Design and self-assembly of albumin nanoclusters as a dynamic-covalent targeting co-delivery and stimuli-responsive controlled release platform†
Abstract
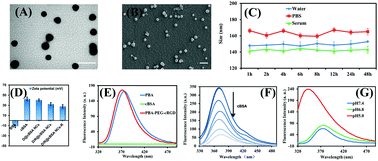
Stimuli-responsive nanomaterial-based drug delivery systems that are able to actively target the tumor microenvironment, enhance intratumoral accumulation and release drugs at target sites are attractive therapeutic platforms at present. Here, the first approach that uses proteins to construct dynamic-covalent targeting stimuli-responsive cationic albumin nanoclusters for the highly efficient co-delivery of siRNA and DOX in vitro and in vivo has been developed and achieved considerable efficacy in combination therapy for the growth, migration and invasion of cancer. In this study, the critical strategy of ‘dynamic-covalent targeting’, as previously mentioned, was developed for the active accumulation of drugs at target sites by fabricating a reversible PEG corona using a pH-sensitive borate ester so as to facilitate the formation of a compact complex and the efficient release of siRNA with or from the nanocluster. In addition, after internalization, the nanocluster can dissociate into cationic albumin via intracellular reduction in an acid environment to achieve stimuli-responsive triggered drug release. As a result, the nanocluster for the co-delivery of DOX and VEGF-siRNA exhibits highly efficient capacity for gene silencing and apoptosis-inducing ability and also markedly suppresses the migration and invasion of cancer cells. Most importantly, it displayed significant combination effects against tumor growth with minimal damage to major organs and provides an effective approach for the safe and efficient delivery of multiple cargoes at target sites.


 Please wait while we load your content...
Please wait while we load your content...