction potential of EGCG under standard conditions is 550 mV, a value lower than that of glutathione (920 mV) and comparable to that of α-tocopherol (480 mV).29 While the tremendous health benefits of the ‘chemical cocktails’ occluded within tea (Fig. 1) are beyond doubt, the actual applications of the chemical reduction power of the myriad of chemicals present in tea is still in its infancy. We hypothesize that, the synergistic reduction potentials of polyphenols including flavonoids, catechins and various phytochemicals present in tea, will reduce gold salts to produce gold nanoparticles for potential applications in medicine and technology. Validation of this hypothesis will have long lasting positive repercussions in the chemical, electronic materials, health and hygiene industries, because such an approach will provide an ideal platform for the production of gold nanoparticles under 100% green chemistry conditions without the intervention of any ‘man-made’ chemicals following all principles of green chemistry. Gold nanoparticles are currently used in a wide spectrum of applications ranging from chemical catalysis to electronic materials design.35–46Gold nanoparticles have also found considerable prominence in the development of biological sensors and in the design and development of diagnostic and therapeutic nanomedicine products.13,35–49 As the nanorevolution, in the realms of medical and technological applications, unfolds, it is imperative to develop environmentally benign and biologically friendly green chemical processes.23,50–85 Naturally grown plants and various plant species which occlude phytochemicals may serve as long lasting and environmentally benign reservoirs for the production of a myriad of metallic nanoparticles. We, herein, present an unprecedented synthetic approach that involves the production of well-defined gold nanoparticles by the simple mixing of an aqueous solution of sodium tetrachloroaurate (NaAuCl4) with an aqueous solution of Darjeeling tea leaves. Production of gold nanoparticles (T-AuNPs 1–4) in this phytochemically-mediated process leads to completion at 25 °C within 30 minutes. Gold nanoparticles generated through this process do not agglomerate suggesting that the combination of thearubigins, theaflavins, catechins and various phytochemicals present in tea also serve as excellent stabilizers on nanoparticles and thus, provide robust shielding from agglomeration. Cellular uptake and cytotoxic studies of T-AuNPs were examined in Human Prostate (PC-3) and Breast cancer cells (MCF-7). The phytochemically coated T-AuNPs showed excellent affinity toward receptors on prostate and breast tumor cells. The details of the synergistic advantages of using tea in this green nanotechnological process for a dual role involving gold nanoparticle production and stabilization in a single process are discussed.
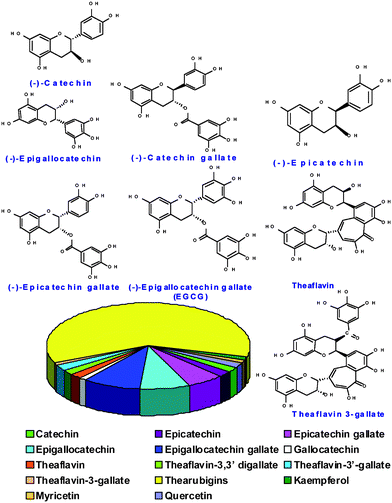 |
| | Fig. 1 Composition of various phytochemicals in black tea leaves. | |
Materials and methods
Chemicals
All chemicals and tea precursors used in the synthesis of gold nanoparticles (AuNPs) were procured from standard vendors: NaAuCl4 (Alfa-Aesar) and tea from organic grocery sources. Transmission electron microscope (TEM) images were obtained on a JEOL 1400 transmission electron microscope (TEM), JEOL Ltd, Tokyo, Japan. TEM samples were prepared by placing 5 µL of gold nanoparticle solution on a 300 mesh carbon-coated copper grid and allowing the solution to stand for five minutes; excess solution was removed carefully and the grid was allowed to dry for an additional five minutes. The average size and size distribution of the gold nanoparticles synthesized were determined by the processing of the TEM image using image processing software such as Adobe Photoshop (with Fovea plug-ins). The absorption measurements were done using Varian Cary 50 UV-Vis spectrophotometers with 1 mL of gold nanoparticle solution in disposable cuvettes of 10 mm path length.
Tea-initiated and -stabilized gold nanoparticles (T-AuNP-1)
To a 10 mL vial was added 6 mL of doubly ionized water (DI), followed by the addition of 100 mg of tea leaves (Darjeeling tea). The reaction mixture was stirred continuously at 25 °C for 15 min. To the stirring mixture was added 100 µL of 0.1 M NaAuCl4 solution (in DI water). The color of the mixture turned purple-red from pale yellow within 5 minutes of the addition, indicating the formation of gold nanoparticles. The reaction mixture was stirred for an additional 15 minutes. The gold nanoparticles thus formed were separated from residual tea leaves immediately using a 5 micron filter and were characterized by UV-Vis absorption spectroscopy and TEM analysis.
Tea-initiated and gum arabic-stabilized gold nanoparticles (T-AuNP-2)
To a 10 mL vial was added 0.012 g of gum arabic, 6 mL of doubly ionized water (DI), followed by the addition of 100 mg of tea leaves (Darjeeling tea). The reaction mixture was stirred continuously at 25 °C for 15 min. To the stirring mixture was added 100 µL of 0.1 M NaAuCl4 solution (in DI water). The color of the mixture turned purple-red from pale yellow within 10 minutes indicating the formation of gold nanoparticles. The reaction mixture was stirred for an additional 15 minutes. The gold nanoparticles thus formed were separated from residual tea leaves immediately using a 5 micron filter and were characterized by UV-Vis absorption spectroscopy and TEM.
Tea-initiated and -stabilized gold nanoparticles at 40 °C (T-AuNP-3)
To a 10 mL vial was added 6 mL of doubly ionized water (DI), followed by the addition of 100 mg of tea leaves (Darjeeling tea). The reaction mixture was stirred continuously at elevated temperature (∼40 °C) for 5 min. To the warm stirring mixture was added 100 µL of 0.1 M NaAuCl4 solution (in DI water). The color of the mixture turned purple-red from pale yellow instantly indicating the formation of gold nanoparticles. The reaction mixture was stirred for an additional 5 minutes. The gold nanoparticles in DI water were separated from residual tea leaves immediately using a 5 micron filter and were characterized by UV-absorption spectroscopy and TEM analysis.
Tea-initiated and gum arabic-stabilized gold nanoparticles at 40 °C (T-AuNP-4)
To a 10 mL vial was added 0.012 g of gum arabic, 6 mL of doubly ionized water (DI), followed by the addition of 100 mg of tea leaves (Darjeeling tea). The reaction mixture was stirred continuously at elevated temperature (∼ 40 °C) for 5 min. To the warm stirring mixture was added 100 µL of 0.1 M NaAuCl4 solution (in DI water). The color of the mixture turned purple-red from pale yellow in about 5–10 min indicating the formation of gold nanoparticles. The reaction mixture was stirred for 5 more minutes. The gold nanoparticles in DI water were separated immediately using a 5 micron filter. The tea/gum arabic stabilized gold nanoparticles (T-AuNP-4) were characterized by UV-absorption spectroscopy and TEM analysis.
In vitro stability studies of gold nanoparticles synthesized using tea leaves
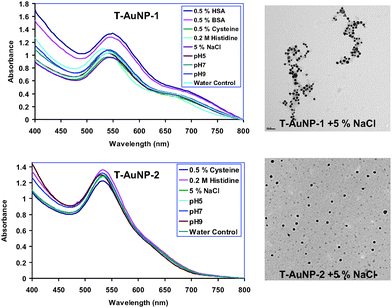
In vitro stabilities of the four different tea-mediated gold nanoparticles (T-AuNPs, 1–4) were tested in the presence of NaCl, cysteine, histidine, HSA and BSA solutions. Typically, 1 mL of gold nanoparticle solution was added to glass vials containing 0.5 mL of 5% NaCl, 0.5% cysteine, 0.2 M histidine, 0.5% HSA and 0.5% BSA solutions respectively and incubated for 30 min. The stability and the identity of gold nanoparticles (T-AuNPs 1–4) were measured by recording UV absorbance after 30 min. The plasmon resonance band at ∼535 nm confirmed the retention of nanoparticulates in all the above mixtures. TEM measurements inferred the retention of the nanoparticulate compositions in all the above four different gold nanoconstructs, signifying the robust nature of these nanoparticles under in vitro conditions.
Cell culture
Minimum essential medium (MEM with nonessential amino acids, powdered), HEPES, bovine insulin, streptomycin sulfate and penicillin-G, were obtained from Sigma Chemical Company (St. Louis, MO); all were “cell culture tested” when available. Bovine calf serum, phenol red (sodium salt), and lyophilized trypsin were obtained from Gibco BRL (Grand Island, NY). MCF-7 breast and PC-3 prostate cancer cells were obtained from ATCC. MCF-7 cells were maintained in MEM with nonessential amino acids, 10 pg ml−1phenol red, 10 mM HEPES, 6 ng ml−1insulin, 100 units ml−1penicillin, 100 pg ml−1streptomycin, and 5% charcoal-stripped calf serum (maintenance medium). PC-3 cells were maintained in RPMI medium supplemented with 4.5 g L−1D-glucose, 25 mM HEPES, 0.11 g L−1sodium pyruvate, 1.5 g L−1sodium bicarbonate, 2 mM L-glutamine and 10% FBS and antibiotics.
Cell internalization studies
About 16,000 cells were plated into each well in a 6 well plate and this plate was incubated at 37 °C for 24 h to allow the cells to recover. After the cells were recovered, the medium from each well was aspirated and fresh growth medium was added (about 4 mL per well). Cells were allowed to grow for 3 days and the media was changed every alternate day. On the 5th day, 25 micromolar T-AuNP-1 (Au atoms) solution was added to each well. After adding the sample, the plate was incubated for 4 h at 37 °C. Medium was aspirated from each well after 4 h and the cell layer was rinsed with CMFH–EDTA (Calcium-Magnesium-Free-Hark's + HEPES–EDTA) solution to remove all traces of serum. About 1 mL of trypsin–EDTA solution was added to each well and the cells were observed under an inverted microscope until the cell layer was dispersed. 4.0 mL of complete growth medium was added to each well and the cells were aspirated by gently pipetting. The cell suspension was transferred into a centrifuge tube and centrifuged at approximately 125 × g for 5 to 10 minutes. The cells were washed thoroughly with chilled PBS, pelleted by centrifugation and fixed with 0.1 M Na-cacodylate buffer containing 2% glutaraldehyde and 2% para-formaldehyde. The pellets were post fixed with 1% osmium tetraoxide, dehydrated and embedded in Epon/Spurr's resin and 80 nm sections were collected and placed on TEM grids followed by sequential counter staining with uranyl acetate and lead citrate. TEM grids were observed under TEM (JEOL 1400) and images were recorded at different magnifications.
Cytotoxicity evaluations
An MTT Cell Proliferation Assay kit was obtained from ATCC. For the cytotoxicity evaluation of these nanoparticles, MTT assay was done as described by the supplier. Briefly, 1 × 105 ml−1cells at the exponential growth phase were placed in a flat-bottomed 96-well polystyrene-coated plate and were incubated for 24 h in a CO2 incubator at 5% CO2 and 37 °C. A series of dilutions of 10, 25, 50, 100, and 150 µM of T-AuNP-1 were made in the medium. Each concentration was added to the plate in quadruplet. After 24 h of incubation, 10 µl/well MTT (stock solution 5 mg ml−1 PBS) was added for 6 h and the formazan crystals formed were dissolved in 100 µl detergent. The plates were read in a microplate reader (Dynastic MR 5000, USA) operating at 570 nm. Wells with complete medium, nanoparticles, and MTT, but without cells were used as blanks. All experiments were performed three times in quadruplet, and the average of all the experiments has been shown as a cell-viability percentage in comparison with the control experiment, while gold untreated controls were considered as 100% viable.
Results and discussion
As part of our ongoing efforts toward the design and development of biocompatible gold nanoparticles for subsequent use in medical applications, we have initiated studies on the direct intervention of phytochemicals for the production of gold nanoparticles. Our new process for the production of gold nanoparticles uses the direct interaction of sodium tetrachloaurate with black Darjeeling tea leaves in aqueous media (Scheme 1) without the intervention of any external man-made chemicals. Therefore, this reaction pathway satisfies all the conditions of a 100% green chemical process.86–88 The composition of various phytochemicals in black tea is outlined in Fig. 1. Gold nanoparticles produced by this process did not require any external chemicals for the stabilization of the nanoparticulate matrix. Phytochemicals present in tea (Fig. 1) are presumably responsible for the creation of a robust coating on gold nanoparticles and thus, rendering the nanoparticles stable against agglomeration.
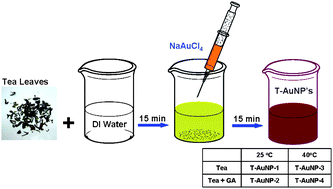 |
| | Scheme 1 Synthesis of T-AuNPs from black Darjeeling tea leaves. | |
Absorption measurements indicated that the plasmon resonance wavelength, λmax of T-AuNPs is ∼535 nm. The sizes of T-AuNPs are in the range of 15–42 nm as measured by TEM techniques (Fig. 2). The phenolics and other phytochemicals within tea (Fig. 1) not only result in effective reduction of gold salts to nanoparticles but their chemical framework is also effective at wrapping around the gold nanoparticles to provide excellent robustness against agglomeration. The current discovery of the unique chemical power of phytochemicals in tea in initiating nanoparticle formation is of paramount importance in the context of the production of gold nanoparticles for medical and technological applications under non-toxic conditions.23,55,57–61 One of the paramount prerequisites of utilizing AuNPs for in vivo imaging and therapy applications is that the nanoparticles be produced and stabilized in biologically benign media.23,55,57–61,49,89 With the currently available methods of producing AuNPs, it is often necessary to remove unreacted toxic chemicals and byproducts. Typical known methods of making gold nanoparticles utilize harsh conditions, such as the application of sodium borohydride to reduce AuCl4−.90,91 Although such processes lead to the efficient production of gold nanoparticles, the presence of sodium borohydride, even in trace amounts, is unsuitable for use in biomedical applications of gold nanoparticles. The high reduction capabilities of sodium borohydride result in reduction of biogenic chemical functionalities present on peptide backbones, thus either reducing or eliminating the biospecificity of biomolecules. Normally, thiol-containing organic compounds are employed to stabilize AuNPs against agglomeration.91Thiol–gold interaction is strong and makes gold nanoparticles highly stable. Therefore, such AuNPs once stabilized by thiols cannot be further conjugated to useful drug moieties including peptides, proteins and various biochemical vectors that are normally used to target diagnostic and therapeutic gold nanoparticles onto tumors and various disease sites in the body. This means that the thiol-stabilized AuNPs will have limited applicability in the development of AuNP-labeled biomolecules for use in the design of target-specific nanoscale imaging or therapeutic agents. Other methods that have been described in the literature utilize a cocktail of chemicals in their production processes. Such techniques are not environmentally friendly and have many drawbacks that impede the efficient utilization of AuNPs in biomedical applications.
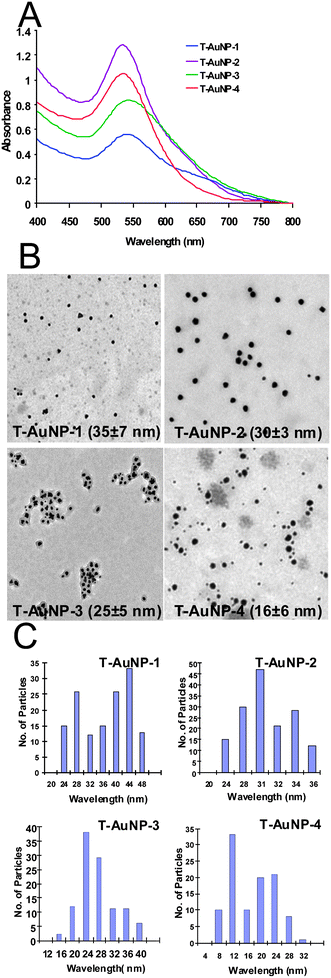 |
| | Fig. 2 (A) UV-visible absorption spectra, (B) TEM images and (C) size distribution histograms of T-AuNPs. | |
Nanoparticle size characterization and size distribution
Physicochemical properties, such as size, charge, and morphology of gold nanoparticles generated using aqueous solutions of tea leaves, were determined by three independent techniques viz.transmission electron microscopy (TEM), differential centrifugal sedimentation (DCS, Disc Centrifuge, CPS Instruments), and dynamic light scattering (DLS). TEM and DCS were used to determine the core size of gold nanoparticles and DLS was used to evaluate the size of tea phytochemical-coated gold.
Size and morphology.
TEM measurements on T-AuNPs synthesized using tea leaves show that the particles are spherical in shape within the size range of 15–45 nm (Table 1). Size distribution analysis of T-AuNPs confirms that particles are well dispersed (Fig. 2 and Table 1). DCS technique measures the sizes of nanoparticles by determining the time required for nanoparticles to traverse a sucrose density gradient created in a disc centrifuge. Both the techniques, TEM and DCS, provide sizes of metallic-gold cores. Gold nanoparticulate sizes measured by TEM and DCS, are in good agreement and are in the range of 15–45 nm (Fig. 2 and Table 1). Dynamic light scattering method was employed to calculate the sizes of gold nanoparticles coated with phytochemicals (hydrodynamic radius). The tea phytochemical coatings on gold nanoparticles are expected to cause substantial changes in the hydrodynamic radii of T-AuNPs. The hydrodynamic diameters of T-AuNP-1 and T-AuNP-2 as determined from DLS measurements gave values of 105 ± 1 and 165 ± 1 respectively, suggesting that tea phytochemicals (catechins, theaflavins and thearibigins) are capped on gold nanoparticles. The measurement of charge on nanoparticles, zeta potential (ζ), provides crucial information on the stability of nanoparticle dispersion. The magnitude of the measured zeta potential is an indication of the repulsive forces that are present and can be used to predict the long-term stability of the nanoparticulate dispersion. The stability of nanoparticulate dispersion depends upon the balance of the repulsive and attractive forces that exist between nanoparticles as they approach one another. If all the particles have a mutual repulsion then the dispersion will remain stable. However, little or no repulsion between particles, lead to aggregation. The negative zeta potential of −32 ± 1 and −25 ± 1 for T-AuNP-1 and T-AuNP-2 indicates that the particles repel each other and there is no tendency for the particles to aggregate (Table 1 and Fig. 2).
Table 1 Physicochemical data parameters of T-AuNPs
| Sample |
TEM
|
DLSa |
CPS |
Zeta potential/mV |
|
Hydrodynamic diameter.
|
|
T-AuNP-1
|
35 ± 7 |
105 ± 1 |
28 ± 1 |
−32 ± 1 |
|
T-AuNP-2
|
30 ± 3 |
165 ± 1 |
25 ± 1 |
−25 ± 1 |
Role of tea phytochemicals
Synthetic conditions have been optimized for the quantitative large scale conversions of NaAuCl4 to the corresponding AuNPs using tea leaves. Specific details on the nature and chemical roles of different phytochemicals in tea leaves responsible for the production of T-AuNPs are summarized in the following sections. The main phytochemicals present in black tea leaves consist of water soluble catechins (catechin, epicatechin, epicatechin gallate, epigallocatechin, epigallocatechin gallateetc.,), theaflavins (theaflavin, theaflavin 3-gallate, theaflavin 3′-gallate, theaflavin 3,3′-gallateetc.,) and Thearubigins which are oligomers of catechins of unknown structure. Generation of T-AuNPS using tea leaves involves aqueous media. Therefore, water soluble phytochemicals of tea (Fig. 1) may be playing a major role in the overall reduction reactions of NaAuCl4. We have systematically investigated the roles of catechins and theaflavins for the generation and stabilization of AuNPs through independent experiments.
(i) Role of catechins.
In order to understand the critical roles of the various catechins present in black tea leaves on the overall reduction of NaAuCl4 to the corresponding gold nanoparticles, we have performed a series of independent experiments using directly the commercially available family of catechins which include: catechin, epicatechin, epicatechin gallate, catechin gallate, epigallocatechin, and epigallocatechin gallate (see ESI†). Results of our experiments using commercially available catechins have unambiguously confirmed that catechins are excellent reducing and stabilizing agents to reduce Au(III) to the corresponding gold nanoparticles. These reactions proceeded to completion within 30 min. Absorption measurements indicated that the plasmon resonance wavelength, λmax, for all the T-AuNPs is ∼530 nm (Fig. 3). The sizes of the T-AuNPs were found to be in the 15–42 nm range as measured from the TEM images. (Fig. 3). The gold nanoaparticles obtained using catechin, and epigallocatechin gallate (EGCG) showed excellent stability which was confirmed by their in vitro stability studies. However, gold nanoparticles generated using epigallocatechin and epicatechin displayed minimum stability as shown by characteristic broad plasmon bands (Fig. 3A). Our studies have unequivocally confirmed that epigallocatechin and epicatechin are very effective in reducing tetrachloroaurate salt to the corresponding AuNPs. However, these phytochemicals failed to provide effective coating to shield the nanoparticles from agglomeration. In order to capitalize on the reduction powers of epigallocatechin and epicatechin, we have utilized gum arabic (a glycoprotein) as a naturally available stabilizing agent in our reactions. Results from these experiments have revealed that all the catechins (catechin, epicatechin, epicatechin gallate, epigallocatechin, eatechin gallate and epigallocatechin gallate) act as excellent reducing agents to reduce the Au(III) to the corresponding gold nanoparticles. The nanoparticles thus generated were coated with gum arabic stabilizing agent and showed significant stability when challenged with various salts and serum proteins (Fig. 5). These experiments have unambiguously confirmed that catechin and epigallocatechin gallate (EGCG) serve dual roles as reduction and stabilizing agents whereas epigallocatechin and epicatechin, can be used only for the reduction of gold salts and require gum arabic as an external stabilizing agent.
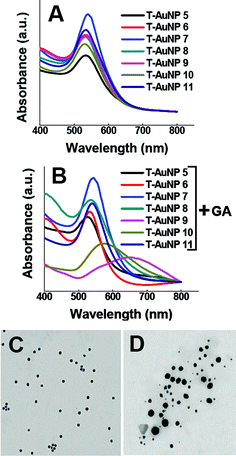 |
| | Fig. 3
UV-Vis
absorption spectrum of gold nanoparticles generated using (A) commercially available phytochemicals from tea, (B) commercially available phytochemicals from tea and gum arabic. T-AuNP-5 to T-AuNP-11 correspond to the gold nanoparticles generated using thiaflavins, epicatechin gallate, catechin, catechin gallate, epicatechin, epigallocatechin and EGCG respectively. TEM images of gold nanoparticles obtained using (C) catechin only (D) epigallocatechin gallate only. | |
(ii) Role of theaflavins.
We have also investigated the role of theaflavins in the generation of gold nanoparticles. Commercially available tea extract (Sigma) which contains >80% of theaflavins was used in these experiments (see ESI†). Addition of aqueous solution of NaAuCl4 to aqueous solution of theaflavin at 25 °C resulted in the formation of purple colored solutions within 30 minutes. The gold nanoparticles thus obtained by using theaflavin were characterized by UV-Vis absorption spectroscopy and TEM. A plasmon resonance band at ∼535 nm indicated the formation of gold nanoparticles (Fig. 3). TEM measurements confirmed the size distribution of gold nanoparticles. Detailed in vitro stabilities of the gold nanoparticles confirmed that the nanoparticles are extremely stable under various conditions. These results convincingly demonstrate the dual reduction and stabilizing capabilities of mixtures of theaflavins present in tea. The reservoir of non-toxic phytochemicals in tea (Fig. 1 and Fig. 4) is pivotal as they serve as a source of non-toxic reducing agents with capabilities for in vivo administration in situations that require the generation of gold nanoparticles under in vivo conditions.
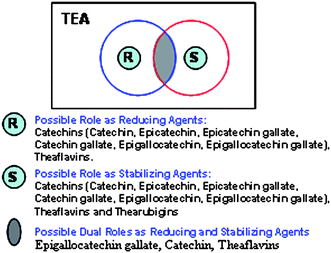 |
| | Fig. 4 Venn diagram showing the possible role of phytochemicals in tea for the generation and stabilization of gold nanoparticles. | |
It is remarkable to note that intervention of a second green component, in the form of gum arabic (GA), in the above reactions provides additional advantages. The use of GA along with tea leaves resulted in an increase in the optical density (absorbance) in the UV-Vis spectra of reaction mixtures (Fig. 2).51 This observation demonstrates that GA may be presumably serving as a biochemical platform to drive such reactions to completion with the consequent production of well-defined and uniform spherical gold nanoparticles. We have also investigated the effect of temperature on the formation of gold nanoparticles using only tea leaves as well as a combination of tea leaves and GA. Results of these reactions performed at 40 °C have revealed that nanoparticle formation at elevated temperatures results in randomly distributed spherical gold nanoparticles of sizes varying from 15–30 nm (Fig. 2).
In vitro stability studies
An issue of critical importance for in vivo molecular imaging applications is the stability of AuNPs over a reasonable time period. The stability of T-AuNPs was evaluated by monitoring the plasmon (λmax) in 0.5% cysteine, 0.2 M histidine, 0.5% Human Serum Albumin (HSA), 0.5% Bovine Serum Albumin (BSA) or 5% NaCl solutions over 30 min. We have also investigated the stability of T-AuNPs at pH 5, 7 and pH 9 phosphate buffer solutions. The plasmon wavelengths in all the above formulations show minimal shifts of ∼1–5 nm. Our results from these in vitro stability studies have confirmed that the AuNPs are intact and thus, demonstrate excellent in vitro stability of T-AuNPs in biological fluids at physiological pH (Fig. 5). For various biomedical applications which require lower concentrations of AuNPs, it is vitally important that dilutions of AuNP solutions do not alter their characteristic chemical and photophysical properties. We have undertaken a detailed investigation to ascertain the effect of dilution on the stability of T-AuNP-1. In order to establish the stability of T-AuNPs under dilution, the plasmon resonance wavelength (λmax) was monitored after every successive addition of 0.1 mL of doubly ionized (DI) water to 1 mL of AuNP solutions. The absorption intensity at λmax is found to be linearly dependent on the concentration of AuNPs, in accordance with the Beer–Lambert law. It is important to recognize that λmax of the AuNPs did not change at very dilute conditions (Fig S1†). These are typical concentrations encountered when working at cellular levels.
It is conceivable that the cocktail of phytochemicals in tea along with non-toxic phytochemical gum arabic (Fig. 1) are acting synergistically in stabilizing gold nanoparticles from any agglomeration in solution. It is also remarkable that this ‘nano-compatible’ structural motif of phytochemicals in tea offers stability to gold nanoparticles in aqueous media for over a month. These results suggest that the green nanotechnological process reported herein provides both the production and stabilization processes under mild conditions without the intervention of any man made harsh chemicals.
Cellular interactions of T-AuNPs
Cellular internalization studies of gold nanoparticle solutions provide insights into cellular uptake and such information will enhance the scope of gold nanoparticles in biomedicine. Gold nanoparticles are currently investigated for their potential applications as diagnostic/therapeutic agents, therapeutic delivery vectors, and intracellular imaging agents.92–97 Selective cell and nuclear targeting of gold nanoparticles will provide new pathways for the site specific delivery of gold nanoparticles as diagnostic/therapeutic agents. A number of studies have demonstrated that phytochemicals in tea have the ability to penetrate the cell membrane and internalize within the cellular matrix.98–101 Cancer cells are highly metabolic and porous in nature and are known to internalize solutes rapidly compared to normal cells.101–103 Therefore, we hypothesized that tea-derived phytochemicals, if coated on gold nanoparticles, would show internalization within cancer cells. We undertook cellular interaction and uptake studies via incubation of aliquots of T-AuNP-1 with prostate (PC-3) and breast cancer (MCF-7) cells. TEM images of prostate (PC-3) and breast tumor (MCF-7) cells post treated with T-AuNP-1 unequivocally validated our hypothesis. Significant internalization of nanoparticlesviaendocytosis within the MCF-7 and PC-3 cells was observed (Fig. 6). The internalization of nanoparticles within cells could occur via processes including phagocytosis, fluid-phase endocytosis, and receptor-mediated endocytosis. The viability of both PC-3 and MCF-7 cells post internalization of T-AuNP-1 suggests that the phytochemical coating renders the nanoparticles to be non toxic to cells. Such internalization of gold nanoparticles, keeping the cellular machinery intact, will provide new opportunities for probing cellular processesvia nanoparticulate-mediated imaging.
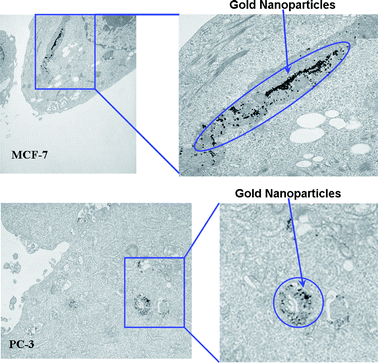 |
| | Fig. 6
TEM images showing endocytosis of T-AuNP-1 in cells. | |
Cytotoxicity studies
The cytotoxicity of T-AuNP-1 under in vitro conditions in prostate (PC-3) and breast (MCF-7) cancer cells was examined in terms of the effect of gold nanoparticles on cell proliferation by the MTT assay. Untreated cells as well as cells treated with 10, 25, 50, 100, and 150 µM concentrations of gold nanoparticles for 24 h were subjected to the MTT assay for cell viability determination. In this assay, only cells that are viable after 24 h exposure to the sample are capable of metabolizing a dye (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) efficiently and produce purple coloured crystals which are dissolved in a detergent and analyzed sphectrophotometrically. After 24 h of post treatment, PC-3 and MCF-7 cells showed excellent viability even up to 150 µM concentrations of T-AuNP-1 (Fig. 7). These results clearly demonstrate that the phytochemicals within tea provide a non-toxic coating on gold nanoparticles and corroborate the results as seen in the internalization studies discussed above. It is also important to recognize that a vast majority of gold (I) and gold (III) compounds exhibit varying degrees of cytotoxicity to a variety of cells.104,105 The lack of any noticeable toxicity of T-AuNP-1 provides new opportunities for the safe delivery and applications of such nanoparticles in molecular imaging and therapy.
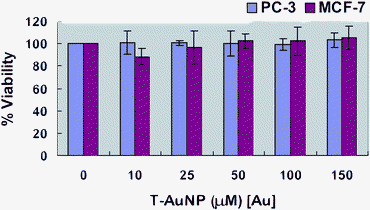 |
| | Fig. 7 Dose dependent MTT cytotoxicity assay of T-AuNP-1 in MCF-7 breast cancer cells and PC-3 prostate cancer cells. | |
Conclusions
The unique chemical, physical, photophysical, topological and radiological properties rendered by nanoparticulate gold (and of other metals/non metals) will continue to unravel a new knowledge base to invent a plethora of new technologies and products for medical, civilian, defense, environmental and space exploration applications. Over the next decade, advances in nanomedicine will likely impact all of us. Although there is no question of the scientific power and the positive impact of nanoscience and nanotechnology in transforming medical diagnosis and therapy, the potential toxic side effects of nanoparticles administered via intravenous or oral pathways cannot be discounted. Therefore, concerted efforts must be invested in the development of non-toxic nanoparticles for utility in a wide spectrum of applications. The studies reported in this paper serve as a unique example of the kinetic propensity of phytochemicals, present in tea, to reduce gold metal at micro or picomolar/sub-nanomolar concentrations to the corresponding gold nanoparticles. The versatile phytochemical-mediated green nanotechnological process has been shown to be effective in both the generation and stabilization of non-toxic gold nanoparticles for direct applications in a myriad of diagnostic and therapeutic applications. Occlusion of cancer fighting phytochemicals in various plant species and their future utility in the development of tumor specific gold nanoparticles will provide unprecedented opportunities toward the design and development of functional gold nanoparticles that can be safely produced, stored and shipped world wide.
Acknowledgements
This work has been supported by generous support from the National Institutes of Health/National Cancer Institute under the Cancer Nanotechnology Platform program (grant number: 5R01CA119412-01), NIH – 1R21CA128460-01 and University of Missouri-Research Board – Program C8761 RB 06-030.
References
- D. L. McKay and J. B. Blumberg, J. Am. Coll. Nutr., 2002, 21, 1–13 Search PubMed.
- A. Amin and M. Buratovich, Recent Pat. Anti-Cancer Drug Discovery, 2007, 2, 109–117 Search PubMed.
- M. Friedman, Mol. Nutr. Food Res., 2007, 51, 116–134 CrossRef CAS.
- M. L. Fraser, G. S. Mok and A. H. Lee, Complement. Ther. Med., 2007, 15, 46–53 CrossRef.
- R. L. Thangapazham, A. K. Singh, A. Sharma, J. Warren, J. P. Gaddipati and R. K. Maheshwari, Cancer Lett., 2007, 245, 232–241 CrossRef CAS.
- T. D. Way, H. H. Lee, M. C. Kao and J. K. Lin, Eur. J. Cancer., 2004, 40, 2165–2174 CrossRef CAS.
- J. M. Yuan, Y. T. Gao, M. J. Lee, C. S. Yang, R. K. Ross and M. C. Yu, Am. J. Epidemiol., 2006, 163, S107–S107.
- C. S. Yang, J. D. Lambert, J. Ju, G. Lu and S. Sang, Toxicol. Appl. Pharmacol., 2007, 224, 265–273 CrossRef CAS.
- J. M. Hodgson, Clin. Exp. Pharmacol. Physiol., 2006, 33, 838–841 CrossRef CAS.
- M. Hamer, Nutr. Res., 2007, 27, 373–379 CrossRef CAS.
- J. Wang, N. D. Zhou, Z. Q. Zhu, J. Y. Huang and G. X. Li, Anal. Bioanal. Chem., 2007, 388, 1199–1205 CrossRef CAS.
- T. P. T. Cushnie and A. J. Lamb, Int. J. Antimicrob. Agents, 2005, 26, 343–356 CrossRef CAS.
- T. A. Gasiewicz, C. Palermo and C. Westlake, J. Nutr., 2005, 135, 3033s–3034s.
- T. Hatano, M. Kusuda, M. Hori, S. Shiota, T. Tsuchiya and T. Yoshida, Planta Med., 2003, 69, 984–989 CrossRef CAS.
- Z. Q. Hu, W. H. Zhao, Y. Yoda, N. Asano, Y. Hara and T. Shimamura, J. Antimicrob. Chemother., 2002, 50, 1051–1054 CrossRef CAS.
- Y. K. Lee, N. D. Bone, A. K. Strege, T. D. Shanafelt, D. F. Jelinek and N. E. Kay, Blood, 2004, 104, 788–794 CrossRef CAS.
- Y. S. Lee, C. H. Han, S. H. Kang, S. J. Lee, S. W. Kim, O. R. Shin, Y. C. Sim, S. J. Lee and Y. H. Cho, Int. J. Urol., 2005, 12, 383–389 CrossRef.
- G. Simonetti, N. Simonetti and A. Villa, J. Chemother., 2004, 16, 122–127 Search PubMed.
- V. Stangl, M. Lorenz and K. Stangl, Mol. Nutr. Food Res., 2006, 50, 218–228 CrossRef CAS.
- P. D. Stapleton, S. Shah, J. C. Anderson, Y. Hara, J. M. T. Hamilton-Miller and P. W. Taylor, Int. J. Antimicrob. Agents, 2004, 23, 462–467 CrossRef CAS.
- P. D. Stapleton, S. Shah, Y. Hara and P. W. Taylor, Antimicrob. Agents Chemother., 2006, 50, 752–755 CrossRef CAS.
- R. P. Tiwari, S. K. Bharti, H. D. Kaur, R. P. Dikshit and G. S. Hoondal, Indian J. Med. Res., 2005, 122, 80–84 Search PubMed.
- G. Zhang, B. Keita, A. Dolbecq, P. Mialane, F. Secheresse, F. Miserque and L. Nadjo, Chem. Mater., 2007, 19, 5821–5823 CrossRef.
- W. H. Zhao, Z. Q. Hu, S. Okubo, Y. Hara and T. Shimamura, Antimicrob. Agents Chemother., 2001, 45, 1737–1742 CrossRef CAS.
- D. A. Balentine, S. A. Wiseman and L. C. M. Bouwens, Crit. Rev. Food Sci., 1997, 37, 693–704 CrossRef CAS.
- G. H. Cao, E. Sofic and R. L. Prior, J. Agric. Food Chem., 1996, 44, 3426–3431 CrossRef CAS.
- C. Lozano, L. Julia, A. Jimenez, S. Tourino, J. J. Centelles, M. Cascante and J. L. Torres, FEBS J., 2006, 273, 2475–2486 CrossRef CAS.
- W. Luczaj and E. Skrzydlewska, Prev. Med., 2005, 40, 910–918 CrossRef CAS.
- K. Maeta, W. Nomura, Y. Takatsume, S. Izawa and Y. Inoue, Appl. Environ. Microbiol., 2007, 73, 572–580 CAS.
- J. Peterson, J. Dwyer, S. Bhagwat, D. Haytowitz, J. Holden, A. L. Eldridge, G. Beecher and J. Aladesanmi, J. Food Compos. Anal., 2005, 18, 487–501 CrossRef CAS.
- A. Rietveld and S. Wiseman, J. Nutr., 2003, 133, 3285s–3292s CAS.
- F. Sari, N. Turkmen, G. Polat and Y. S. Velioglu, Food Sci. Technol. Res., 2007, 13, 265–269 Search PubMed.
- E. Satoh, N. Tohyama and M. Nishimura, J. Pharmacol. Sci., 2005, 97, 220p–220p.
- B. L. Zhao, J. Clin. Biochem. Nutr., 2006, 38, 59–68 Search PubMed.
- G. Han, P. Ghosh and V. M. Rotello, Nanomedicine (London, U. K.), 2007, 2, 113–123 Search PubMed.
- X. H. Huang, P. K. Jain, I. H. El-Sayed and M. A. El-Sayed, Nanomedicine (London, U. K.), 2007, 2, 681–693 Search PubMed.
- K. K. Jain, Clin. Chem., 2007, 53, 2002–2009 CrossRef CAS.
- H. W. Liao, C. L. Nehl and J. H. Hafner, Nanomedicine (London, U. K.), 2006, 1, 201–208 Search PubMed.
- D. Boyer, P. Tamarat, A. Maali, B. Lounis and M. Orrit, Science, 2002, 297, 1160–1163 CrossRef CAS.
- L. Cognet, C. Tardin, D. Boyer, D. Choquet, P. Tamarat and B. Lounis, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 11350–11355 CrossRef CAS.
- A. G. Cuenca, H. B. Jiang, S. N. Hochwald, M. Delano, W. G. Cance and S. R. Grobmyer, Cancer, 2006, 107, 459–466 CrossRef CAS.
- J. F. Hainfeld, D. N. Slatkin, T. M. Focella and H. M. Smilowitz, Br. J. Radiol., 2006, 79, 248–253 Search PubMed.
- M. Hu, J. Y. Chen, Z. Y. Li, L. Au, G. V. Hartland, X. D. Li, M. Marquez and Y. N. Xia, Chem. Soc. Rev., 2006, 35, 1084–1094 RSC.
- X. H. Huang, I. H. El-Sayed, W. Qian and M. A. El-Sayed, J. Am. Chem. Soc., 2006, 128, 2115–2120 CrossRef CAS.
- S. M. Moghimi, A. C. Hunter and J. C. Murray, FASEB J., 2005, 19, 311–330 CrossRef CAS.
- X. M. Qian, X. H. Peng, D. O. Ansari, Q. Yin-Goen, G. Z. Chen, D. M. Shin, L. Yang, A. N. Young, M. D. Wang and S. M. Nie, Nat. Biotechnol., 2008, 26, 83–90 CrossRef CAS.
- R. Kannan, V. Rahing, C. Cutler, R. Pandrapragada, K. K. Katti, V. Kattumuri, J. D. Robertson, S. J. Casteel, S. Jurisson, C. Smith, E. Boote and K. V. Katti, J. Am. Chem. Soc., 2006, 128, 11342–11343 CrossRef CAS.
- V. Kattumuri, M. Chandrasekhar, S. Guha, K. Raghuraman, K. V. Katti, K. Ghosh and R. J. Patel, Appl. Phys. Lett., 2006, 88 Search PubMed -.
- V. Kattumuri, K. Katti, S. Bhaskaran, E. J. Boote, S. W. Casteel, G. M. Fent, D. J. Robertson, M. Chandrasekhar, R. Kannan and K. V. Katti, Small, 2007, 3, 333–341 CrossRef CAS.
- R. Zsigmondy, Z. Anorg. Allg. Chem., 1916, 96, 265–288 CrossRef.
- R. Zsigmondy, Z. Elektrochem. Angew. Phys. Chem., 1916, 22, 102–104.
- S. S. Shankar, A. Ahmad and M. Sastry, Biotechnol. Prog., 2003, 19, 1627–1631 CrossRef CAS.
- S. S. Shankar, S. Bhargava and M. Sastry, J. Nanosci. Nanotechnol., 2005, 5, 1721–1727 CrossRef CAS.
- S. S. Shankar, A. Rai, A. Ahmad and M. Sastry, J. Colloid Interface Sci., 2004, 275, 496–502 CrossRef CAS.
- S. S. Shankar, A. Rai, B. Ankamwar, A. Singh, A. Ahmad and M. Sastry, Nat. Mater., 2004, 3, 482–488 CrossRef CAS.
- Z. J. Wang, Q. X. Zhang, D. Kuehner, A. Ivaska and L. Niu, Green Chem., 2008, 10, 907–909 RSC.
- I. A. Siddiqui, V. M. Adhami, D. J. Bharali, B. B. Hafeez, M. Asim, S. I. Khwaja, N. Ahmad, H. Cui, S. A. Mousa and H. Mukhtar, Cancer Res., 2009, 69, 1712–1716 CrossRef CAS.
- J. L. Huang, Q. B. Li, D. H. Sun, Y. H. Lu, Y. B. Su, X. Yang, H. X. Wang, Y. P. Wang, W. Y. Shao, N. He, J. Q. Hong and C. X. Chen, Nanotechnology, 2007, 18, 105104–105114 CrossRef.
- P. Mohanpuria, N. K. Rana and S. K. Yadav, J. Nanoparticle Res., 2008, 10, 507–517 CrossRef CAS.
- P. Raveendran, J. Fu and S. L. Wallen, J. Am. Chem. Soc., 2003, 125, 13940–13941 CrossRef CAS.
- N. Vigneshwaran, R. P. Nachane, R. H. Balasubramanya and P. V. Varadarajan, Carbohydr. Res., 2006, 341, 2012–2018 CrossRef CAS.
- B. Ankamwar, C. Damle, A. Ahmad and M. Sastry, J. Nanosci. Nanotechnol., 2005, 5, 1665–1671 CrossRef CAS.
- A. Bharde, A. Kulkarni, M. Rao, A. Prabhune and M. Sastry, J. Nanosci. Nanotechnol., 2007, 7, 4369–4377 CrossRef CAS.
- D. R. Bhumkar, H. M. Joshi, M. Sastry and V. B. Pokharkar, Pharm. Res., 2007, 24, 1415–1426 CrossRef CAS.
- S. P. Chandran, M. Chaudhary, R. Pasricha, A. Ahmad and M. Sastry, Biotechnol. Prog., 2006, 22, 577–583 CrossRef CAS.
- A. Gole, C. Dash, C. Soman, S. R. Sainkar, M. Rao and M. Sastry, Bioconjugate Chem., 2001, 12, 684–690 CrossRef CAS.
- P. Mukherjee, A. Ahmad, D. Mandal, S. Senapati, S. R. Sainkar, M. I. Khan, R. Ramani, R. Parischa, P. V. Ajayakumar, M. Alam, M. Sastry and R. Kumar, Angew. Chem., Int. Ed., 2001, 40, 3585–3588 CrossRef CAS.
- P. Mukherjee, S. Senapati, D. Mandal, A. Ahmad, M. I. Khan, R. Kumar and M. Sastry, ChemBioChem, 2002, 3, 461–463 CrossRef CAS.
- P. R. Selvakannan, S. Mandal, R. Pasricha and M. Sastry, J. Nanosci. Nanotechnol., 2003, 3, 372–374 CrossRef CAS.
- Q. Zhang, L. Zhang and J. H. Li, J. Nanosci. Nanotechnol., 2007, 7, 4557–4561 CrossRef CAS.
- Y. Yang and T. Coradin, Green Chem., 2008, 10, 183–190 RSC.
- W. Yang, Y. Ma, J. Tang and X. R. Yang, Colloids Surf., A, 2007, 302, 628–633 CrossRef CAS.
- W. J. Yan, R. Wang, Z. Q. Xu, J. K. Xu, L. Lin, Z. Q. Shen and Y. F. Zhou, J. Mol. Catal., A: Chem., 2006, 255, 81–85 CrossRef CAS.
- C. C. Wu and D. H. Chen, Gold Bull., 2007, 40, 206–212 CAS.
- Q. L. Wei, S. Z. Kang and J. Mu, Colloids Surf., A, 2004, 247, 125–127 CrossRef CAS.
- C. Y. Tai, Y. H. Wang and H. S. Liu, AIChE J., 2008, 54, 445–452 CrossRef CAS.
- T. Premkumar and K. E. Geckeler, J. Phys. Chem. Solids, 2006, 67, 1451–1456 CrossRef CAS.
- N. N. Mallikarjuna, X. Y. Zhang, A. M. Wu, H. Kolla and S. Manohar, Abstr. Pap. Am. Chem. Soc., 2005, 229, U51–U51.
- Z. H. Lin, Y. W. Lin, K. H. Lee and H. T. Chang, J. Mater. Chem., 2008, 18, 2569–2572 RSC.
- S. K. Li, Y. H. Shen, A. J. Xie, X. R. Yu, L. G. Qiu, L. Zhang and Q. F. Zhang, Green Chem., 2007, 9, 852–858 RSC.
- J. H. Li, C. L. Ren, X. Liu, Z. De Hu and D. S. Xue, Mater. Sci. Eng., A, 2007, 458, 319–322 CrossRef.
- H. Z. Huang and X. R. Yang, Carbohydr. Res., 2004, 339, 2627–2631 CrossRef CAS.
- J. Fu and P. Raveendran, Abstr. Pap. Am. Chem. Soc., 2003, 226, U119–U119.
- J. Chen, J. Wang, X. Zhang and Y. L. Jin, Mater. Chem. Phys., 2008, 108, 421–424 CrossRef CAS.
- M. A. Albrecht, C. W. Evans and C. L. Raston, Green Chem., 2006, 8, 417–432 RSC.
- M. Poliakoff, J. M. Fitzpatrick, T. R. Farren and P. T. Anastas, Science, 2002, 297, 807–810 CrossRef CAS.
- M. Poliakoff and P. Licence, Nature, 2007, 450, 810–812 CrossRef CAS.
- S. L. Y. Tang, R. L. Smith and M. Poliakoff, Green Chem., 2005, 7, 761–762 RSC.
- D. Kim, S. Park, J. H. Lee, Y. Y. Jeong and S. Jon, J. Am. Chem. Soc., 2007, 129, 7661–7665 CrossRef CAS.
- M. C. Daniel and D. Astruc, Chem. Rev., 2004, 104, 293–346 CrossRef CAS.
- M. Brust, M. Walker, D. Bethell, D. J. Schiffrin and R. Whyman, J. Chem. Soc., Chem. Commun., 1994, 801–802 RSC.
- B. D. Chithrani and W. C. W. Chan, Nano Lett., 2007, 7, 1542–1550 CrossRef CAS.
- R. Hong, G. Han, J. M. Fernandez, B. J. Kim, N. S. Forbes and V. M. Rotello, J. Am. Chem. Soc., 2006, 128, 1078–1079 CrossRef CAS.
- T. Nakai, T. Kanamori, S. Sando and Y. Aoyama, J. Am. Chem. Soc., 2003, 125, 8465–8475 CrossRef CAS.
- F. Osaki, T. Kanamori, S. Sando, T. Sera and Y. Aoyama, J. Am. Chem. Soc., 2004, 126, 6520–6521 CrossRef CAS.
- P. C. N. Rensen, L. A. J. M. Sliedregt, A. Ferns, E. Kieviet, S. M. W. van Rossenberg, S. H. van Leeuwen, T. J. C. van Berkel and E. A. L. Biessen, J. Biol. Chem., 2001, 276, 37577–37584 CrossRef CAS.
- P. H. Yang, X. S. Sun, J. F. Chiu, H. Z. Sun and Q. Y. He, Bioconjugate Chem., 2005, 16, 494–496 CrossRef CAS.
- H. Mizuno, Y. Y. Cho, F. Zhu, W. Y. Ma, A. M. Bode, C. S. Yang, C. T. Ho and Z. G. Dong, Mol. Carcinogen., 2006, 45, 204–212 Search PubMed.
- H. K. Na and Y. J. Surh, Mol. Nutr. Food Res., 2006, 50, 152–159 CrossRef CAS.
- H. Pelicano, D. Carney and P. Huang, Drug Resist. Update, 2004, 7, 97–110 Search PubMed.
- D. J. Sun, Y. Liu, D. C. Lu, W. Kim, J. H. Lee, J. Maynard and A. Deisseroth, Hum. Pathol., 2007, 38, 1047–1056 CrossRef CAS.
- M. Benhar, I. Dalyot, D. Engelberg and A. Levitzki, Mol. Cell. Biol., 2001, 21, 6913–6926 CrossRef CAS.
- M. A. Liebman, B. R. Williams, K. M. Daley and J. Sharon, Immunol. Lett., 2004, 91, 179–188 CrossRef CAS.
- C. Basset, J. Vadrot, J. Denis, J. Poupon and E. S. Zafrani, Liver Int., 2003, 23, 89–93 Search PubMed.
- C. F. Shaw, Chem. Rev., 1999, 99, 2589–2600 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Preparation of T-AuNP-5–11 and Fig. S1: Change in plasmon absorption maximum of T-AuNP-1 under various dilution conditions. See DOI: 10.1039/b822015h |
|
| This journal is © The Royal Society of Chemistry 2009 |
Click here to see how this site uses Cookies. View our privacy policy here.