Lithium intercalation and exfoliation of layered bismuth selenide and bismuth telluride†
Zhongfen
Ding
a,
Sabah K.
Bux
b,
Daniel J.
King
b,
Feng L.
Chang
c,
Tai-Hao
Chen
c,
Shu-Chuan
Huang
*c and
Richard B.
Kaner
*b
aMST-7, Los Alamos National Laboratory, MS E544, Los Alamos, New Mexico 87545, USA. E-mail: zding@lanl.gov; Fax: +1-505-6672185; Tel: +1-505-6062078
bDepartment of Chemistry and Biochemistry and California NanoSystems Institute, University of California Los Angeles, 607 Charles E. Young Drive Box 951569, Los Angeles, California 90095, USA. E-mail: kaner@chem.ucla.edu; Fax: +1-310-206-4038; Tel: +1-310-825-5346
cDepartment of Chemistry, National Dong Hwan University, No.1, Sec.2, Da Hsueh Rd., Shoufeng, Hualien, 97401, Taiwan. E-mail: schuang@mail.ndhu.edu.tw
First published on 27th February 2009
Abstract
Alloys of bismuth telluride (Bi2Te3) are commonly used in thermoelectric devices. These materials possess a hexagonal layered structure comprised of five atom thick stacks of Te–Bi–Te–Bi–Te held together by weak van der Waals forces. Lithium cations can be intercalated between the layers using the reducing power of solvated electrons in liquid ammonia. After intercalation, lithium can be removed by exfoliation to create a stable colloidal suspension of thin sheets of Bi2Te3 or Bi2Se3 in water. Zeta potential measurements indicate that the colloids are charge stabilized. These colloidal suspensions can be deposited onto a variety of substrates to create two-dimensional thin films. Atomic force microscopy indicates that initially individual layers are deposited. The films are partially oriented as observed using X-ray powder diffraction. Annealing at temperatures as low as 85 °C can produce highly oriented films. Thus intercalation, exfoliation and deposition from a charge-stabilized colloid can provide a scalable process for synthesizing bulk quantities of nanostructured thermoelectric materials.
Introduction
Thermoelectric (TE) materials can be used in a variety of applications from waste heat recovery via the Seebeck effect to electronic refrigeration via the Peltier effect. These solid-state devices have no moving parts and are therefore quiet, durable and reliable. They can be considered environmentally friendly since no ozone depleting chlorofluorocarbons (CFCs) are produced. For the past fifty years, the major obstacle has been the relatively low conversion efficiency of thermoelectric based devices.1 The thermoelectric conversion efficiency of a material is directly related to the Carnot efficiency and the dimensionless figure of merit, ZT, as given in Equation 1:| ZT = S2σT/κ | (1) |
Low dimensional materials have been suggested as a means to significantly increase the ZT via quantum confinement of the charge carriers.3–6 Recent work on Si nanowires has demonstrated that a doubling of ZT is possible.7,8 However, experiments indicate that the ZT enhancement was not the result of quantum confinement effects. Instead, a significant decrease in the thermal conductivity of the nanostructured materials via phonon scattering was observed along with preservation of the relatively high carrier mobilities.7–10 Here we focus on developing routes to create low dimensional forms of the materials with the highest known room temperature ZT values—bismuth telluride and its alloys.11 Bismuth telluride can be made either n-type by substitutional doping using Se to form Bi2(Se,Te)3 or p-type by substitutional doping of Sb to form (Sb,Bi)2Te3.2 The goal of this research is to develop scalable routes to nanostructured thermoelectric materials with the idea that phonon scattering at grain boundaries should ultimately reduce thermal conductivity and enhance the thermoelectric figure of merit. Bismuth telluride and its alloys are hexagonal layered materials comprised of five atom thick covalently bonded stacks of Te–Bi–Te–Bi–Te within each layer with the sheets held together by van der Waals forces. The layered structure and weak van der Waals forces can be exploited using intercalation chemistry followed by exfoliation to form nanostructured materials.12 This study describes the intercalation of Bi2Te3 and Bi2Se3 with lithium and exfoliation with water to yield stable colloids. The colloidal suspensions can in turn be deposited onto a variety of substrates to create highly oriented thin films as demonstrated using atomic force microscopy (AFM) and powder X-ray diffraction (XRD). An outline of the entire process is presented in Fig. 1.
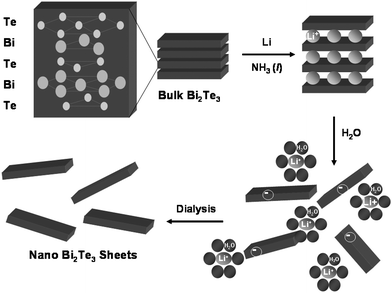 | ||
| Fig. 1 Schematic illustration of how layered Bi2Te3 can be intercalated with lithium and exfoliated with water to form a colloidal suspension of Bi2Te3 nano-sheets. Each layer of bulk Bi2Te3 is five atoms thick alternating between Te and Bi. In the presence of liquid ammonia (NH3), lithium dissolves to form Li cations and solvated electrons that in turn reduce Bi2Te3 to LiyBi2Te3. On exposure to water the reduced LiyBi2Te3 layers exfoliate forming a charge stabilized colloid. Dialysis of the suspension results in nano-sheets of Bi2Te3 in water that can be subsequently deposited and dried to form two-dimensional films. | ||
Results and discussion
Intercalation of layered bismuth telluride was originally attempted by electrochemical methods using a lithium anode.13,14 Unfortunately, the amount of lithium that could be intercalated was very limited since as the bismuth telluride lattice expanded pieces spalled off the cathode. Here we avoid this problem by using a chemical intercalation method. The strongest chemically reducing solution known is comprised of solvated electrons. By dissolving alkali metals in liquid ammonia, solvated electrons can be readily made. These solutions have been used successfully to intercalate many layered compounds from single layer graphite15 to the three atom thick layers found in transition metal dichalcogenides such as TiS216 and MoS2.17 Here we use lithium dissolved in liquid ammonia to intercalate Bi2Te3 and Bi2Se3. In a helium filled drybox, a stoichiometric amount of lithium is cut and put in a Pyrex vessel along with the Bi2Te3 or Bi2Se3. On a vacuum line, high purity ammonia is then condensed on top of the starting reagents using liquid nitrogen (at −196 °C). The ammonia is allowed to thaw slowly in an isopropanol bath cooled with an electronic cold finger that maintains the temperature at −45 °C. As the ammonia warms up and liquefies, the lithium rapidly dissolves forming Li+ ions and solvated electrons as given in Equation 2:| Li + NH3(l) → Li+ + e−(NH3) | (2) |
The solution containing solvated electrons is readily apparent from its characteristic blue color. The solvated electrons immediate begin to reduce the bismuth telluride or bismuth selenide with the concomitant intercalation of lithium cations to maintain charge balance as given in Equation 3:
| Bi2Te3 + yLi+ + ye− → Li+y[Bi2Te3]y− | (3) |
The resulting lithiated materials, i.e. LiyBi2Te3, appear to be stable as long as they are kept dry. Inductively coupled plasma mass spectrometry (ICP-MS) analysis shows that a lithium content greater than one can readily be achieved. This indicates that fully intercalated first stage compounds have been produced. Note that y values greater than one are known for other intercalation compounds.18
On exposure to water, the lithium ions in LiyBi2Te3 or LiyBi2Se3 rapidly become solvated producing lithium hydroxide (LiOH) and hydrogen gas. The rapid expansion leads to exfoliation and the formation of colloidal suspensions. The Li+ ions can then be removed via diffusion through a dialysis membrane leaving the counter ion, H+ from the dissociation of H2O to balance the charge. ICP-MS shows that only trace amounts of lithium are present in both bulk Bi2Te3 or Bi2Se3 before intercalation and in the nanostructured Bi2Te3 or Bi2Se3 produced via intercalation, exfoliation and dialysis.19
Here we will first look at the Bi2Se3 colloids and films made from them and then turn to Bi2Te3 colloids and films. Dynamic light scattering indicates that colloidal suspensions of Bi2Se3 have particle sizes centered around 10 nm. With time the particles begin to grow reaching an average size of 20 nm after approximately one day. After one week most of the particles have flocculated out of the colloidal suspension, while those remaining exhibit an average particle size of 450 nm. From this study it is clear that Bi2Se3 colloidal suspensions are stable for at least a day which allows sufficient time for their use in film formation.
Zeta potential analysis was carried out to determine what gives the colloidal suspensions their stability. Zeta potentials of three different concentrations of LiyBi2Se3 suspensions are presented in Fig. 2 as a function of time. The relatively high zeta potential values measured (−48.6 to −56.3 mV) indicate that charge stabilized colloidal suspensions have been created. The colloidal suspensions are stable for hours, but slowly lose their high surface charge over the course of several days leading to flocculation which is consistent with the dynamic light scattering experiments. Note that when a salt such as AlCl3 is added to the colloid, precipitation begins immediately. This experimental result provides further evidence for a charge stabilized colloid.20
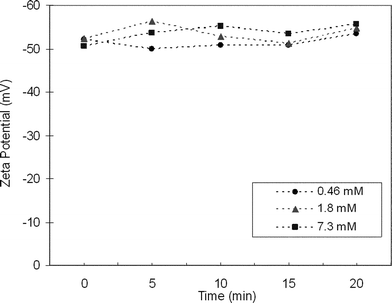 | ||
| Fig. 2 Zeta potential measurements for colloidal suspensions of 0.46 mM, 1.8 mM and 7.3 mM concentrations of LiBi2Se3 in water as a function of time. The high negative zeta potential values suggest that the colloids are charge stabilized. | ||
Highly ordered thin films of bismuth selenide can be made using the colloidal suspensions. First, dialysis is used to remove the lithium ions. After dialysis, the essentially lithium-free colloids can be deposited on almost any substrate simply by placing a clean substrate in a Petri dish and then pouring the colloidal suspension on top of the substrate or by drop-casting using a pipette and either waiting for the liquid to evaporate or providing heat to speed the process.
When Bi2Se3 is deposited on Pyrex glass slides it forms a film that is highly oriented along the c-axis as can be seen in the powder X-ray diffraction patterns presented in Fig. 3. The as-deposited film shows somewhat broadened yet intense (0 0 6) and (0 0 15) peaks which differ significantly from those obtained from bulk material. The observed peak broadening is indicative of small crystallites. This is confirmed using a line broadening calculation based on the Scherrer equation that indicates an average crystallite size of 20 nm. The 20 nm size is also consistent with the dynamic light scattering experiments carried out on the colloidal suspensions.
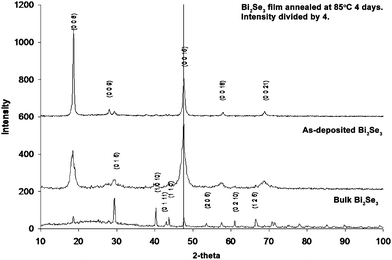 | ||
| Fig. 3 Powder X-ray diffraction patterns of bismuth selenide (Bi2Se3) as synthesized from the pure elements at 700 °C (bottom), as deposited on Pyrex from a colloidal suspension at room temperature (middle) and after annealing the deposited material at 85 °C for one week (top). Bulk Bi2Se3 possesses strong (0 1 5), (1 0 10) and (1 1 0) peaks, while the as-deposited film shows only strong (0 0 l) reflections indicating that it is oriented along the c-axis. Annealing the film at 85 °C results in a highly c-axis oriented film. | ||
Bismuth selenide has a hexagonal crystal structure that produces strong (0 1 5), (1 0 10) and (1 1 0) X-ray diffraction peaks in the bulk material. However, the XRD pattern of the as-deposited Bi2Se3 thin film mainly exhibits intense (0 0 l) peaks and is therefore clearly oriented in the c-axis direction. This indicates that the Bi2Se3 thin sheets lie parallel to the substrate surface. Annealing can be used to increase the crystallinity of the Bi2Se3 thin films. The top X-ray pattern in Fig. 3 shows that a deposited film becomes highly crystalline when annealed, even at temperatures as low as 85 °C, as sharp (0 0 l) peaks dominate the diffraction pattern.
To investigate what happens in the initial stages of deposition, atomic force microscopy (AFM) was used to study Bi2Se3 thin films deposited on mica (Fig. 4). Freshly cleaved mica provides a nice flat surface that is useful in AFM experiments. The AFM image shows that a very thin film of Bi2Se3, approximately 1 µm in width and 0.7 μm2 in area, has been deposited on the mica substrate. The height profiles A, B, C and D were measured at different locations on the film and indicate that the film has a consistent thickness of ∼20 Å, which corresponds to 2 unit layers since each layer is 10.16 Å thick.21 Occasionally, single layer, i.e. 10 Å thick, films have been observed using AFM, but these films usually occupy smaller areas and often exhibit greater surface roughness. Three and four layer thick films have also been observed suggesting that layers are deposited sequentially as the colloid dries.
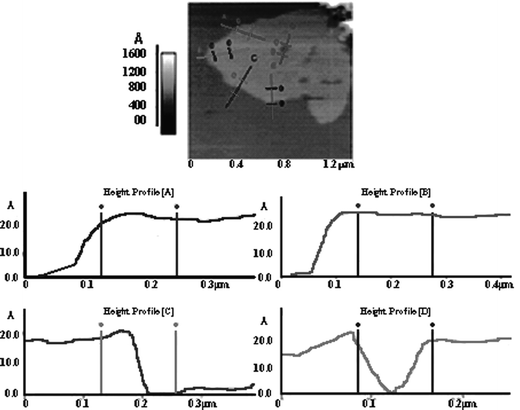 | ||
| Fig. 4 Atomic force microscopy height profile measurements of a bismuth selenide (Bi2Se3) thin film deposited on a freshly cleaved piece of mica. Four scanning paths are shown with the resulting height profiles labeled A through D. Each of the profiles indicates that the height of the film is around 20 Å, which is equivalent to two unit layers of Bi2Se3. Profile D shows a spot where there is a discontinuity in the film coverage. | ||
Colloidal suspensions of Bi2Te3 can be produced by exfoliating LiyBi2Te3 in water. Dynamic light scattering indicates that the as-formed colloids have an average particle size of 20 nm. Zeta potential analyses of three different concentrations of LiyBi2Te3 are presented in Fig. 5 as a function of time. The relatively high zeta potential values (−42.6 to −59.7 mV) indicate that the colloidal suspensions are charge stabilized. Like the Bi2Se3 colloids, the particle size increases slowly over several hours and will flocculate out of solution within days. Although the Bi2Te3 colloids are not quite as stable as the Bi2Se3 colloids, the Bi2Te3 colloids are certainly sufficiently stable for casting films.
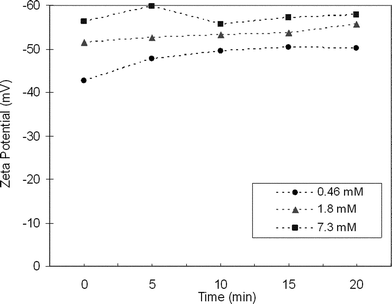 | ||
| Fig. 5 Zeta potential measurements for colloidal suspensions of 0.46 mM, 1.8 mM and 7.3 mM concentrations of LiBi2Te3 in water as a function of time. The high negative zeta potential values suggest that the colloids are charge stabilized. | ||
Thin films of Bi2Te3 deposited on Pyrex glass at room temperature show little preferred orientation as can be seen from the X-ray diffraction patterns presented in Fig. 6. The as-deposited film has a broad (0 1 5) peak which is the strongest peak observed for bulk Bi2Te3.22 The crystallinity of the film can be increased by annealing for an hour as can be seen in Fig. 6. Heating the film to 350 °C significantly increases the crystallinity and brings out the (1 0 10) and (0 0 15) reflections as the next most prominent peaks in the X-ray powder pattern. Although these peaks with large l components suggest some c-axis orientation, the (1 0 10) and (0 0 15) reflections are also the next strongest peaks observed in the X-ray diffraction pattern of bulk Bi2Te3.
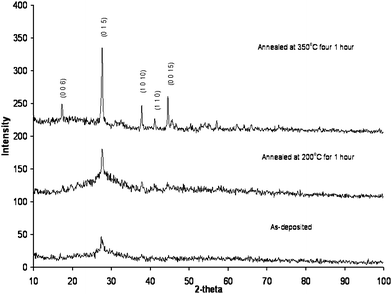 | ||
| Fig. 6 Powder X-ray diffraction patterns of bismuth telluride (Bi2Te3) as-deposited on Pyrex from a colloidal suspension at room temperature (bottom), after annealing at 200 °C for one hour (middle) and after annealing at 350 °C for 1 hour (top). The most intense peak from each of the patterns is the (0 1 5) reflection which is also the most intense peak found in bulk Bi2Te3. Annealing increases the crystallinity of Bi2Te3, but the films are only partially oriented along the c-axis. | ||
Although Bi2Te3 and Bi2Se3 have similar structures and form comparable colloids, they have very different film forming properties. One might expect comparable films since in each case the deposition process occurs as water evaporates leaving thin sheets from the colloid deposited on the substrate. The expectation is that the sheets will lie flat on the surface, producing films that are highly c-axis oriented. Indeed this is exactly what occurs with Bi2Se3 films, but the deposition of Bi2Te3 films produces much less orientation. This can be thought of in terms of competing forces that determine the quality and orientation of the deposited colloids. These forces include those that favor oriented sheets: gravity, the interaction between the substrate and the colloidal particles and electrostatic repulsion between the negatively charged thin sheets themselves. The force that works against orientation is the minimization of surface energy which favors aggregation. The bismuth selenide sheets with high aspect ratio as observed by AFM thus produce more highly oriented thin films than the lower aspect ratio bismuth telluride particles. Future experiments will explore making larger bismuth telluride sheets along with controlling the pH of the colloids to see if more highly oriented films can be made.
Materials preparation and characterization
Bulk materials synthesis and lithium intercalation
Bulk crystals of Bi2Te3 and Bi2Se3 were synthesized using classical solid state chemistry in which freshly ground elements are combined in stoichiometric amounts and heated to 700 °C under vacuum as described previously.23 A schematic diagram for the intercalation and exfoliation of Bi2Se3 and Bi2Te3 is illustrated in Fig. 1. The intercalation of lithium using liquid ammonia was carried out as previously described in more detail in reference 12. A molar ratio of 1.5 equivalents of Li to Bi2Te3 or Bi2Se3 was used to assure complete intercalation. The exfoliation was carried out by adding ultra-pure water to the lithium intercalated compound. The lithium ions were subsequently removed by dialysis.Substrate preparation
Pyrex substrates of 0.6 cm × 1.0 cm pieces were boiled in piranha solution (H2O2:H2SO4, 1:1) for 30 minutes to remove any surface organic contaminants, then rinsed with water and dried in an oven. A cleaned Pyrex® substrate was placed in a Petri dish and colloidal suspensions were decanted into the dish. The water was left to evaporate and the resulting deposited films were subsequently annealed to increase crystallinity.Mica substrates were first cut into 0.6 cm × 1.0 cm pieces. Then a piece of Scotch™ tape was used to peel off some surface layers, exposing clean, smooth surfaces. The films were drop-cast from the colloidal suspension and dried. The resulting thin films were used for AFM analysis.
Characterization
Dynamic light scattering of colloidal suspensions was measured using a MICROTRAC ultrafine particle analyzer with water (refractive index = 1.33, viscosity = 0.924 at 23 °C) as the solvent. The particle density is 7.74 g/cm3 for Bi2Te3 and 7.50 g/cm3 for Bi2Se3. Inductively coupled plasma-mass spectrometry (ICP-MS) was used to establish the lithium content. A zeta potential analyzer (Malvern, Nano Zen3600 Zetasizer) was used to determine the surface charge on the particles in the colloidal suspensions.X-Ray powder diffraction patterns were taken in 0.1° 2θ steps at 3 sec/step using a Crystal Logic diffractometer with Ni-filtered Cu Kα radiation for the Bi2Te3 and Bi2Se3 thin films. Atomic force microscopy scans were performed in contact mode on an Autoprobe (Park Scientific Instruments) using v-shaped silicon nitride cantilevers (Veeco, USA).
Conclusions
Using the reducing power of solvated electrons, lithium can be intercalated into the layered thermoelectric materials bismuth telluride and bismuth selenide. The intercalated materials can then be exfoliated in water to create colloidal suspensions. Zeta potential analysis indicates that the colloids are charge stabilized. Dialysis can then be used to remove the lithium ions. The colloidal suspensions of Bi2Te3 and Bi2Se3 can be deposited as thin films on various substrates. Bi2Se3 films show a preferred c-axis orientation. An AFM height profile of a typical Bi2Se3 film at the beginning stage of growth shows a 20 Å thick slab corresponding to 2 unit layers of material. This suggests that the material deposits layer by layer. Powder X-ray diffraction indicates that c-axis oriented bismuth selenide is deposited at room temperature. Annealing at temperatures as low as 85 °C creates highly oriented films. Bismuth telluride can also be readily intercalated with lithium and exfoliated in water to produce a colloid. Zeta potential experiments again indicate that a charge stabilized colloid is formed. Although bismuth telluride films deposited on glass show much less preferred orientation when compared to the bismuth selenide films, annealing at 300 °C produces crystalline films. The intercalation, exfoliation and deposition method for layered compounds such as Bi2Te3 and Bi2Se3 represents a scalable route to nanostructured thermoelectric materials.Acknowledgements
The authors are grateful for support from the National Science Foundation: DMR-0453121 (RBK) and IGERT Fellowships DGE-0114443 and DGE-0654431 (SKB and DJK); and a thermoelectrics subcontract from the Jet Propulsion Laboratory/Caltech-1308818.References
- G. J. Snyder and E. S. Toberer, Nature Materials, 2008, 7, 105–114 CrossRef CAS.
- E. J. Winder, A. B. Ellis and G. C. Lisensky, Journal of Chemical Education, 1996, 73, 940–946 CrossRef CAS.
- L. D. Hicks and M. S. Dresselhaus, Physical Review B, 1993, 47, 16631–16634 CAS.
- L. D. Hicks and M. S. Dresselhaus, Physical Review B, 1993, 47, 12727–12731 CAS.
- L. D. Hicks, T. C. Harman and M. S. Dresselhaus, Applied Physics Letters, 1993, 63, 3230–3232 CrossRef CAS.
- S. Cronin, T. Koga, X. Sun, Z. Ding, S.-C. Huang, R. Kaner, M. S. Dresselhaus, in Materials Research Society Symposium Proceedings, 1998, p. p. 397 Search PubMed.
- A. I. Boukai, Y. Bunimovich, J. Tahir-Kheli, J.-K. Yu, W. A. Goddard Iii and J. R. Heath, Nature, 2008, 451, 168–171 CrossRef CAS.
- A. I. Hochbaum, R. Chen, R. D. Delgado, W. Liang, E. C. Garnett, M. Najarian, A. Majumdar and P. Yang, Nature, 2008, 451, 163–167 CrossRef CAS.
- M. S. Dresselhaus, G. Chen, M. Y. Tang, R. G. Yang, H. Lee, D. Z. Wang, Z. F. Ren, J. P. Fleurial and P. Gogna, Advanced Materials, 2007, 19, 1043–1053 CrossRef CAS.
- M. S. Dresselhaus, G. Chen, Z. Ren, J.-P. Fleurial, P. Gogna, M. Y. Tang, D. Vashaee, H. Lee, X. Wang, G. Joshi, G. Zhu, D. Wang, R. Blair, S. Bux, R. Kaner, in Materials Research Society Conference Proceedings, 2007, pp. U02–04 Search PubMed.
- B.C.M., D. M. Rowe, Modern Thermoelectrics, Holt, Rinehart and Winston, London. 1983 Search PubMed.
- Z. Ding, L. Viculis, J. Nakawatase and R. B. Kaner, Advanced Materials, 2001, 13, 797–800 CrossRef CAS.
- I. D. Kozmik, I. I. Grigorchak, Z. D. Kovalyuk, B. P. Bakhmatyuk, S. V. Gavrilyuk and M. V. Tovarnitskii, Zhurnal Fizicheskoi Khimii, 1990, 64, 840–843 CAS.
- S. N. Chizhevskaya, T. E. Svechnikova, S. Y. Skipidarov, N. A. Tsvetkova and N. N. Nemtsov, Inorganic Materials, 1992, 28, 231–234.
- W. Rudorff, Advances in Inorganic Chemistry, 1959, 1, 223–266 CAS.
- M. S. Whittingham, Science, 1976, 192, 1126–1127 CAS.
- W. Rudorff, Angewandte Chemie-International Edition, 1959, 71, 487–491 Search PubMed.
- D. Yang and R. F. Frindt, Journal of Physics and Chemistry of Solids, 1996, 57, 1113–1116 CrossRef CAS.
- Z. Ding, in Chemistry and Biochemistry, University of California, Los Angeles, Los Angeles, 2001 Search PubMed.
- A. Sheludko, Colloid Chemistry, Elsevier, Amsterdam, 1966 Search PubMed.
- R. W. G. Wyckoff, Landolt-Börnstein, New Series III/17f, Group III: Crystal and Solid State Physics, Springer, Berlin, 1983 Search PubMed.
- I. C. f. D. Data, 2003, PDF-2, #00-015-0863.
- Z. Ding, S.-C. Huang, D. Marcus, R. B. Kaner, ICT'99. Proc. 18th International Conference on Thermoelectrics, IEEEBaltimore, MD. 1999721–724 Search PubMed.
Footnote |
| † This paper is part of a Journal of Materials Chemistry theme issue on Layered Materials. Guest editors: Leonardo Marchese and Heloise O. Pastore. |
| This journal is © The Royal Society of Chemistry 2009 |
