Overcoming blood–brain barrier by HER2-targeted nanosystem to suppress glioblastoma cell migration, invasion and tumor growth†
Abstract
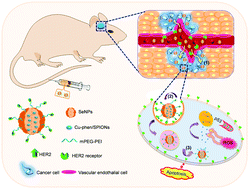
Efficient therapy of glioblastoma remains a big clinical challenge due to the existence of a blood–brain barrier (BBB) that prevents the delivery of anti-cancer drugs to the brain. In this study, a HER2 antibody-conjugated selenium nanoparticle platform was rationally designed and synthesised and was found to be capable of overcoming the BBB efficiently and delivering anti-cancer cargoes precisely to brain tissues. The BBB-overcoming efficacy of the nanosystem (HER2@NPs) was evaluated using in vitro cell co-culture model and in vivo mouse model. The results demonstrated that HER2 functionalization effectively enhanced BBB permeability of the NPs, which could significantly inhibit the growth of U251 glioblastoma tumor spheroids. Examination of the action mechanisms revealed that HER2@NPs entered the cancer cells through receptor-mediated endocytosis and then triggered DNA damage-mediated p53 signalling pathways. Moreover, by using superparamagnetic iron oxide nanoparticles (SPIONs) as a probe in a clinically used magnetic resonance imaging (MRI) system, we found that HER2@NPs effectively deliver SPIONs into the brain. Taken together, this study provides a cancer-targeting nanosystem for the delivery of anti-cancer drugs and MRI contrast agents overcoming the BBB to enable a precise future theranosis of malignant glioma in humans.


 Please wait while we load your content...
Please wait while we load your content...