Manipulation of the band gap and efficiency of a minimalist push–pull molecular donor for organic solar cells†
Abstract
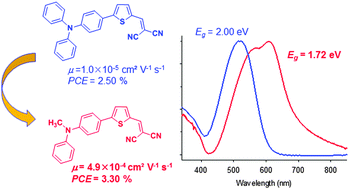
A small push–pull compound involving a methyl-diphenylamine donor block connected to a dicyanovinyl acceptor group through a 2,5-thienyl spacer has been synthesized (1). Comparison with a reference compound containing a triphenylamine block (2) shows that the replacement of a phenyl group by a methyl group has limited effects on the electronic properties of the molecule but induces major changes in the structure and electronic properties of the resulting material such as a ∼0.30 eV reduction of the optical band gap and fifty-fold increase of the hole-mobility. Results of X-ray diffraction on single crystals show that the replacement of a phenyl by a methyl group results in a change from head-to-tail to face-to-face arrangement of the dipolar molecules in the crystal. A preliminary evaluation of the potential of the new compound as a donor material in basic bilayer cells of 0.28 cm2 with C60 as the acceptor reveals a ca. 33% increase of the power conversion efficiency compared to the reference compound.

 Please wait while we load your content...
Please wait while we load your content...