Inorganic intermolecular motifs, and their energies†
Abstract
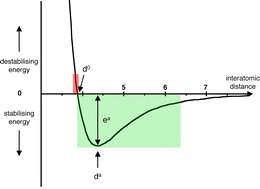
This article focuses on materials containing inorganic molecules and coordination complexes, assembled via intermolecular interactions. For inorganic systems that can contain the full range of elements and exhibit the full diversity of chemistry, some clarification is needed for fundamental concepts about molecular and non-molecular structure. An underlying theme is the need for knowledge of intermolecular energy potentials as a prerequisite for reliable design and preparation of molecular materials. Relationships between intermolecular potentials, histograms of intermolecular distances, and van der Waals surfaces, are discussed. A density functional method for efficient calculation of intermolecular potentials is evaluated, and some results presented, particularly for compounds with high-Z atoms where the integrity of molecular surfaces is diminished. In the context of the higher ordering of molecular assemblies, the main types of concerted intermolecular embraces between arylated molecules and representative coordination complexes are reviewed. When charged polyatomic molecules are assembled, and homo-charged molecules are segregated, there are basic questions about the relative magnitudes and influences of electrostatic energies: this issue is considered in the context of observed embraces between homo-charged polyatomic molecules and coordination complexes.
- This article is part of the themed collection: Molecular Crystal Engineering

 Please wait while we load your content...
Please wait while we load your content...