DOI:
10.1039/B100890K
(Paper)
CrystEngComm, 2001,
3, 57-59
Crystal structures of two polymorphs of thiodiglycolamide via the formation of dimeric and catemeric hydrogen bonds
Received
25th January 2001
, Accepted 9th February 2001
Abstract
Thiodiglycolamide crystallizes in two different crystalline forms, triclinic and tetragonal, from methanol and dimethyl sulfoxide solvents respectively. While the triclinic form possesses dimeric hydrogen bond coupling between the adjacent amide molecules, the tetragonal form has a catemeric hydrogen bond pattern.
Introduction
Polymorphism – crystallization of a compound into different crystalline forms from different solvents – in fact, is the phenomenon that laid the foundation for the development of solid-state organic chemistry with the identification of different photochemical reactivities in different polymorphs of trans-cinnamic acid crystals.1–3 The pharmaceutical industry is one of the few areas to recognize the importance of the phenomenon as several drug molecules often crystallize in different polymorphs, with varying properties towards drug activity.4–6 In this connection, the well-known and unprecedented Ranitidine patent case is a representative example.7 However, the phenomenon was often considered to be a nuisance in general chemistry as no definite means of either prediction or deliberate synthesis of the polymorhic forms were established, as exemplified by Dunitz and Bernstein recently in their article ‘Disappearing polymorphs’.8 Also, it is believed that the success of obtaining a new polymorph of a specific compound is proportional to the number of man-hours spent crystallizing the compounds (a test of patience!).9 As a result, synthesis of new polymorphic forms becomes practically unattainable in a target synthesis both in terms of economics as well as time-scales involved.
However, in a recent statistical analysis10 on known polymorphic structures, polymorphs of some compounds with multifunctional groups capable of interacting with each other have been found to be easily distinguishable from each other by the nature of intermolecular bonds, such as hydrogen bonds, formed by the constituent functional groups. For instance, 2,6-dihydroxybenzoic acid forms two polymorphs with different types of hydrogen bonds formed by OH and COOH groups as shown in Scheme 1. This suggests that deliberate synthesis of polymorphic structures is possible if the molecules possess a functional group capable of forming different types of intermolecular bonds. Indeed, it appears to be feasible, as noted from the analysis of the two polymorphic forms – triclinic and tetragonal – of thiodiglycolamide, obtained serendipitously in the process of co-crystallisation of the amide with 4,4′-bipyridyl, and these results are discussed in this article.
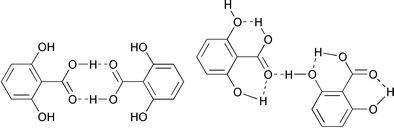 |
| | Scheme 1
Schematic representation of different kinds of hydrogen bond patterns in different polymorphs of a hydroxybenzoic acid.
| |
Results and discussion
While the triclinic11 form of 2 was obtained from a methanol solution, the tetragonal form was obtained from a dimethyl sulfoxide (DMSO) solution (for crystal data for 1 and 2 see Table 1). Packing analysis reveals the intriguing features for both of the structures with distinct differences in the packing arrangements. The two polymorphs form different kinds of N–H⋯O hydrogen bond couplings (see Scheme 2), leading to unique packing arrangements in each structure.
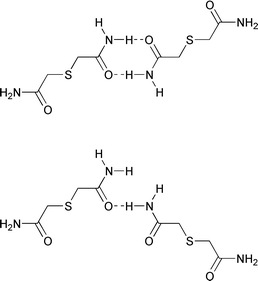 |
| | Scheme 2
Schematic representation of the difference in the hydrogen bonding network between the two polymorphs of thiodiglycolamide.
| |
| |
1
|
2
|
|
Full-matrix, least square refinement on F2. H atoms were obtained from Fourier difference maps.
Click b100890k.txt for full crystallographic data (CCDC 157832–157833).
|
| Formula |
C4H8N2O2S |
C4H8N2O2S |
| Crystal dimensions/mm |
0.30⊕×⊕0.20⊕×⊕0.25 |
0.25⊕×⊕0.20⊕×⊕0.25 |
| Crystal geometry |
Rod |
Diamond |
| Crystal system |
Triclinic |
Tetragonal |
| Space group |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P41212 |
|
a/Å |
5.142(1) |
5.172(1) |
|
b/Å |
8.125(1) |
5.172(1) |
|
c/Å |
8.221(1) |
25.085(1) |
|
α/° |
83.75(1) |
90 |
|
β/° |
117.2(1) |
90 |
|
γ/° |
85.47(1) |
90 |
|
U/Å3 |
325.2(1) |
670.9(2) |
|
Z
|
2 |
4 |
|
T/K |
293 |
293 |
|
F(000) |
156 |
312 |
|
μ(Mo-Kα)/mm−1 |
0.42 |
0.41 |
| Number of reflections (independent) |
1366 (736) |
2904 (1944) |
|
R
1
|
0.085 |
0.025 |
|
wR
2
|
0.170 |
0.070 |
In the triclinic form, the adjacent molecules are held together by cyclic N–H⋯O hydrogen bond couplings generally known for amide compounds [Scheme 3(a)].12 In addition, interestingly, the adjacent molecules form a cyclic dimer by connecting in a head-to-tail manner as shown in Fig. 1. The H⋯O distances observed are 2.05 and 2.17 Å. Other characteristics of these bonds are given in Table 2. These cyclic units, connected together by a centrosymmetric cyclic C–H⋯O hydrogen bond coupling, form a sheet structure in the two-dimensional arrangement. The corresponding H⋯O distance is 2.63 Å. In the three-dimensional arrangement, the sheets comprise cyclic networks stacked along [100] and connect together through typical N–H⋯O hydrogen bonds perpendicular to the cyclic hydrogen bond motif, involving the second hydrogen on the NH2 group of the amide group. It is worthy of mention that this kind of packing arrangement, as well as being present in the formation of cyclic supermolecules, was noted earlier in a 1∶1 complex of thiodiglycolic acid and 4,4′-bipyrdiyl.13
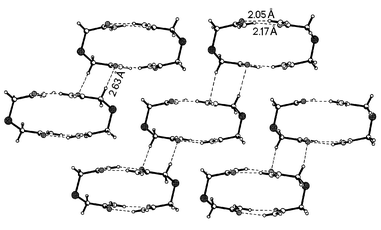 |
| | Fig. 1
Formation of ring dimers via cyclic N–H⋯O hydrogen bonds in the triclinic form of thiodiglycolamide. Notice the C–H⋯O hydrogen bonds connecting the rings. Click image or 1.htm to access a 3D representation.
| |
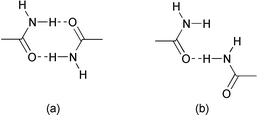 |
| | Scheme 3
Schematic representation of (a) dimeric hydrogen bond and (b) catemeric hydrogen bond patterns of CONH2.
| |
Table 2
Hydrogen bonding parameters for the triclinica and tetragonalb polymorphic modifications of thiodiglycolamide
| Hydrogen bond |
X–H⋯O/Å |
X⋯O/Å |
X–H⋯O/° |
|
Shown as normal font.
Shown as bold font; X⊕=⊕N or C.
|
| N–H⋯O |
2.05 |
2.88 |
164 |
| |
2.17 |
3.02 |
170 |
|
2.15
|
2.94
|
152
|
|
2.26
|
2.95
|
137
|
| C–H⋯O |
2.63 |
3.35 |
131 |
| |
2.05
|
2.94
|
174
|
|
2.26
|
2.95
|
130
|
However, in the crystal structure of the tetragonal form, the adjacent molecules are held together by a single N–H⋯O hydrogen bond in a catemeric fashion (as shown in Fig. 2) with a H⋯O distance of 2.05 Å
(Table 2), forming linear chains. These linear chains are further held together by a N–H⋯O hydrogen bond (H⋯O, 2.26 Å) formed by the second amide group. It is interesting to note that neither of the amide groups forms the generally known centrosymmetric coupling for the amide functional group.
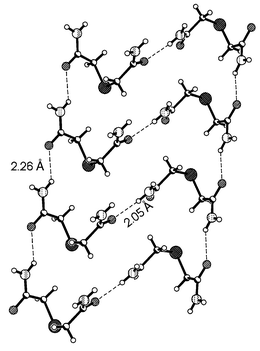 |
| | Fig. 2
Linear chain arrangement between the adjacent molecules of thiodiglycolamide in the tetragonal form. Notice the formation single N–H⋯O hydrogen bonds, unlike the cyclic rings formed in the triclinic form. Click image or 2.htm to access a 3D representation.
| |
The noted differences in the formation of different types of hydrogen bond couplings by the amide moiety has prompted a systematic study of these patterns. In this regard, a search performed on the Cambridge Structural Database (CSD),14 comprising of 207![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 507 crystal structures, (version 5.18) reveals quite interesting features. There is a total of 1070 entries containing the amide functional group (excluding organometallic compounds) for which three-dimensional coordinates are available. While 301 structures of these compounds possess a cyclic hydrogen bond pattern [Scheme 3(a)], the catemeric hydrogen bond pattern [Scheme 3(b)] is present in a nearly similar number of structures (245 structures), suggesting equal occurrence of both types of hydrogen bond pattern. A comparative study of the characteristics of both these types of hydrogen bond, shown in Fig. 3, further ascertains that these patterns are very similar with nearly equal mean H⋯O distances of 2.08 and 2.19 Å, respectively, for cyclic and catemeric hydrogen bonds. This intriguing hydrogen bond feature in the amide compounds suggests that amide functionality may be a better choice as steering group, if not the best, for the synthesis of polymorphic forms. In conclusion, it appears that the target synthesis of polymorphs could be carried out by employing the CONH2 moiety, and that successful results could be obtained with other functional groups as well, provided that more precise knowledge of the interplay of intermolecular interactions formed by such functional groups is well understood (as exemplified recently by Allen et al.15 for various types of hydrogen bond formed by COOH).
507 crystal structures, (version 5.18) reveals quite interesting features. There is a total of 1070 entries containing the amide functional group (excluding organometallic compounds) for which three-dimensional coordinates are available. While 301 structures of these compounds possess a cyclic hydrogen bond pattern [Scheme 3(a)], the catemeric hydrogen bond pattern [Scheme 3(b)] is present in a nearly similar number of structures (245 structures), suggesting equal occurrence of both types of hydrogen bond pattern. A comparative study of the characteristics of both these types of hydrogen bond, shown in Fig. 3, further ascertains that these patterns are very similar with nearly equal mean H⋯O distances of 2.08 and 2.19 Å, respectively, for cyclic and catemeric hydrogen bonds. This intriguing hydrogen bond feature in the amide compounds suggests that amide functionality may be a better choice as steering group, if not the best, for the synthesis of polymorphic forms. In conclusion, it appears that the target synthesis of polymorphs could be carried out by employing the CONH2 moiety, and that successful results could be obtained with other functional groups as well, provided that more precise knowledge of the interplay of intermolecular interactions formed by such functional groups is well understood (as exemplified recently by Allen et al.15 for various types of hydrogen bond formed by COOH).
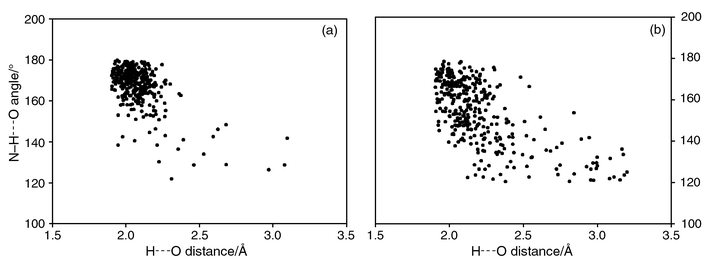 |
| | Fig. 3
Scatter plots showing similarity between (a) cyclic hydrogen bond and (b) catemeric hydrogen bond patterns present in amide compounds.
| |
Acknowledgements
I thank Dr Paul Ratnasamy and Dr K. N. Ganesh, respective Director and Head of the Division of Organic Chemistry, National Chemical Laboratory, Pune, India, for providing grant assistance for computational equipment used to carry out structure determinations.
References
-
(a) G. M. J. Schmidt, Pure Appl. Chem., 1971, 27, 647 CrossRef CAS;
(b) M. Lahav and G. M. J. Schmidt, J. Chem. Soc. B, 1967, 239 RSC.
-
G. R. Desiraju, Organic Solid State Chemistry, Elsevier, Amsterdam, 1987. Search PubMed.
-
G. R. Desiraju, Crystal Engineering: The Design of Organic Solids, Elsevier, Amsterdam, 1989. Search PubMed.
-
S. R. Byrn, Solid State Chemistry of Drugs, Academic Press, New York, 1982. Search PubMed.
-
W. C. McCrone, Polymorphism in Physics and Chemistry of the Organic Solid State, Vol. 2, ed. D. Fox and M. M. Labes, Weinberger, Interscience, New York, 1965, p. 725. Search PubMed.
-
(a) A. Nangia and G. R. Desiraju, Chem. Commun., 1999, 605 RSC;
(b) R. J. Davey, N. Blagden, G. D. Potts and R. Docherty, J. Am. Chem. Soc., 1997, 119, 1767 CrossRef CAS;
(c) V. R. Thalladi, M. Muthuraman, A. Nangia and G. R. Desiraju, Acta Crystallogr., Sect. C, 1999, 55, 698 CrossRef.
-
W. H. Decamp, in Proceedings of 3rd International Workshop on Crystal Growth of Organic Materials, ed. A. S. Myerson, D. A. Green and P. Meenan, ACS series, Washington DC, 1996. Search PubMed.
-
(a) J. D. Dunitz and J. Bernstein, Acc. Chem. Res., 1995, 28, 193 CrossRef CAS;
(b)
T. Liad, in Comprehensive Medicinal Chemistry, ed. P. D. Kennewell, Pergamon, Oxford, 1990, vol. 5, p. 545. Search PubMed.
-
M. R. Caira, in Design of Organic Solids, ed. E. Weber, Springer-Verlag, Berlin, 1998, p. 164. Search PubMed.
-
A. Nangia and G. R. Desiraju, in Design of Organic Solids, ed. E. Weber, Springer, Berlin, 1998, pp. 57–95. Search PubMed.
-
(a) Siemens, SMART System, Siemens Analytical X-Ray Instruments Inc., Madison, WI, 1995. Search PubMed;
(b)
G. M. Sheldrick, SHELXTL, Users Manual, Siemens Analytical X-Ray Instruments Inc., Madison WI, 1993. Search PubMed.
- Y. Weisinger-Lewin, F. Frolow, R. K. McMullan, T. F. Koetzle, M. Lahav and L. Leiserowitz, J. Am. Chem. Soc., 1989, 111, 1035 CrossRef CAS.
- V. R. Pedireddi, S. Chatterjee, A. Ranganathan and C. N. R. Rao, Tetrahedron, 1998, 54, 9457 CrossRef CAS.
- F. H. Allen and O. Kennard, Chem. Des. Autom. News, 1993, 8, 31 Search PubMed.
- F. H. Allen, W. D. S. Motherwell, P. R. Raithby, G. P. Shields and R. Taylor, New J. Chem., 1999, 23, 25 RSC.
|
| This journal is © The Royal Society of Chemistry 2001 |
Click here to see how this site uses Cookies. View our privacy policy here. 

![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif)



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 507 crystal structures, (version 5.18) reveals quite interesting features. There is a total of 1070 entries containing the amide functional group (excluding organometallic compounds) for which three-dimensional coordinates are available. While 301 structures of these compounds possess a cyclic hydrogen bond pattern [Scheme 3(a)], the catemeric hydrogen bond pattern [Scheme 3(b)] is present in a nearly similar number of structures (245 structures), suggesting equal occurrence of both types of hydrogen bond pattern. A comparative study of the characteristics of both these types of hydrogen bond, shown in Fig. 3, further ascertains that these patterns are very similar with nearly equal mean H⋯O distances of 2.08 and 2.19 Å, respectively, for cyclic and catemeric hydrogen bonds. This intriguing hydrogen bond feature in the amide compounds suggests that amide functionality may be a better choice as steering group, if not the best, for the synthesis of polymorphic forms. In conclusion, it appears that the target synthesis of polymorphs could be carried out by employing the CONH2 moiety, and that successful results could be obtained with other functional groups as well, provided that more precise knowledge of the interplay of intermolecular interactions formed by such functional groups is well understood (as exemplified recently by Allen et al.15 for various types of hydrogen bond formed by COOH).
507 crystal structures, (version 5.18) reveals quite interesting features. There is a total of 1070 entries containing the amide functional group (excluding organometallic compounds) for which three-dimensional coordinates are available. While 301 structures of these compounds possess a cyclic hydrogen bond pattern [Scheme 3(a)], the catemeric hydrogen bond pattern [Scheme 3(b)] is present in a nearly similar number of structures (245 structures), suggesting equal occurrence of both types of hydrogen bond pattern. A comparative study of the characteristics of both these types of hydrogen bond, shown in Fig. 3, further ascertains that these patterns are very similar with nearly equal mean H⋯O distances of 2.08 and 2.19 Å, respectively, for cyclic and catemeric hydrogen bonds. This intriguing hydrogen bond feature in the amide compounds suggests that amide functionality may be a better choice as steering group, if not the best, for the synthesis of polymorphic forms. In conclusion, it appears that the target synthesis of polymorphs could be carried out by employing the CONH2 moiety, and that successful results could be obtained with other functional groups as well, provided that more precise knowledge of the interplay of intermolecular interactions formed by such functional groups is well understood (as exemplified recently by Allen et al.15 for various types of hydrogen bond formed by COOH).
