Electrochemical-mediated gelation of catechol-bearing hydrogels based on multimodal crosslinking†
Abstract
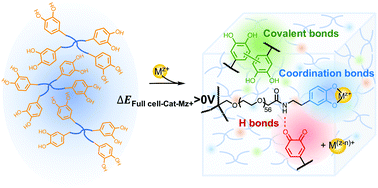
Catechol-bearing polymers form hydrogel networks through cooperative oxidative crosslinking and coordination chemistry. Here we describe the kinetics of cation-dependent electrochemical-mediated gelation of precursor solutions composed of catechol functionalized four-arm poly(ethylene glycol) combined with select metal cations. The gelation kinetics, mechanical properties, crosslink composition, and self-healing capacity is a strong function of the valency and redox potential of metal ions in the precursor solution. Catechol-bearing hydrogels exhibit highly compliant mechanical properties with storage moduli ranging from G′ = 0.1–5 kPa depending on the choice of redox active metal ions in the precursor solution. The gelation kinetics is informed by the net cell potential of redox active components in the precursor solution. Finally, redox potential of the metal ion precursor can differentially alter the effective density of crosslinks in networks and confer properties to hydrogels such as self-healing capacity. Taken together, this parametric study generates new insight to inform the design of catechol-bearing hydrogel networks formed by electrochemical-mediated multimodal crosslinking.
- This article is part of the themed collection: Hydrogel properties and applications


 Please wait while we load your content...
Please wait while we load your content...
