Predicted photovoltaic performance of lead-based hybrid perovskites under the influence of a mixed-cation approach: theoretical insights
Abstract
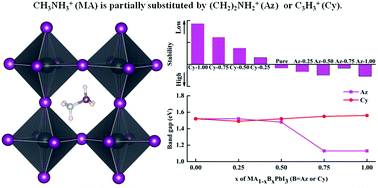
Hybrid organic–inorganic halide perovskite solar cells have recently attracted much attention because of their highly efficient photovoltaic performance. However, perovskite solar cells suffer from intrinsic instabilities, which limit their commercial applications. In this study, we partially substituted the methyl-ammonium (MA) cation in MA lead iodide perovskite (MAPbI3) and investigated the geometric structures and electronic and optical properties by first-principles calculations to determine their thermodynamic stabilities and optical band gaps. The results suggest that substitution of the Az cation in the A-site can enhance the stability of the MA1−xAzxPbI3 system. However, the formation energies indicate that the MA1−xCyxPbI3 series is less stable. The estimated power conversion efficiency of MA0.50Az0.50PbI3 is 23.06%, which is higher than that of MAPbI3. Notably, MA1−xAzxPbI3 (0.50 < x < 0.75) is predicted to be a promising candidate for photovoltaic applications because it has the highest theoretical power conversion efficiency, which is attributed to its optimal band gap. These results suggest that an organic cation mixing strategy can be used to tune the stability, band gap, and photovoltaic performance of hybrid perovskites, which will be particularly useful for designing light-harvesting materials for perovskite solar cells.


 Please wait while we load your content...
Please wait while we load your content...