Almost complete radiationless energy transfer from excited triplet state of a dim phosphor to a covalently linked adjacent fluorescent dye in purely organic tandem luminophores doped into PVA matrix†
Abstract
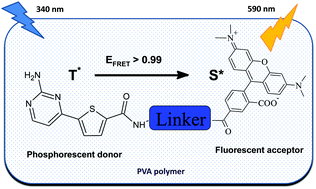
In solid PVA matrix some thiophene- and selenophene-comprising heteroaromatic compounds reveal moderate room temperature phosphorescence with emission peaks between 550 and 600 nm and possessing lifetime in millisecond range upon excitation with near-UV radiation. In tandem luminophores almost complete intramolecular interchromophore energy transfer takes place by FRET mechanism from the named phosphors in excited triplet state to adjacent covalently linked fluorescent dyes, leading to emission of light from the fluorophore. Variation of length of the linker connecting the chromophores enables tuning of luminescence lifetime in the range from 5 ms to 100 μs. The delayed emission spectrum of the tandem luminophores originates from the acceptor fluorophore. Triplet–singlet energy transfer outperforms other relaxation pathways of the excited triplet state even at 75 °C, making the luminescence lifetime temperature independent. Thus a universal strategy is provided for converting inherently dim organic phosphors into bright long-lifetime luminescence emitters. This discovery may open new avenues for construction of efficient purely organic light emitting devices and photoluminescent sensors for measurement of various analytes.
- This article is part of the themed collection: Functional Organic Materials for Optoelectronic Applications


 Please wait while we load your content...
Please wait while we load your content...