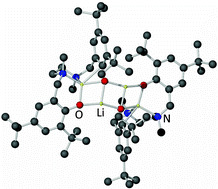
Two lithium and one sodium diamine bis(phenolate) complexes have been prepared and characterised by X-ray crystallography and NMR spectroscopy. Two parent diamine bis(phenol) ligands were utilised in the study (1-H2 and 2-H2). Dimeric (1-Li2)2 was prepared by treating 1-H2 with two molar equivalents of n-butyllithium in hydrocarbon solvent. It adopts a ladder-like structure in the solid state, which appears to deaggregate in C6D6 solution. The monomeric (hence, dinuclear) TMEDA-solvated species [2-Li2·(TMEDA)] has two chemically unique Li atoms in the solid state and is prepared by reacting 2-H2 with two molar equivalents of n-butyllithium in hydrocarbon solvent, in the presence of N,N,N′,N′-tetramethylethylenediamine (TMEDA). Finally, the dimeric sodium-based [2-Na2·(OEt2)]2 was prepared by reacting 2-H2 with two molar equivalents of freshly prepared n-butylsodium in a hydrocarbon–diethyl ether medium. The complex adopts a Na4O4 cuboidal structure in the solid state, which appears to remain intact in C6D6 solution.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...