The impact of light irradiation timing on the efficacy of nanoformula-based photo/chemo combination therapy†
Abstract
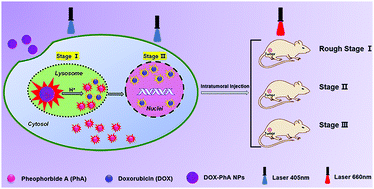
Photo/chemo combination therapy has been demonstrated to be a generally more powerful strategy for treating cancers than a single treatment modality. However, it is unknown whether the timing of light irradiation has any impact on therapeutic efficacy. We designed a carrier-free and self-monitoring nanodrug to monitor the entire dual-drug release profile and determined the impact of photodynamic therapy (PDT) at different time points. The designed nanodrug consists of the chemotherapeutic doxorubicin (DOX) and the photosensitizer pheophorbide A (PhA). The drugs form a fluorescence resonance energy transfer (FRET) pair (DOX transferring energy to PhA) when present at a precise ratio in the combination nanodrug. Due to the FRET effect, the DOX-PhA nanoparticles (NPs) show PhA fluorescence in a normal pH environment (such as cytoplasm). However, the FRET effect is lost when the NPs are disassembled in an acidic environment (such as lysosomes), and the DOX fluorescence is recovered. By real-time fluorescence variation monitoring, we determined the key time points when the drugs reached various subcellular locations, which helped us to determine the PDT-triggering time points and investigate the impact on the therapeutic effect in the combination therapy. Furthermore, the PDT was triggered at these established time points both in vitro and in vivo, which revealed that the best PDT-triggering time point in the combination therapy was achieved after nuclear entry of DOX. The study suggests that the optimization of combination therapy, not only photo/chemo but also chemo/chemo combination therapy, may require not only a controlled drug ratio but also a controlled drug release profile and target arrival time.


 Please wait while we load your content...
Please wait while we load your content...