Efficient Pt(ii) emitters assembled from neutral bipyridine and dianionic bipyrazolate: designs, photophysical characterization and the fabrication of non-doped OLEDs†
Abstract
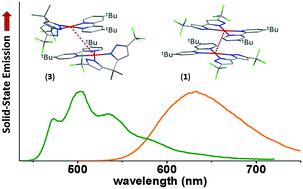
Potential dianionic chelates, 5,5′-bis(trifluoromethyl)-2H,2′H-3,3′-bipyrazole (bipzH2) and 5,5′-(1-methylethylidene)-bis(3-trifluoromethyl-1H-pyrazole) (mepzH2), were synthesized from Claisen condensation employing ethyl trifluoroacetate with 2,3-butanedione and with 3,3-dimethyl-2,4-pentanedione, followed by hydrazine cyclization. These chelates were then utilized in the preparation of four emissive Pt(II) metal complexes [Pt(tbbpy)(bipz)] (1), [Pt(msbpy)(bipz)] (2), [Pt(tbbpy)(mepz)] (3) and [Pt(msbpy)(mepz)] (4), where tbbpy and msbpy represent 4,4′-di-t-butyl-2,2′-bipyridine and 4,4′-dimesityl-2,2′-bipyridine, respectively. Single crystal X-ray structural analyses of 2 and 3 were executed to unveil the basic coordination geometry around the Pt(II) center as well as the π–π stacking interaction in the solid state. These complexes are essentially non-emissive in solution (Q. Y. = 0.2–0.4%), but are highly luminescent in the solid state with QY of 52% and 83% and τobs of 368 ns and 8.37 μs for 1 and 3, respectively. Their photophysical properties were measured and discussed on the basis of computational approaches. For applications, non-doped organic light emitting diodes (OLEDs) were fabricated using 1 and 3 as emitters, exhibiting red-orange emission with a maximum luminance of 43 000 cd m−2, an EQE of 19.0%, a CE of 21.0 cd A−1 and a PE of 15.5 lm W−1, and yellow emission with a maximum luminance of 5100 cd m−2, an EQE of 7.1%, a CE of 21.0 cd A−1 and a PE of 11.3 lm W−1, respectively. The particularly higher OLED efficiencies of 1versus3 highlight the design principle of Pt(II) based phosphors, particularly for the fabrication of a non-doped OLED architecture.

 Please wait while we load your content...
Please wait while we load your content...