Exploring the possible existence of oxygen-bridged planarized 4-aminopyridine: promising structure, charge transport and nonlinear optical properties†
Abstract
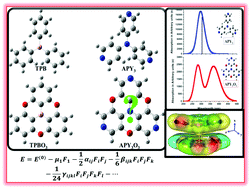
In the present quantum chemical investigation, for the first time, we shed light on the promising structure, charge transport and nonlinear optical properties (NLO) of oxygen-bridged planarized 4-aminopyridine (APY3O3). Density functional theory (DFT) and time dependent DFT methods are used to elucidate the novel structure, optical and NLO properties of open 4-aminopyridine (APY3) as well as oxygen-bridged closed APY3O3 systems. The calculated third-order polarizability 〈γ〉 for APY3 and APY3O3 is found to be 76.85 and 89.16 × 10−36 esu with the B3LYP method and the 6-311G** basis set, respectively, which is ∼10 and 12 times larger than that of para-nitroaniline (a prototype NLO molecule). Calculation of the electron affinity and reorganization energy suggests that good electron transfer properties are feasible by changing the geometry from open APY3 to closed APY3O3 through oxygen bridging. Interestingly, the configuration of a dimer having APY3O3 and oxygen-bridged triphenylborane (TPBO3) monomers demonstrates remarkable enhancement in NLO response through a novel non-covalent intermolecular charge transfer process. The present investigation not only evokes scientific interest regarding the promising structure of APY3O3 but also highlights its several optical and NLO properties that can be beneficial for its use as an advanced functional material in modern hi-tech applications.


 Please wait while we load your content...
Please wait while we load your content...