Structure–property relationship of D–A type copolymers based on phenanthrene and naphthalene units for organic electronics†
Abstract
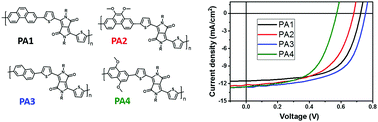
Four donor–acceptor (D–A) type conjugated polymers (PA1, PA2, PA3 and PA4) based on phenanthrene and naphthalene as the donating units with or without dimethoxy substitution were synthesized for organic field effect transistors (OFETs) and bulk-heterojunction organic photovoltaics (OPVs). Dimethoxy substituents have significant effects on the optical, electrochemical, charge transport and photovoltaic properties depending on the donor-polyaromatic (PA) compounds. The optical band gaps of these PA-based copolymers from the smallest to the largest are as follows: 1.52 eV (1,5-dimethoxy substituted naphthalene (PA4)), 1.59 eV (unsubstituted naphthalene (PA3)), and 1.63 eV (unsubstituted phenanthrene (PA1), and substituted 9,10-dimethoxy phenanthrene (PA2)). While the values vary depending on the compounds, both PA2 and PA4 are found to have higher highest occupied molecular orbital (HOMO) energy levels than those of PA1 and PA3 due to the electron donating nature of dimethoxy substituents. The PA based copolymers without dimethoxy substituents showed highly balanced ambipolar behavior with ∼1 cm2 V−1 s−1, whereas the electron mobility of dimethoxy modified PA (MeOPA) based copolymers was suppressed. The inverted bulk heterojunction OPVs based on PA1 and PA3 exhibited power conversion efficiency (PCE) as high as 5.3% and 5.8%, respectively. The PCEs of PA copolymer-based OPV devices were mainly affected by an increase in the open circuit voltage rather than by the photocurrent or fill factor.


 Please wait while we load your content...
Please wait while we load your content...