Controlling morphology and charge transfer in ZnO/polythiophene photovoltaic films†
Abstract
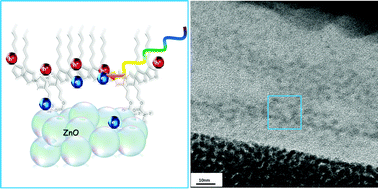
The organic–inorganic interfacial chemical composition and interaction have a critical influence on the performance of corresponding hybrid photovoltaic devices. Such interfaces have been shown to be controlled by using surfactants to promote contact between the organic electron-donating conjugated polymer species and the inorganic electron-accepting metal oxides. However, the location of the surfactant at the organic–inorganic interface often hinders donor–acceptor charge transfer and limits the device performance. In this study we have replaced the conventional surfactant with an optically and electronically active amphiphilic polythiophene that both compatibilizes the conjugated polymer and metal oxide, and plays an active role in the charge generation process. Specifically, a polythiophene with hydrophilic urethane side-groups allows the formation of a fine continuous ZnO network in P3HT with an organic–inorganic interfacial chemical interaction and a high surface area. A combination of FTIR, absorption and photoluminescence life-time spectroscopies yields insights into the composition and nano-scale interaction of the components, while high-resolution electron microscopy reveals the morphology of the films. The control over morphology and electronic coupling at the hybrid interface is manifested in photovoltaic devices with improved performances.

 Please wait while we load your content...
Please wait while we load your content...