Co-titration of AgNO3 and HAuCl4: a new route to the synthesis of Ag@Ag–Au core–frame nanocubes with enhanced plasmonic and catalytic properties†
Abstract
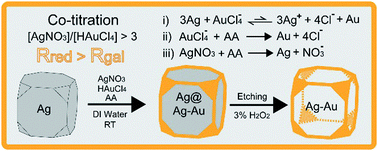
We report a new strategy for the synthesis of Ag@Ag–Au core–frame nanocubes by co-titrating AgNO3 and HAuCl4 concomitantly into an aqueous suspension of Ag nanocubes in the presence of ascorbic acid (AA) and poly(vinyl pyrrolidone) at room temperature. When the molar ratio of AgNO3 to HAuCl4 was larger than three, we discovered that the added Ag+ ions could effectively push the galvanic replacement reaction between Ag nanocubes and HAuCl4 backward and thus inhibit it, making it possible to achieve the co-reduction of the two precursors by AA without involving any galvanic replacement. By increasing the volumes of the two co-titrated precursors, we validated that the added AgNO3 and HAuCl4 were completely reduced to Ag and Au atoms, respectively, followed by their co-deposition onto the edges, corners, and then side faces of the Ag nanocubes in a fashion similar to seeded growth. As a result, the co-titration process offers an exquisite control over the relative amounts of Ag and Au atoms being deposited by simply varying the feeding ratio between the two precursors. We also demonstrated that the Ag@Ag–Au core–frame nanocubes exhibited unique plasmonic properties. Upon etching of the Ag templates from the core–frame nanocubes by an oxidant, we obtained Ag–Au nanoframes that could serve as an active catalyst for the reduction of 4-nitrophenol by NaBH4.
- This article is part of the themed collection: 2015 Journal of Materials Chemistry C Hot Papers

 Please wait while we load your content...
Please wait while we load your content...