A novel ratiometric SERS biosensor with one Raman probe for ultrasensitive microRNA detection based on DNA hydrogel amplification†
Abstract
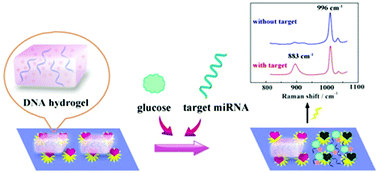
In this work, a novel ratiometric SERS biosensor with only one Raman probe was fabricated in combination with the DNA hydrogel-captured glucose oxidase (GOx) amplification method to realize an accurate and sensitive assay for microRNA (miRNA) 122, which could overcome the complex operational process and chemical waste issues of the traditional ratiometric approach with two different signal probes. Here, 3-mercaptophenylboronic acid (3-MPBA) as a standard reference with a unique Raman peak at 996 cm−1 was first connected to silica@Au nanoflower (Si@AuNF) SERS substrates. When the DNA hydrogel containing GOx was opened by the released DNA (R) from the target miRNA-induced cycle amplification process, GOx could be released from the DNA hydrogel. Therefore, GOx could catalyze the oxidation of glucose to produce H2O2 on Si@AuNFs. Subsequently, the acquired H2O2 could further react with 3-MPBA to produce 3-hydroxythiophenol (3-HTP) with a new Raman peak at 883 cm−1. During this time, the intensity of the peak at 996 cm−1 almost remained the same, which could act as a reference standard. The intensity ratio of 883 cm−1 to 996 cm−1 increased with the increase in the concentration of target miRNA 122, thus achieving quantitative detection of miRNA 122. As a consequence, our SERS biosensor could sensitively detect miRNA 122 from 10 aM to 100 pM, and the detection limit was 7.75 aM. Our strategy adopts a novel ratiometric method with one Raman probe to detect miRNA, opening a new avenue for the detection of trace amounts of biological samples with high sensitivity and accuracy.


 Please wait while we load your content...
Please wait while we load your content...