Electrochromics for smart windows: thin films of tungsten oxide and nickel oxide, and devices based on these
Abstract
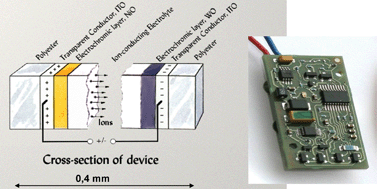
Electrochromic (EC) materials are able to change their optical properties, reversibly and persistently, by the application of an electrical voltage. These materials can be integrated in multilayer devices capable of modulating the optical transmittance between widely separated extrema. We first review the recent literature on inorganic EC materials and point out that today's research is focused on tungsten oxide (colouring under charge insertion) and nickel oxide (colouring under charge extraction). The properties of thin films of these materials are then discussed in detail with foci on recent results from two comprehensive investigations in the authors' laboratory. A logical exposition is obtained by covering, in sequence, structural features, thin film deposition (by sputtering), electronic band structure, and ion diffusion. A novel conceptual model is given for structural characteristics of amorphous W oxide films, based on notions of defects in the ideal amorphous state. It is also shown that the conduction band density of states is obtainable from simple electrochemical chronopotentiometry. Ion intercalation causes the charge-compensating electrons to enter localized states, implying that the optical absorption underlying the electrochromism can be described as ensuing from transitions between occupied and empty localized conduction band states. A fully quantitative theory of such transitions is not available, but the optical absorption can be modeled more phenomenologically as due to a superposition of transitions between different charge states of the W ions (6+, 5+, and 4+). The Ni oxide films were found to have a porous structure comprised of small grains. The data are consistent with EC coloration being a surface phenomenon, most likely confined to the outer parts of the grains. Initial electrochemical cycling was found to transform hydrated Ni oxide into hydroxide and oxy-hydroxide phases on the grain surfaces. Electrochromism in thus stabilized films is consistent with reversible changes between Ni hydroxide and oxy-hydroxide, in accordance with the Bode reaction scheme. An extension of this model is put forward to account for changes of NiO to Ni2O3. It was demonstrated that electrochromism is associated solely with proton transfer. Data on chemical diffusion coefficients are interpreted for polycrystalline W oxide and Ni oxide in terms of the lattice gas model with interaction. The later part of this review is of a more technological and applications oriented character and is based on the fact that EC devices with large optical modulation can be accomplished essentially by connecting W-oxide-based and Ni-oxide-based films through a layer serving as a pure ion conductor. Specifically, we treat methods to enhance the bleached-state transmittance by mixing the Ni oxide with other oxides characterized by wide band gaps, and we also discuss pre-assembly charge insertion and extraction by facile gas treatments of the films, as well as practical device manufacturing and device testing. Here the emphasis is on novel flexible polyester-foil-based devices. The final part deals with applications with emphasis on architectural “smart” windows capable of achieving improved indoor comfort jointly with significant energy savings due to lowered demands for space cooling. Eyewear applications are touched upon as well.

 Please wait while we load your content...
Please wait while we load your content...