Dye-sensitized solar cells using cobalt electrolytes: the influence of porosity and pore size to achieve high-efficiency†
Abstract
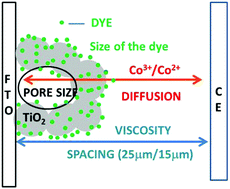
Diffusion impediments in redox shuttles are a major limitation to realizing high power conversion efficiencies (PCEs) in dye sensitized solar cells (DSCs) using cobalt complexes. Mass transport limitations are strongly dependent on the pore size and the porosity of mesoporous TiO2 films. Herein, we examine the major factors responsible for mass transport limitations and identify ways to minimize them, achieving highly efficient cobalt based DSCs. Depending on the electrolyte and dye used, the pore size and the porosity of the TiO2 films must be modulated to facilitate diffusion of cobalt complexes through pores. The influence of porosity, pore size and viscosity of the electrolyte used on the performance of DSCs with different maximum obtainable JSCs are studied in detail. These effects were studied using two dye solutions, Y123 (0.2 mM) and a cocktail solution of SM342/Y123 (0.2 mM each, 7 : 1). The JSC is affected primarily by lowered pore sizes as a result of the TiCl4 post-treatment. Transient photocurrent decay measurements were performed on devices featuring photoanodes fabricated with differing porosities and pore sizes. This study indicates that in cells featuring only Y123, recombination rates decreased with decreasing porosity of TiO2 films (i.e. higher TiCl4 concentrations used in their post-treatment). Conversely, in DSCs fabricated with the SM342/Y123 dye cocktail, an intermediate TiCl4 post-treatment concentration (20 mM) afforded the lowest recombination rate, resulting in the preservation of a high open-circuit potential (VOC). It was found that this intermediate concentration regime is optimal in order to achieve an efficiency of over 12.7% at full sun intensity with the SM342/Y123 dye cocktail. Finally, it was concluded that the porosity and pore size must be modulated for different dyes and electrolytes to minimize diffusion limitations of the redox species.


 Please wait while we load your content...
Please wait while we load your content...