Tuning the supramolecular chirality and optoelectronic performance of chiral perylene diimide nanowires via N-substituted side chain engineering†
Abstract
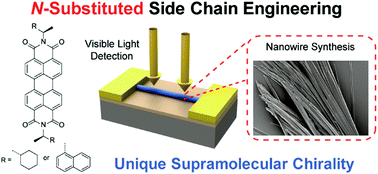
Supramolecular chirality has drawn a great deal of attention due to promising applications in chiral recognition, sensing, catalysis, and device design. Here, we studied the influence of the side chains of chiral perylene diimides (PDIs) on the supramolecular chirality and optoelectronic performance. PDIs with various N-substituted alkyl chains were synthesized and self-assembled into one-dimensional nanostructures. PDI nanowires (NWs) with less bulky alkyl chains (CPDI-Cy) exhibited the maximum electron mobility of 0.67 cm2 V−1 s−1 and showed better optoelectronic performance than those with bulkier substituents (CPDI-Naph), with three orders of magnitude improvements in photoresponsivity, photosensitivity, external quantum efficiency, and detectivity (D*). Particularly, the D* of CPDI-Cy NWs was two or three orders of magnitude higher than those of other chiral PDI nanomaterials and PDI-based polymers, and is one of the highest among chiral organic semiconductors. Judging from density functional theory calculations and molecular dynamics simulations, the higher optoelectronic performance of CPDI-Cy NWs mainly originated from their denser packing and favorable molecular arrangements for charge transport. These findings demonstrate the impact of side chain engineering on tuning the self-assembly process of PDI molecules, supramolecular chirality, and their consequent device performance.


 Please wait while we load your content...
Please wait while we load your content...