DNA adducts and the total sum of at-risk DNA repair alleles in the nasal epithelium, a target tissue of tobacco smoking-associated carcinogenesis
Abstract
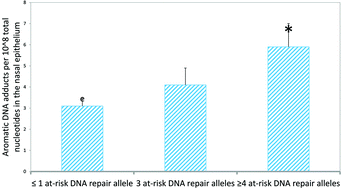
Tobacco smoking is a leading cause of death and disability. Interindividual variability in DNA adducts has been shown in subjects exposed to similar amounts of environmental carcinogens, including tobacco smoke. We have investigated the effects of smoking on DNA adducts in the nasal epithelium instead of peripheral blood in 42 volunteers, considering a panel of at-risk alleles by 32P-postlabeling and PCR. In detail, we have studied the association of DNA damage with tobacco smoke habits considering genes involved in DNA repair, including X-ray repair cross complementing protein 1 (XRCC1) Arg194Trp, XRCC protein 3 Thr241Met, and excision repair cross complementation group 2/xeroderma pigmentosum D (ERCC2/XPD) Lys751Gln polymorphisms. Then, we have analysed the combinations of the variant alleles of XRCC1 and ERCC2/XPD together with the wild type allele of XRCC3 by calculating the sum of at-risk alleles for lung cancer. DNA adducts were significantly higher in smokers with respect to nonsmokers (P < 0.001). An overall significant increase in adducts in heavy and long term smokers was found (P-values for trend <0.001, respectively). Multivariate regression analysis showed that adducts were linearly correlated to the number of cig per day (P < 0.001). Individuals with XRCC1 194Trp variant have a significant increment of DNA damage (P < 0.05), whereas XRCC3 241Met variant was inversely associated with adducts (P < 0.05). A null association was found with ERCC2/XPD. The levels of DNA adducts in participants with ≥4 at-risk alleles were two-fold increased with respect to those with one or fewer alleles (P < 0.01). A significant trend was observed (P-value for trend <0.05). Particularly, these results indicate a functional role for the XRCC1 and XRCC3 polymorphisms in genotoxic susceptibility related to the sensitivity to mutagens contained in tobacco smoke. Interindividual differences in at-risk alleles influence significantly the repair of damage in a tissue target of tobacco smoking-associated carcinogenesis. Our results support a functional role for the studied polymorphisms related to differences in cellular response to tobacco smoke mutagens. Phenotypes characterized by high levels of damage in susceptible individuals may be due to disorders in mechanisms designed to maintain cell homeostasis and DNA integrity.

 Please wait while we load your content...
Please wait while we load your content...