Metal–organic supramolecular architecture containing cationic cavities: synthesis and single crystal investigation of {[Co(bpe)2(H2O)2](ClO4)2·(H2O)2}n
Eringathodi
Suresh
*a and
Mohan Madhav
Bhadbhade
*b
aSilicates and Catalysis Discipline, Central Salt and Marine Chemicals Research Institute, G.B.Marg, Bhavnagar-364 002, Gujarat, India. E-mail: salt@csir.res.in
bNational Chemical Laboratory, Physical Chemistry Division, Pune-411 008, India. E-mail: mohanb@sil.ncl.res.in
Abstract
The one-dimensional cavity-containing cationic complex [Co(bpe)2(H2O)2](ClO4)2·(H2O)2 is synthesized and characterized. Single crystal analysis reveals that the metal centres are doubly bridged via bpe ligands (bpe⊕=⊕1,2-bis(4-pyridyl)ethane) creating cationic channels with dimensions 9.29⊕×⊕9.94 Å across Co–Co(1) and C(6)–C(6A) atoms respectively, which are occupied by perchlorate anions and lattice water molecules along the c axis. The linear 1D polymeric chains run along the b axis and the versatile bpe ligand bridges the metal centres in a syn conformation.
Introduction
Molecular architecture constructed from metal-based coordination is an expanding field which leads to the development of new classes of functional materials. These complexes have potential application in the field of magnetic materials,1 molecular adsorption,2 host–guest chemistry3 and catalysis.4 However, coordination polymers with open framework structures are of interest from a structural point of view and their mimicry of microporous solids. The various factors which can influence the structural topologies in coordination polymers are the coordination geometry of the metals, structure of the spacer ligand, counter ions and reaction conditions. A building block approach has been utilized for the design of coordination polymers, which are assembled from suitable metal centres and organic ligands of different size and nature. A family of metal bridging building blocks containing 4-pyridyl donors having two coordination sites of the rigid ligands are often used to construct coordination polymers.5 Because of their ability to form H-bonded networks, the incorporation of the flexible 4-pyridyl ligands as linkers opens up more research avenues for coordination polymers to create high-dimensional networks. We chose 1,2-bis(4-pyridyl)ethane as it exhibits different conformations: anti (extended), syn (folded) (Fig. 1) and also because of its versatile nature as a spacer in any extended system. The dimensions of the polymer motif can be increased by cross linking the low-dimensional polymer by weaker intermolecular interactions such as hydrogen bonding and stacking in the solid state. In an endeavour to understand the network topology of the flexible bridging ligand 1,2-bis(4-pyridyl)ethane) (bpe) with the Co(II) metal centre by taking advantage of its conformational flexibility and the versatile binding mode, the {[Co(bpe)2(H2O)2](ClO4)2·2H2O}n complex was synthesized and structurally characterized.Experimental
All reagents were commercially available and used as received. Elemental analysis (C, H, N) was performed on a model 2400 Perkin-Elmer elemental analyzer. The FTIR spectrum was recorded from KBr pellets in the range 4000–400 cm−1 on a Perkin-Elmer Spectrum GX FTIR spectrophotometer.Synthesis of {[Co(bpe)2(H2O)2](ClO4)2·(H2O)2}n
To a solution of Co(NO3)2·6H2O (0.291 g, 1 mmol) in an EtOH–water mixture (15 ml, 2∶1 (v/v)), a solution of bpe (0.368 g, 2 mmol) in EtOH (10 ml) was added slowly with stirring over 15 min at 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C. To this a solution of sodium perchlorate (0.245 g, 2 mmol) in water (10 ml) was added and the resulting solution was refluxed on a water bath for 1 additional hour. The yellow solution was filtered, kept at 25
°C. To this a solution of sodium perchlorate (0.245 g, 2 mmol) in water (10 ml) was added and the resulting solution was refluxed on a water bath for 1 additional hour. The yellow solution was filtered, kept at 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C and stable crystals suitable for diffraction studies appeared within one week. Calc: C⊕=⊕41.26, H⊕=⊕4.29, N⊕=⊕8.02. Found: C⊕=⊕41.34, H⊕=⊕4.31, N⊕=⊕8.20%. υ/cm−1: 3224–2931 br, 1613 vs, 1561 m, 1503 m, 1423 vs, 1225 m, 1090 sh, 1061 m, 930 w, 830 sh, 811 m, 622 sh, 549 sh.
°C and stable crystals suitable for diffraction studies appeared within one week. Calc: C⊕=⊕41.26, H⊕=⊕4.29, N⊕=⊕8.02. Found: C⊕=⊕41.34, H⊕=⊕4.31, N⊕=⊕8.20%. υ/cm−1: 3224–2931 br, 1613 vs, 1561 m, 1503 m, 1423 vs, 1225 m, 1090 sh, 1061 m, 930 w, 830 sh, 811 m, 622 sh, 549 sh.
X-Ray crystallographic analysis
Data was collected on an Enraf-Nonius CAD-4 diffractometer with graphite monochromatised MoKα radiation (λ⊕=⊕0.7107 nm). Cell constants and the orientation matrix for data collection were obtained from least squares refinement using 25 high-angle reflections in the θ range 8–12°. The ω-2θ scan mode with a maximum θ value of 22.5° was used to collect the intensity data for the complex. Preliminary data, cell refinement and intensity data collection were carried out using the program CAD4-PC6 and the data reduction using NRCVAX.7 The data were corrected for Lorentz Polarization effects but not for absorption. Structure solution was carried out using the Patterson method with full-matrix least squares refinement on F2 using SHELX-97.8 Scattering factors and anomalous dispersions were taken from international tables for X-ray crystallography.9 Hydrogen atoms were located either from difference Fourier maps or kept fixed using the riding model, except for the lattice water hydrogens which were not available from the difference Fourier map. Crystallographic data and refinement for the compound are presented in Table 1.| a Click b009706n.txt for full crystallographic data (CCDC 154062) | |
|---|---|
| Formula | C12H16ClCo0.5N2O6 |
| M | 349.18 |
| Crystal system | Monoclinic |
| Space group | C2/c |
| Crystal dimensions/mm | 0.16⊕×⊕0.08⊕×⊕0.06 |
| a/Å | 18.522(7) |
| b/Å | 9.293(5) |
| c/Å | 18.782(5) |
| β/° | 104.20(3) |
| V/Å3 | 3143(2) |
| D c/g cm−1 | 1.480 |
| F(000) | 1444 |
| Total number of reflections | 2061 |
| Number of observed reflections [I⊕>⊕2σ(I)] | 1196 |
| Number of refined parameters | 223 |
| R1 | 0.0608 |
| wR2 | 0.1491 |
Results and discussion
An ORTEP10 view of the cationic linear coordination network is shown in Fig. 2. The structure of [Co(bpe)2(H2O)2] consists of linear cationic chains extending along the crystallographic b axis. The coordination sphere around Co(II) can be defined by four nitrogen atoms from the bpe ligands and two oxygen atoms from the water molecules forming slightly distorted octahedral geometry. The equatorial positions are occupied by four nitrogen atoms from doubly bridged bpe ligands, whereas the axial coordination is provided by the two oxygen atoms of the symmetry-related water molecules. The average Co–Nbpe (2.15 Å) and Co–OH2O (2.143 Å) distances are well within the range of reported values11 and all the angles around the metal centre are closer to the ideal value giving rise to a slightly distorted octahedral geometry around the Co2+. The molecule possesses a centre of symmetry passing through the metal centre which occupies the (0.5, 0.275, 0.25) postion. The Co(II) ion makes a good equatorial plane with the coordinated bpe nitrogens; both the trans nitrogens are slightly displaced in opposite directions by only ±0.026 Å. Each Co(II) centre is doubly bridged by the terminal nitrogen atoms of the bidentate bpe ligands to form a 22-membered cyclic ring extending the linear polymeric chain along the b axis. The adjacent metal centres bridged by the bpe ligands are separated by 9.293 Å.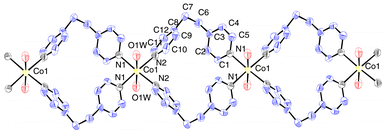 | ||
| Fig. 2 ORTEP view of the molecular structure of complex 1 at a 50% probability level. Perchlorate anions and lattice water molecules are omitted for clarity.Click image or 2.htm to access a 3D representation. | ||
A PLATON12 diagram showing the packing viewed down the c axis is shown in Fig. 3. The linear polymeric chain runs along the b axis with adjacent chains staggered by half the b axis with respect to each other in the ab plane to form effective close packing. The neighbouring chains which are almost aligned along the c axis are separated by 9.39 Å. The bpe units doubly bridged with the metal centre form through channels down the c axis with dimensions 9.293⊕×⊕9.413 Å,13 across the Co–Co(1) and C(6)–C(6A) atoms, respectively. These channels along the c axis are occupied by the perchlorate anions and the lattice water molecules which form alternate layers with the cationic framework. The CPK diagram is shown in Fig. 4.
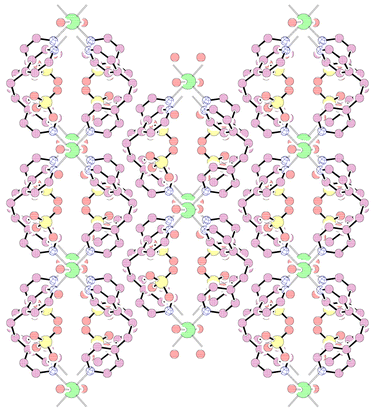 | ||
| Fig. 3 Packing diagram viewed down the c axis showing the staggering of adjacent polymeric chains along the ab plane and through the cavity along the c axis filled with perchlorate anions and lattice water molecules. | ||
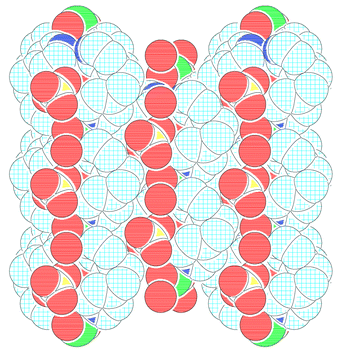 | ||
| Fig. 4 CPK diagram showing the cationic cavity filled with perchlorate anions and lattice water molecules. | ||
The intrachain metal–metal distance is shorter (9.293 Å) than the one found in the Co(II)·μ-4,4′-bipyridine complexes,11a, 14 forming linear polymers, as a result of the folded syn conformation adopted by the bpe ligand. The torsion angle involving the four carbon atoms C3, C6, C7 and C8 (central ethyl carbon atoms and the connecting pyridyl carbon atom) is 63.7(4)°, clearly show the folding of the coordinated bpe ligand. The interchain metal–metal distance down the c axis is 9.417 Å, whereas the nearest metal–metal distance of the polymeric chains which are staggered in the ab plane is 10.361 Å. The cavity-enclosed bpe pyridyl rings are rotated by 44.7(3)° with respect to each other.
Even though structural reports of bpe ligands with transition metals are limited in number, conformational flexibility (both syn and anti) and the versatile coordination ability of the bpe ligand play vital roles in the generation of the various three-dimensional coordination networks. It is interesting to note that the isostructural Cd15 and Co16 structures [M2(μ-syn-bpe)2(μ-anti-bpe)(NO3)4]n (M⊕=⊕Cd2+, Co2+) are reported, in which two syn bpe spacers link the metal centres in a similar fashion to that reported here to create a cyclic closed structure. These macrocyles are further linked viaanti-bpe ligand spacers at the metal centres to create linear polymeric chains. In the same report on Co(II)bpe complexes, Hennigar et al.16 structurally characterized two more isomeric forms [Co(II)(μ-syn-bpe)(μ-anti-bpe)2(NO3)4]n, [Co(II)(μ-anti-bpe)2(μ-anti-bpe)(NO3)4]n, which are possible only due to the conformational flexibility of the bpe ligands. Similar syn coordination of the bpe ligand bridging the metal centres blocked at the cis positions by ethylenediamine to create a dimer with a square cavity has been reported by Fujita et al. where no counter ions or solvent molecules are encapsulated.17 Ferbinteanu et al. reported the interesting structural motif {[Co(II)(μ-anti-bpe)(anti-bpe)(syn-bpe)(H2O)2](ClO4)2·0.5(anti-bpe)·(H2O)}n,18 where the bpe moiety adopts both conformations creating a novel supramolecular architecture utilizing both coordinate and hydrogen bonding interactions. In the structural report of the neutral [Fe(bpe)2(NCS)2]n polymeric compound,19 the bpe ligands exhibit a similar type of coordination to that reported in the present investigation. Both the bpe ligands are in the syn disposition bridging the adjacent metal centres creating the linear polymeric chains, showing weak antiferromagnetic coupling through a M–(bpe)2–M bridge, which is extended via a C–H⋯S H-bonding interactions between the adjacent chains resulting in a two-dimensional sheet-like architecture. The coordinated water molecule in complex 1 makes strong H-bonding interactions with the lattice water molecule O2W and the perchlorate oxygen atom O2 where O1W acts as a donor [O1W⋯O2W⊕=⊕2.728(1) Å, <O1W–H11O⋯O2W⊕=⊕152.64(2)°, symmetry code⊕=⊕1/2⊕+⊕x, 1/2⊕+⊕y, z; O1W⋯O2⊕=⊕3.160(2) Å,<O1W–H21O⋯O2⊕=⊕162.234(2)°, symmetry code⊕=⊕1/2⊕+⊕x, −1/2⊕+⊕y, z]. Although we could not locate the H-atoms of the lattice water molecule it is making good short contacts with the perchlorate oxygens O1 and O2 (O2W⋯O1⊕=⊕2.963 Å, O2W⋯O2⊕=⊕2.939 Å). These hydrogen bonding interactions play a crucial role in the stabilization of the molecule in the crystal lattice and the alignment of the polymeric chains along the c axis creating the cationic cavity.
Conclusions
The cavity-containing cationic linear coordination polymeric network {[Co(bpe)2(H2O)2](ClO4)2·(H2O)2}n has been synthesized and characterized by various physicochemical techniques including single crystal X-ray diffraction. The metal centres are bridged via exobidendate bpe ligands from either end through both terminal nitrogen atoms to create cationic channels which are occupied by the lattice water molecules and the perchlorate anions. The structure reported here illustrates the ability of the flexible bpe ligand to create a cavity-containing coordination network. Selective guest inclusion or anion exchange properties of this class of coordination polymer are yet to be investigated.Acknowledgements
We acknowledge Dr P. K. Ghosh, Director and Dr R. V. Jasra, Deputy Director, CSMCRI, Bhavnagar for their interest and encouragement in this work.Notes and references
- (a) H. O. Stumpt, L. Ouahab, Y. Pei, D. Gajindran and O. Khan, Science, 1993, 261, 447 CrossRef PubMed; (b) J. A. Real, E. Anders, M. C. Munoz, M. Julve, T. Granier, A. Bousseksou and F. Varet, Science, 1995, 268, 265 CrossRef CAS PubMed.
- (a) M. Kondo, T. Yoshitomi, K. Seki, H. Matsuzaka and S. Kitagawa, Angew. Chem., Int. Ed. Engl., 1997, 36, 2076 CrossRef; (b) W. Mori, F. Inoue, K. Yosida, H. Nakayama, S. Takamizawa and M. Kishita, Chem. Lett., 1997, 1219 CrossRef CAS.
- (a) G. B. Gardner, D. Vekataraman, J. S. Moore and S. Lee, Nature, 1995, 374, 792 CrossRef CAS; (b) D. Vekataraman, G. B. Garner, S. Lee and J. S. Moore, J. Am. Chem. Soc., 1995, 117, 11600 CrossRef; (c) O. M. Yaghi, G. Li and H. Li, Nature, 1995, 378, 703 CrossRef CAS; (d) S. Subramanin and M. J. Zawarotko, Angew. Chem., Int. Ed. Engl., 1995, 34, 2127 CrossRef; (e) O. M. Yaghi, H. Li and T. L. Groy, Inorg. Chem., 1997, 36, 4292 CrossRef CAS PubMed; (f) R. Robinson and M. J. Zaworotko, J. Chem. Soc., Chem. Commun., 1995, 2413 RSC; (g) O. M. Yaghi and H. Li, J. Am. Chem. Soc., 1996, 118, 295 CrossRef CAS; (h) O. M. Yaghi and H. Li, J. Am. Chem. Soc., 1995, 117, 10401 CrossRef CAS.
- (a) J. S. Seo, D. Whang, H. Lee, S. I. Jun, J. Oh, Y. J. Jeon and K. Kim, Nature, 2000, 404, 982 CrossRef CAS PubMed; (b) M. Fujita, Y. J. Kwon, S. Washizu and K. Ogura, J. Am. Chem. Soc., 1995, 116, 1151 CrossRef.
- (a) S. Kitagawa and M. Kondo, Bull. Chem. Soc. Jpn., 1998, 71, 1739 CrossRef CAS and references therein.; (b) C. S. Hong and Y. Do, Inorg. Chem., 1998, 37, 4440 Search PubMed; (c) G. D. Munno, D. Armeatano, T. Poerio, M. Julve and J. A. Real, J. Chem. Soc, Dalton Trans., 1999, 1813 RSC.
- CAD4-PC Software, Version 5, Enraf-Nonius, Delft, Netherlands, 1989..
- E. I. Gabe, Y. Le Page, I. P. Charland, F. L. Lee and P. S. White, J. Appl. Crystallogr., 1989, 22, 384 CrossRef CAS.
- G. M. Sheldrick, SHELX-97. Program for Crystal Structure Determination, University of Göttingen, Germany, 1997..
- International Tables for X-Ray Crystallography, Kynoch Press, Birmingham, 1974, vol. IV. Search PubMed.
- C. K. Johnson, ORTEP, Report ORNL-3794, Oak Ridge National Laboratory, Oak Ridge, TN, USA, 1976..
- (a) Y. B. Dong, M. D. Smith, R. C. Layland and H. C. Loye, J. Chem. Soc., Dalton Trans., 2000, 775 RSC; (b) J. Lu, C. Yu, T. Niu, T. Paliwala, G. Crisci, F. Somosa and A. Jacobson, Inorg. Chem, 1998, 37, 4637 CrossRef CAS PubMed.
- A. L. Spek, PLATON 92, University of Utrecht, Utrecht, The Netherlands, 1992..
- The cavity dimensions described here are crystallographic scalar quantitities only and do not take into account the van der Wals radii of the atoms defining the cavity..
- (a) P. Losier and M. J. Zaworotko, Angew. Chem., Int. Ed. Engl., 1996, 35, 2779 CrossRef CAS; (b) L. M. Zheng, X. Fang, K. H. Li, H. H. Song, X. Q. Xin, H. K. Fun, K. Chinnakali and A. Razak, J. Chem. Soc., Dalton Trans., 1999, 2311 RSC.
- M. Fujita, Y. J. Kwon, M. Miyazawa and K. Ogura, J. Chem. Soc., Chem Commun., 1994, 1977 RSC.
- T. L. Hennigar, D. C. MacQuarrie, P. Losier, R. D. Rogers and M. J. Zaworotko, Angew. Chem., Int. Ed. Engl., 1997, 36, 972 CrossRef CAS.
- M. Fujita, S. Nagao, M. Iida, K. Ogata and K. Ogura, J. Am. Chem. Soc., 1993, 115, 1574 CrossRef CAS.
- M. Ferbinteanu, G. Marinescu, H. W. Roesky, M. Noltemeyer, H. G. Schmidt and M. Anrudh, Polyhedron, 1998, 18, 243 CrossRef.
- M. L. Hernàndez, M. G. Barandika, M. K. Urtiaga, R. Cortés, L. Lezama, M. I. Arriortua and T. Rojo, J. Chem. Soc., Dalton Trans., 1999, 1401 RSC.
| This journal is © The Royal Society of Chemistry 2001 |