Nanostructured ion gels from liquid crystalline block copolymers and gold nanoparticles in ionic liquids: manifestation of mechanical and electrochemical properties†
Abstract
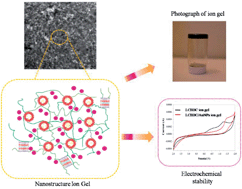
We examine the influence of confining gold nanoparticles on the overall nanoscale morphology, mechanical and electrochemical properties of nanocomposite ion gels. Stimuli-responsive ion gels are generated via the synthesis of gold nanoparticles (AuNPs) within a thiol-functionalized liquid crystalline brush-like block copolymer (LCBBC) and subsequent gelation in ionic liquids. AuNPs are prepared by in situ reduction of the gold ions and stabilized by direct anchoring through coordination bonds with the thiol-functionalized LCBBC. The resulting LCBBC/AuNP nanocomposite comprises a hierarchical structure in which polymer-coated AuNPs are dispersed in a microsegregated LCBBC matrix that further contains both liquid crystalline (LC) and block copolymer ordering. More importantly, this LCBBC/AuNP nanocomposite is solubilized in an ionic liquid (IL), 1-butyl-3-methylimidazolium hexafluorophosphate, to form a nanocomposite ion gel. At 5 wt% concentration, LCBBC/AuNP nanocomposite ion-gel exhibits interesting characteristics including excellent mechanical strength (∼103 Pa), good optical properties with higher ionic conductivity (∼10−2 S cm−1) and long-term electrochemical stability over a larger potential range compared to the plain LCBBC ion gel. Thus, nanocomposite ion gels present tunable optical, thermal and mechanical properties by virtue of their polymer architecture, morphology and functionality. These are versatile and modular hybrid materials to design nanocomposite ion gels with multiple functionalities for applications in electrochemical devices, photonics, and opto-electronics.

 Please wait while we load your content...
Please wait while we load your content...